Utility of Intraoperative Cortico-Cortical Evoked Potentials for the Evaluation of Language Function During Brain Tumor Resection
A B S T R A C T
The cortico-cortical evoked potentials (CCEP) are a relatively recent intraoperative monitoring tool that has shown high reliability in detecting the functional language area and its connections. In this work we describe a patient with a tumor located in the language area who underwent tumor resection. A neurophysiological intraoperative mapping of the frontal and posterior language area with the anesthetized patient was carried out. Then the patient was awaked. To demonstrate the relationship between language function and CCEP, the Ojemann’s stimulation technique was used in the same electrodes were CCEPs were obtained. An alteration in the emission of language had been found on the electrodes where CCEP was obtained and in adjacent electrodes. During tumor resection we used a double check for language function: neurological evaluation and CCEPs. We obtained a biunivocal relationship between warning criteria in the CCEP and immediate language impairment on neurological examination. We always observed a significant decrease in the CCEP associated with language impairment. The CCEP are able to obtain information in real time with a good spatial resolution of the language function. Future studies should be carried out in order to analyse the robustness of this technique to perform it in anesthetized patients.
Keywords
Anaesthetized surgery, awake surgery, cortical mapping, cortico-cortical evoked potentials, intraoperative neurophysiological, monitoring
Introduction
Cortico-cortical evoked potential (CCEP) is a promising technique that has shown to be an extremely useful tool for detecting functional connections in cortical networks [1, 2]. This method uses single pulse electrical stimulation (SPES) applied directly to the cortex through subdural electrodes to elicit CCEPs picked up several centimeters away from the remote cortex [3, 4]. During extra-operative evaluation, this method has been able to map functional cortical networks, including language network between the frontal language area (FLA) and the parieto-temporal language area (PLA), motor-sensory system, limbic system, visual system, and seizure network [2-10]. Besides, recent studies have demonstrated that intraoperative CCEP is a feasible and reproducible tool for monitoring the language system during the resection of brain tumors [2]. CCEP has been described as a neurophysiological monitoring technique in patients with brain tumors in language areas; in most cases awake craniotomies were performed [2, 11].
In this work we describe the use of CCEP as mapping and monitoring in a patient during the surgery for a brain tumor placed near the language area in the language-dominant left hemisphere. We have used a double check to monitor the language function: neurological evaluation with the patient awake in parallel with CCEP. In this way, we have demonstrated a univocally relation between language function and CCEPs.
Case Report
We present the case of a 31-year-old man right-handed with history of morbid obesity, consulting for paroxysmal episodes of numbness of the face and tongue right-side, with a daily frequency in the last month. Also refers occasionally tinnitus and auditory hallucinations. A 1.5-T magnetic resonance imaging (MRI) (General Electric®, Fairfield, CT, USA) showed a low-grade left temporal and insular glioma. Also, the patient was evaluated by neurological, neuropsychological, and psychiatric assessments. No language alterations were found, and the paroxysmal episodes were diagnosed as epileptic seizures and treatment with levetiracetam was started.
The craniotomy and surgery were guided by a neuronavigational system (Brainlab, Feldkirchen, Germany). The tumor was removed using microsurgical techniques, bipolar coagulation, and ultrasonic aspiration. The patient was operated in a sleep-awake-sleep schedule. During the sleep period, general anaesthesia was induced by bolus of propofol and remifentanil and maintained with continuous perfusion (propofol 6.5 mg/kg/h and remifentanil 0.1 µ/kg/min) during the period of cortical mapping (Figure 1). Later, the patient was awaked to evaluate the language function neurologically and by CCEP.
