Elevated Level of Serum Vitamin D Reduce the Chance of Latent Genital Tuberculosis
A B S T R A C T
Aim: To explore the possibility of risk factor between latent genital tuberculosis with association of low secretion anti-mullerian hormone(AMH) and low level of Vitamin D for infertility.
Design: Retrospective study was done in the reproductive medicine unit of Calcutta Fertility Mission, Kolkata-700019, and India
Result: From January 2017 to January 2019, total 150 Subjects (n= 75 TB-PCR positive and n=75 TB-PCR negative cases as control) were included in this study on the basis of inclusion and exclusion criteria. We have found that mean value of AMH and Vitamin D were higher in PCR negative group than positive group. Various level of AMH and Vitamin D concentration for PCR positive and negative cases were statistically significant (p-value < 0.01).
Conclusion: Our finding is that vitamin D deficiency is highly prevalent not only in active tuberculosis but also in latent genital tuberculosis subjects and serum Vitamin D level may help in treatment of latent genital tuberculosis. Elevated level of Vitamin D in serum may rule out latent tuberculosis.
Keywords
Latent genital tuberculosis, anti-mullerian hormone, polymerase chain reaction, in vitro fertilization, cholecalciferol, to 25 hydroxyvitamin D, 1,25-dihydroxyvitamin D, Mycobacterium tuberculosis, DNA, chemiluminescence immunoassay
Introduction
Standing on 2nd decade of twenty 1stcentury, infertility has been considered as a social stigma especially developing countries like India which is also a serious contributory factor in mental stress of an infertile couple, mainly affected woman and became clinical modalities with high incidence in nature. Currently, all over world mainly in developing country, latent genital tuberculosis become endemic in reproductive age span of women [1]. Due to the silent nature of this disease and due to the non-availability of reliable confirmatory diagnostic procedures, it remains undetected for a long time. The virulence of tubercle bacilli determines the immune response generated by the host, leading to either active or asymptomatic, latent infection persisting for several years [2]. Till date, some studies have focused a link between tuberculosis and vitamin D and deficiency of vitamin D also increased the risk of tuberculosis infection and due to deficiency of Vitamin D may possibly increase the rate of susceptibility for developing the tuberculosis [3-6]. Actually, hypovitaminosis D has been more common in low income group in developing country. During direct exposure to sunlight, vitamin D is mainly synthesized in skin [6, 7]. Parental Vitamin D produced endogenously under the influence of UV light which is called as 7-dehydrocholesterol in skin or cholecalciferol, also known as Vitamin D3 [8].
Vitamin D is metabolized first to 25 hydroxyvitamin D (25OHD), then to the hormonal form 1,25-dihydroxyvitamin D (1,25(OH)2D) which is the bioactive form of Vitamin D. Vitamin D may prevent the infection of mycobacterium through the attachment of bioactive Vitamin D to the Vitamin D receptor which regulates the expression of gene [9]. Conversion of Vitamin D inactive form to bioactive form, which help to enhance the expression of antimicrobial peptide that is called cathelicidin. Vitamin D has been considered as an immune-modulatory effector because it has been proved that it may play the crucial role in inducing antimycobacterial activity which is accomplished by inhibiting the growth of Mycobacterium tuberculosis and up-regulating protective innate host responses [10, 11]. Low level of serum Vitamin D is correlated to latent genital tuberculosis. On the other hand, during ovarian reserve test (ORT), low anti-mullarian hormone (AMH) level was observed in latent genital tuberculosis affected women who have low serum vitamin D level also. Mycobacterium tuberculosis may have toxic effects on ovarian reserve, which would explain the poor results observed during the intrauterine insemination or IVF cycle, leading to infertility [12].
Diminished or low level of Vitamin D is a common presentation in women in developing country. In many situations low serum Vitamin D level is detected even in younger patients affected by latent genital tuberculosis. Interest has been created to find out whether low serum Vitamin D level enhances the risk of latent genital tuberculosis and low AMH secretion. The study is aimed at estimating serum Vitamin D level may be one indicator in latent genital tuberculosis affected women. Whether low Vitamin D level enhances the latent genital tubercular infection which reduces ovarian reserve or ovarian AMH secretion is the point of interest or to explore whether serum vitamin D is correspondingly associated with latent genital tuberculosis and low AMH secretion.