Cortical mapping aims to identify all the functional areas near the tumor and was performed under general anaesthesia. We use a 32 channels multimodal intraoperative neurophysiological monitoring (IONM) system (Elite, Cadwell®, Kennewick, Washington, USA). The next neurophysiological tests were performed:
i. Electrocorticography (ECoG), through a grid of 4×5 electrodes of 1.3 mm in diameter and 1 cm center-to-center (Ad-Tech, Racine, Wisconsin, USA), was used to monitor stimulation triggered epileptiform discharges during the direct current stimulation (DCS). Bandwidth 1.5-1,000 Hz notch off.
ii. The pre- and postcentral gyri and the Central Sulcus (CS) were determined via somatosensory evoked potentials (SSEP) phase reversal. Trains of 300 pulses at 7.1 Hz, 200 µs in duration and 20 mA intensity were delivered at contralateral median nerve, bandwidth 10-1,500 Hz, notch off. Recording through subdermal electrodes was performed at Erb’s point and cortical somatosensory-evoked potentials (cSSEP) were directly recorded from subdural grid.
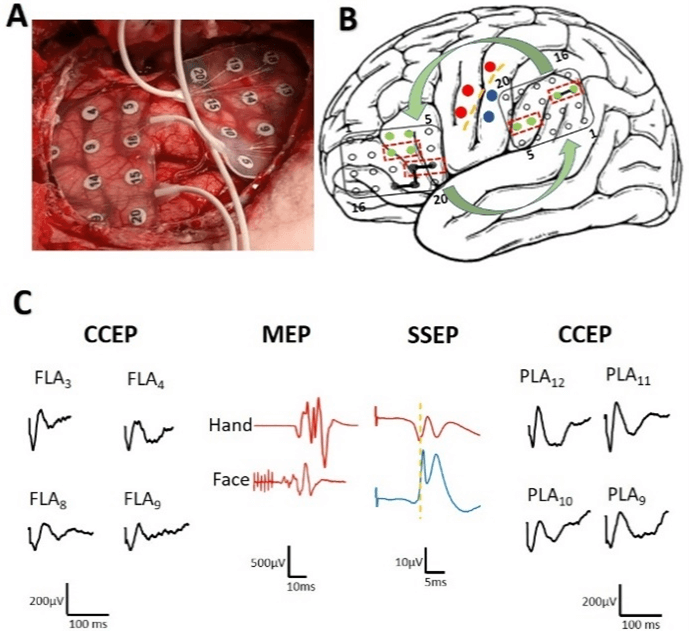
iii. The primary motor cortex (PMC) was identified by DCS using constant-current trains of 6 bi-phasic pulses of 200 μs in duration/phase at 500 Hz (high frequency technique) with intensities between 4-30 mA [12]. Motor-evoked potentials (MEPs) were recorded through pairs of subdermal electrodes (12/18 mm, SGM, Ljubiceva, Croatia) inserted into the muscles of the right side of the body, bandwidth 50-3,000 Hz, notch off, 10 ms/division time-base. The PMC of the hand and face was identified (Figure 1).
iv. Functional language areas were addressed by CCEPs. Two 4 × 5 electrodes grids were located in the FLA and PLA. DCS was applied using SPES method (cathodic monophasic pulse 1 ms duration) on two adjacent electrodes. The stimulus intensity increased stepwise, at 5 mA increments, from 5 mA until the effect was achieved or a maximum of 20 mA was obtained without response (negative stimulation). Bandwidth recording 10-1,500 Hz, notch off. The mapping started by stimulating the area immediately anterior to the PMC of the face (FLA) and recording the PLA. At least three trials were performed separately to confirm the reproducibility of the responses. A positive response was considered if a large N1 peak in the electrodes of the PLA was obtained (higher 200 µV). Then we use electrodes of the PLA grid with CCEP response to stimulate and recording with the grid at FLA to confirm bidirectionality.
It's quite important to keep in mind that ECoG was performed during all the period that DCS was done, to properly monitor excitability of the cortex. After mapping of eloquent areas, the patient was awaked. To demonstrate the relationship between language function and CCEP, the Ojemann’s stimulation technique (low-frequency stimulation) was used in the same electrodes were CCEPs were obtained: 50-Hz trains, 4 seconds duration, 400 μs pulse-width [12, 13]. We found an alteration in the emission of language when we stimulate electrodes in which CCEP had been found and in adjacent electrodes at currents between 4-6 mA (Figure 1). Neurological evaluation of language function was used by means or nomination, repetition, and series of words.
Figure 1: Intraoperative neurophysiological mapping. A) Picture showing the arrangement of the grids. B) Diagram of the grids with the electrodes where a CCEP response was obtained (green) and that of the electrodes that, when stimulated, produced a clinical alteration of language (red dashed line). The pairs of electrodes that, when stimulated, evoke a PECC are shown with a black bar and their directionality with arrows. C) The results of the mapping are displayed. Left: CCEP of the grid in the anterior region (FLA). Maximum amplitude of 258.1 µV and onset peak latency of 30.6 ms (FLA3). Middle: sensory-motor mapping with the MEP of the hand and face and the phase reversal of the PESS. Right: CCEP of the posterior grid (PLA), maximum amplitude 378.9 µV and onset peak latency of 37.5 ms (PLA11).
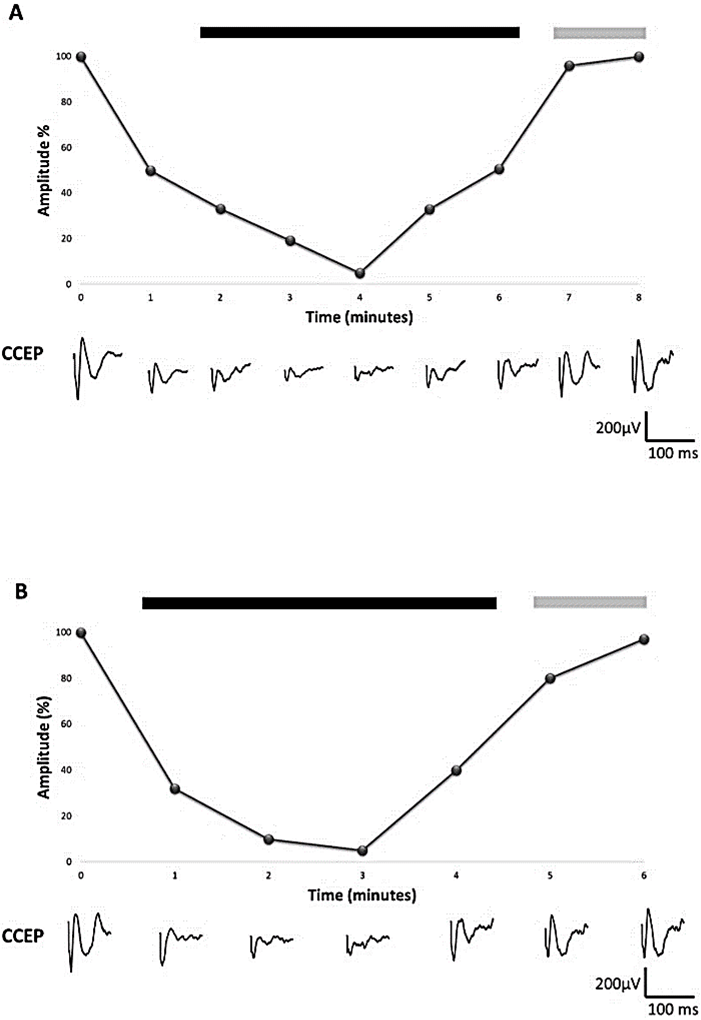
We obtained responses in four electrodes of the PLA grid (PLA9-12), amplitude 295.4-361.2 µV. The onset peak latency ranged between 36.7 to 40.6 ms. SPES stimulation at PLA11-12 electrodes induced responses in four electrodes of the FLA grid (FLA3,4,8,9), amplitude 200.0-258.1 µV and onset peak latencies 30.1-39.3 ms. (Figure 1). During most of the resective period of surgery we used a double check for language function: neurological evaluation and CCEPs. This fact allowed us to demonstrate a biunivocal relationship between warning criteria in the CCEP and an immediate language impairment on neurological examination (n = 12) [11]. Furthermore, after a language alteration was firstly observed, a significant decrease was observed in the subsequent CCEP (n = 7). We never observed language impairment without CCEP decrease. When language impairment occurred, the neurosurgeon was advised and the necessary corrective measures were taken (temporally stop resection, warm serum irrigation), after both the language function and CCEP amplitude recovered (Figure 2).