Materials & Methods
This prospective observational study was done in the reproductive medicine unit of Calcutta Fertility Mission, Kolkata, India from January 2017 to January 2019. Subjects, who were recruited for IVF programme, prescribed for biochemical hormone investigation like anti-mullerian hormone (AMH), follicle stimulating hormone (FSH), luteinizing hormone (LS), prolactine (PRL), testosterone (ST), 25-OH vitamin D (VITD3) and estradiol (E2) assay along with tubercular DNA test. Total 150 Subjects (n= 75 cases and n=75 control) were included in this study between age group 25-40years on the basis of inclusion and exclusion criteria.
I Selection Criteria
Both experimental subjects and control were selected based on the following same criteria: subjects with reproductive age group (20 to 40years), primary or secondary infertility above 2 years from the planning , normal menstrual cycle length was 26-34 days and tubercular DNA PCR positive and negative subjects. Subjects with following criteria were excluded from study both as experimental and controls: subjects with polycystic ovaries or polycystic ovarian syndrome, endometriosis, any vaginal infection like Chlamydia, gonorrhea ,suffered from pulmonary tuberculosis or extra pulmonary tuberculosis other than genital organ, history of previous ATD treatment and having any genetic disorder.
II Ethical Approval
A specific written consent format was designed according to ethical guidelines of Helsinki declaration 1975 and received the ethical clearance from Institutional Ethical Committee of Calcutta Fertility Mission, Kolkata, India and informed consent form was obtained from the each subject prior to sample collection
III DNA-PCR Study
Using IUI cannula by the specialist via cervical os, menstrual blood on the 2nd of cycle and in between 21 to 23rd day for endometrium tissue of her natural cycle were collected, who were recruited for this study. After collecting the menstrual blood and endometrium tissue, the contents were transferred to lysis buffer and freezed in -20oc for extraction to DNA. RNA kit was very expensive so we could not extract the RNA from sample. DNA was extracted by QIAamp DNA Mini Kit (Qiagen, Hilden, Germany) from the menstrual blood as well as endometrium tissue and eluted by 150 microliter of elution buffer, was stored at -20oc for PCR. A multiplex PCR was carried out by three sets of primer to identify the tuberculosis genome. Details of three sets of primer and condition of Multiplex PCR was described in elsewhere [13].
IV Vitamin D3 and AMH
Serum was separated by centrifugation from fasting morning sample of 5 ml of whole venous blood in the time of pre IVF biochemical investigation tests by venipuncture from all those subjects and preserved in -20oc for vitamin D3 and AMH assay. Vitamin D was estimated by Calbiotech 25-OH vitamin D total test system ELISA kit (Calbiotech, Cordell ct, California) and quantities determination of Vitamin D concentration in subjects serum were measured by chemiluminescence immunoassay (CLIA) (Tosoh, Lake-forest, California, USA).In this technique 25 ul of serum sample, vitamin D calibrators poured into assigned well for measurement of Vitamin D3 level. Estimation was done as per manufacture’s instruction. According to our reference interval, concentration of Vitamin D3 was <10ng/ml -Deficiency, 10-30ng/ml -Insufficiency, 30-100ng/ml -Sufficiency, >100ng/ml -Intoxication. AMH level was estimated by AMH Gen II Enzyme Linked Immuno Sorbent Assay (ELISA) from Beckman Coulter Inc. USA. In this technique 25 ul of serum sample, AMH calibrators, were poured into assigned well for measurement the concentration of AMH followed by the manufacturer’s instructions. According to our reference interval, concentration of AMH was 0-0.29ng/ml -very low AMH, 0.30- 2.19ng/ml- low AMH, 2.20-3.99ng/ml -satisfactory AMH, 4-6.80ng/ml -optimal.