Figure 2: Monitoring of CCEP during tumor resection. A graph is displayed with the percent change in amplitude over time. The panel below shows the CCEPs during each of the times. A) Decrease in the amplitude of the CCEP is observed (50%) that is followed by an alteration in the emission of language (black bar) that persists until the potential recovers at least 90 % of amplitude (gray bar). B) Reverse situation in which the clinical alteration of the language was first evidenced (negative bar) and was confirmed with a fall in the amplitude of the CCEP (>70%). The recovery was also reversible after the corrective measures taken (gray bar).
During the last part of tumor resection, the patient was too tired and was asleep again. During all this period IONM of language function was performed only through CCEPs. In the immediate postoperative period, the patient was asymptomatic. At six months of evolution he was still asymptomatic and free of epileptic seizures. The control MRI demonstrated a partial resection of the tumor.
Discussion
In this work we have shown that CCEP is a reliable tool to identify and monitor language function during resection of language-related cerebral structure. This technique can be performed with the patient asleep, since, as our case illustrates the mapping with the anesthetized patient corresponds in a high degree with the cortical areas of language function, as demonstrated the presence of language alterations with Ojemann’s technique when the patient was awake. This strong relationship between language function and CCEPS was even enhanced with the temporal association between CCEP amplitude decrease and clinical language impairment. This is an important issue; since it suggests that this procedure can be performed in anesthetized patients, in addition to provide new insights into the neurophysiological organization of speech function.
It's of great relevance to remind that patient was epileptic. Therefore, there is a real increased risk of seizures during cortical electrical stimulation. Continuous performance of ECoG can avoid the appearing of seizures precociously detecting post-discharge or, even, the first phases of a seizure [12]. So far, CCEP have been carried out as a complementary technique for clinical examination and imaging techniques, but not as only tool to monitoring language function during brain surgery [2, 5, 6, 11].
We expected to observe changes in CCEP features by the effect of anaesthesia; however, we did not find any change in the morphology or latency of the CPEC with the patient anesthetized or awake. The question of whether anesthetic drugs can change the characteristics of potentials is still a matter of debate, since until now the technique has been performed in patients who have been awake or asleep throughout the procedure [11].
The main advantage of CCEP is that access to functional connectivity in real time and with a good spatial resolution, unlike diffusion tensor image (DTI) that although depict the connections between two areas, cannot resolve neither the function nor the direction of functional connectivity. Not to mention that in patients with structural lesions the functional tract can be distorted or interrupted due to brain edema or infiltration of the tumor [14]. Also, functional magnetic resonance imaging (fMRI) provides dynamic understanding of cortical function, but it does not indicate the actual dynamics of information flow.
There are some limitations in the procedure described. To achieve the widest possible coverage with the electrodes, craniotomy must be widespread, besides operating time can be extended [11, 15]. However, this flaw could be saved if, as we illustrated in our case, a motor mapping is performed first for better orientation and the procedure is carried out without waking up the patient. The limitations of awake surgery must be considered seriously. During such surgeries, the awake patient is maintained with the head fixed and covered with cloth and may be kept awake for several hours. Hence, patients must have both adequate cognitive function and the emotional maturity necessary to withstand such an environment. Nevertheless, use in mentally handicapped patients remains problematic or impossible [16].
It’s a medical obligation to offer the best treatment at hand with the lowest degree of annoyance to the patient. We believe that CCEP can fit this mandatory in a perfect way when language function is compromised during a brain surgery. Although we are conscious that much more clinical and scientific work must be done before accepting this possibility as the gold standard technique for this kind of surgery. New studies with intraoperative CCEP in anesthetized patients are necessary to analyse the robustness of this technique.
Conflicts of Interest
None.
Funding
This work was financed by a grant from the Ministerio de Sanidad FIS PI17/02193 and was partially supported by FEDER (Fonds Europeen de Developpement Economique et Regional).
Acknowledgements
None.