Results and Analysis
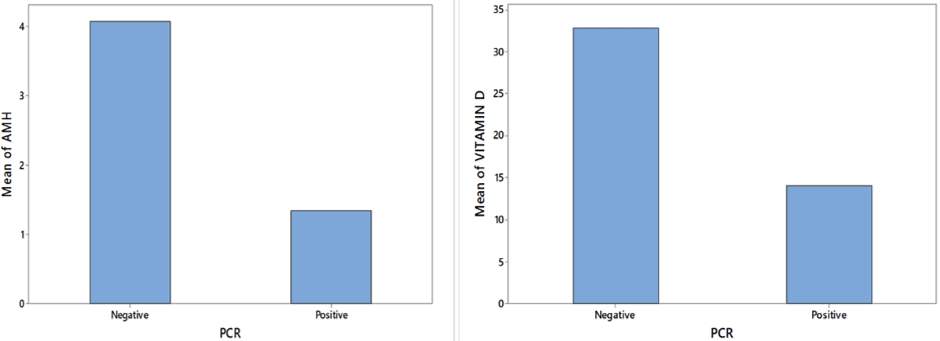
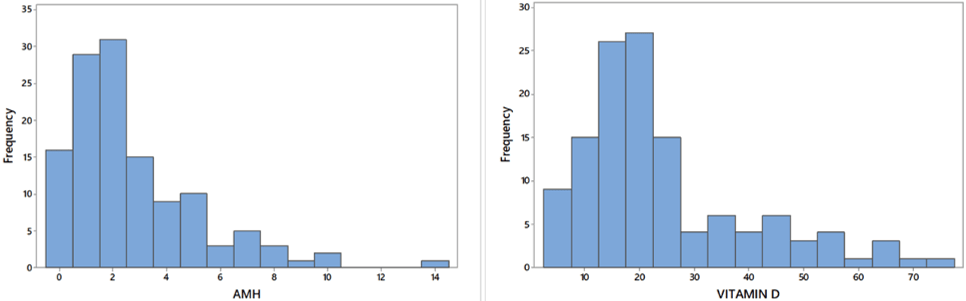
Out of 150 cases, 75cases were tubercular PCR positive (two consecutive cycles) and 75 cases were tubercular PCR (two consecutive cycles) negative. AMH and Vitamin D level is tabulated against PCR positive and negative group and diagrammed by bar chart in (Figure 1). From this bar chart we have found that mean value of AMH and Vitamin D were higher in PCR negative group than positive group. From (Figure 2), frequency of Vitamin D and AMH were histogramatically described. The mean value and Interquartile Range Formula (IQR) of AMH and Vitamin D concentration in case of PCR positive and negative group were presented in (Table 1). From (Table 2), number of positive and negative PCR cases is calculated from lower level to upper level of AMH and Vitamin D according to concentration. From (Table 3), we got a significance relation in median value and Interquartile Range of PCR positive and negative group versus various level of AMH and Vitamin D concentration. This suggests that various level of AMH and Vitamin D concentration for PCR positive and negative cases were statistically significant (p-value < 0.01).
Table 1: Test for difference in Vitamin D and AMH level against PCR
|
|
Vitamin D |
P-value |
AMH level |
P-value |
||
|
PCR |
Median |
IQR |
Median |
IQR |
||
|
Negative |
26.76 |
23.25 |
0.000 |
3.61 |
3.44 |
0.000 |
|
Positive |
13.69 |
8.00 |
1.04 |
1.36 |
||
Table 2: Test for association between levels of Vitamin D, AMH and PCR
|
|
|
PCR |
P-value |
|
|
|
|
Negative |
Positive |
|
|
Vitamin D |
VERY SEVERE VIT D DEFFICIENCY |
0 |
3 |
0.000 |
|
SEVERE VIT D DEFFICIENCY |
1 |
20 |
||
|
VIT D DEFFICIENCY |
9 |
44 |
||
|
SUB OPTIMAL VITMIN D PROVISION |
32 |
6 |
||
|
OPTIMAL VITAMIN D LEVEL |
21 |
2 |
||
|
UPPER VIT D LEVEL |
12 |
0 |
||
|
AMH level |
VERY LOW AMH |
0 |
11 |
0.000 |
|
LOW AMH |
19 |
52 |
||
|
SATISFACTORY AMH |
26 |
10 |
||
|
OPTIMAL AMH |
30 |
2 |
||
Table 3: Test for difference in levels of Vitamin D, AMH level against PCR
|
|
|
PCR |
P-value |
|||
|
|
|
Negative |
Positive |
|||
|
|
|
Median |
IQR |
Median |
IQR |
|
|
Vitamin D |
VERY SEVERE VIT D DEFFICIENCY |
|
|
3.59 |
1.25 |
0.000 |
|
SEVERE VIT D DEFFICIENCY |
7.25 |
- |
7.15 |
3.40 |
||
|
VIT D DEFFICIENCY |
18.19 |
2.88 |
14.97 |
4.38 |
||
|
SUB OPTIMAL VITMIN D PROVISION |
22.32 |
4.22 |
24.53 |
2.27 |
||
|
OPTIMAL VITAMIN D LEVEL |
38.72 |
9.09 |
48.11 |
- |
||
|
UPPER VIT D LEVEL |
58.13 |
14.04 |
- |
- |
||
|
AMH level |
VERY LOW AMH |
- |
- |
0.13 |
0.18 |
0.000 |
|
LOW AMH |
1.63 |
0.71 |
1.03 |
0.93 |
||
|
SATISFACTORY AMH |
2.97 |
1.00 |
2.64 |
0.55 |
||
|
OPTIMAL AMH |
6.14 |
2.14 |
6.84 |
- |
||
Figure 1: Bar chart of AMH and Vitamin D level against PCR
Figure 2: Histogram of AMH level and Vitamin D level