Article Info
Article Type
Case ReportPublication history
Received: Mon 09, Nov 2020Accepted: Fri 20, Nov 2020
Published: Thu 10, Dec 2020
Copyright
© 2023 Jesús Pastor Gómez. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2020.12.09
Author Info
Lorena Vega-Zelaya Marta Navas Pilar Martín Jesús Pastor Gómez
Corresponding Author
Jesús Pastor GómezClinical Neurophysiology, Hospital Universitario de la Princesa, Madrid, Spain
Figures & Tables
References
- Kunieda T, Yamao Y, Kikuchi T, Matsumoto R (2015) New Approach for Exploring Cerebral Functional Connectivity: Review of Cortico-cortical Evoked Potential. Neurol Med Chir 55: 374-382. [Crossref]
- Yamao Y, Suzuki K, Kunieda T, Matsumoto R, Arakawa Y et al. (2017) Clinical impact of intraoperative CCEP monitoring in evaluating the dorsal language white matter pathway. Hum Brain Mapp 38: 1977-1991. [Crossref]
- Valentin A, Anderson M, Alarcón G, Seoane JJG, Selway R et al. (2002) Responses to single pulse electrical stimulation identify epileptogenesis in the human brain in vivo. Brain 125: 1709-1718. [Crossref]
- Lacruz ME, Seoane JJG, Valentin A, Selway R, Alarcón G (2007) Frontal and temporal functional connections of the living human brain. Eur J Neurosci 26: 1357-1370. [Crossref]
- Matsumoto R, Nair DR, LaPresto E, Najm I, Bingaman W et al. (2004) Functional connectivity in the human language system: a cortico-cortical evoked potential study. Brain 127: 2316-2330. [Crossref]
- Matsumoto R, Nair DR, Ikeda A, Fumuro T, Lapresto E et al. (2012) Parietofrontal network in humans studied by cortico-cortical evoked potential. Hum Brain Mapp 33: 2856-2872. [Crossref]
- Swann NC, Cai W, Conner CR, Pieters TA, Claffey MP et al. (2012) Roles for the pre-supplementary motor area and the right inferior frontal gyrus in stopping action: electrophysiological responses and functional and structural connectivity. Neuroimage 59: 2860-2870. [Crossref]
- Enatsu R, Martinez JG, Bulacio J, Kubota Y, Mosher J et al. (2015) Connections of the limbic network: a corticocortical evoked potentials study. Cortex 62: 20-33. [Crossref]
- Matsuzaki N, Juhasz C, Asano E (2013) Cortico-cortical evoked potentials and stimulation elicited gamma activity preferentially propagate from lower- to higher-order visual areas. Clin Neurophysiol 124: 1290-1296. [Crossref]
- Valentín A, Alarcón G, Seoane JJG, Lacruz ME, Nayak SD et al. (2005) Single-pulse electrical stimulation identifies epileptogenic frontal cortex in the human brain. Neurology 65: 426-435. [Crossref]
- Saito T, Tamura M, Muragaki Y, Maruyama T, Kubota Y et al. (2014) Intraoperative cortico-cortical evoked potentials for the evaluation of language function during brain tumor resection: initial experience with 13 cases. J Neurosurg 121: 827‐838. [Crossref]
- Zelaya LV, Pastor J (2015) Intraoperative Neurophysiological Monitoring Techniques for the Resection of Malignant Brain Tumors Located in Eloquent Cortical Areas. Austin J Neurosurg 2: 1038.
- Hamer PCDW, Robles SG, Zwinderman AH, Duffau H, Berger MS et al. (2012) Impact of intraoperative stimulation brain mapping on glioma surgery outcome: a meta-analysis. J Clin Oncol 30: 2559-2565. [Crossref]
- Bizzi A, Nava S, Ferre` F, Castelli G, Aquino D et al. (2012) Aphasia induced by gliomas growing in the ventrolateral frontal region: Assessment with diffusion MR tractography, functional MR imaging and neuropsychology. Cortex 48: 255-272. [Crossref]
- Araki K, Terada K, Usui K, Usui N, Araki Y et al. (2014) Bidirectional neural connectivity between basal temporal and posterior language areas in humans. Clin Neurophysiol 19: S1388-S2457.
- Zelaya LV, Sola RG, Pulido P, Pastor J (2018) Do we Need to Wake Patients up during Cortical Surgery? J Cancer Res 7: 84-96.