I Statistical Analysis
The bar chart shows much higher mean level of Vitamin D and AMH level with PCR negative cases than PCR positive cases (Figure 1). The histograms of AMH level and Vitamin D level show a positively skewed distribution for both the levels. Table 1 provides result of median test for difference in median of Vitamin D and AMH level for two levels of PCR. This suggests that significant differences in medians of AMH level and Vitamin D level exist for two levels of PCR (p-value < 0.01). There exists significant association between different levels of Vitamin D and PCR. Similarly, a significant association exists between different levels of AMH level and PCR.
Discussion
Vitamin D, AMH and female genital tuberculosis are closely related with infertility and still a contentious issue in developing country. In our study, we have found insufficient AMH and latent genital tuberculosis especially in female are interrelated to low amount of Vitamin D level, associated to increase risk of infertility. This finding suggests that vitamin D play a major role in up-regulation of latent genital tuberculosis in female, as a result low AMH level due to ovarian dysfunction [14]. Probable association between pulmonary tuberculosis and vitamin D has been reported from many countries [15]. Attempts have been made1st time, to establish, the existence of association among latent genital tuberculosis, AMH, Vitamin D as well as infertility and accordingly was characterized in this study. Our observation that AMH is significantly lower in female latent genital tuberculosis cases, which is probably not surprising, assuming the same relationship between Vitamin D and female latent genital tuberculosis. However, in this study we found that both AMH level and incidence of female latent genital tuberculosis were reduced due to Vitamin D, suggest that vitamin D may have prognostic value for the assessment of latent genital tuberculosis.
We also found that AMH and Vitamin D were lower in female latent genital tuberculosis cases than in controls. Low Vitamin D levels can be implicated in latent genital tuberculosis and that could be due to nutritional factors, however this is unlikely to be a single factor. Vitamin D is a fat-soluble vitamin and exists in two mail forms as cholecalciferol (Vitamin D3) which is synthesized by skin from 7-dehydrocholesterol in response to sun light exposure and Ergocalciferol (Vitamin D2) present in dietary sources. Both cholecalciferol and Erfocalciferol are converted to 25(OH) vitamin D in liver. Therefore, due to reduced exposure to sunlight or lack of proper nutrition, resultant vitamin D deficiency is strongly related with progression of latent genital tuberculosis. Alteration in the expression of antimicrobial peptide linkage also lead to the persistence of latent genital tuberculosis [16].
Conclusion
Association between Mycobacterium tuberculosis and Vitamin D deficiency is long established when its ill effect comes from the pulmonary tuberculosis or other forms of active tuberculosis. Our finding is that vitamin D deficiency is highly prevalent not only in active tuberculosis but also in latent female genital tuberculosis, resultant low AMH secretion, leading to infertility. Regular sun exposure and Vitamin D rich diet are essential to fight against latent genital tuberculosis. Thus, serum Vitamin D level may play an important role in treatment of latent genital tuberculosis and it can be used as a risk factor or clinical indicator for latent genital tuberculosis.
Limitation
In future large scale, well designed studies are needed to verify these results.
Funding
The study was funded by an intramural grant from Calcutta fertility mission.
Acknowledgement
The authors sincerely acknowledge Mr. Piyush Barg and Mr. Sambhu Chatterjee for laboratory assistance and Asst. Prof. Dr. Arabinda Das for statistical analysis.
Conflicts of Interest
None.
Key Message
Low level of AMH indicates poor ovulatory response to stimulation is observed in women undergoing IVF programme who has latent tuberculosis in genital tract and low level of Vitamin D in serum.
Article Info
Article Type
Research ArticlePublication history
Received: Mon 16, Dec 2019Accepted: Tue 14, Jan 2020
Published: Wed 22, Jan 2020
Copyright
© 2023 Siddhartha Chatterjee. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.CMR.2019.01.05
Author Info
Abira Datta Basudev Bhattacharya Rajib Gon Chowdhury Siddhartha Chatterjee Subhankar Chowdhury
Corresponding Author
Siddhartha ChatterjeeDepartment of Reproductive Medicine, Calcutta fertility mission
Figures & Tables
Table 1: Test for difference in Vitamin D and AMH level against PCR
|
|
Vitamin D |
P-value |
AMH level |
P-value |
||
|
PCR |
Median |
IQR |
Median |
IQR |
||
|
Negative |
26.76 |
23.25 |
0.000 |
3.61 |
3.44 |
0.000 |
|
Positive |
13.69 |
8.00 |
1.04 |
1.36 |
||
Table 2: Test for association between levels of Vitamin D, AMH and PCR
|
|
|
PCR |
P-value |
|
|
|
|
Negative |
Positive |
|
|
Vitamin D |
VERY SEVERE VIT D DEFFICIENCY |
0 |
3 |
0.000 |
|
SEVERE VIT D DEFFICIENCY |
1 |
20 |
||
|
VIT D DEFFICIENCY |
9 |
44 |
||
|
SUB OPTIMAL VITMIN D PROVISION |
32 |
6 |
||
|
OPTIMAL VITAMIN D LEVEL |
21 |
2 |
||
|
UPPER VIT D LEVEL |
12 |
0 |
||
|
AMH level |
VERY LOW AMH |
0 |
11 |
0.000 |
|
LOW AMH |
19 |
52 |
||
|
SATISFACTORY AMH |
26 |
10 |
||
|
OPTIMAL AMH |
30 |
2 |
||
Table 3: Test for difference in levels of Vitamin D, AMH level against PCR
|
|
|
PCR |
P-value |
|||
|
|
|
Negative |
Positive |
|||
|
|
|
Median |
IQR |
Median |
IQR |
|
|
Vitamin D |
VERY SEVERE VIT D DEFFICIENCY |
|
|
3.59 |
1.25 |
0.000 |
|
SEVERE VIT D DEFFICIENCY |
7.25 |
- |
7.15 |
3.40 |
||
|
VIT D DEFFICIENCY |
18.19 |
2.88 |
14.97 |
4.38 |
||
|
SUB OPTIMAL VITMIN D PROVISION |
22.32 |
4.22 |
24.53 |
2.27 |
||
|
OPTIMAL VITAMIN D LEVEL |
38.72 |
9.09 |
48.11 |
- |
||
|
UPPER VIT D LEVEL |
58.13 |
14.04 |
- |
- |
||
|
AMH level |
VERY LOW AMH |
- |
- |
0.13 |
0.18 |
0.000 |
|
LOW AMH |
1.63 |
0.71 |
1.03 |
0.93 |
||
|
SATISFACTORY AMH |
2.97 |
1.00 |
2.64 |
0.55 |
||
|
OPTIMAL AMH |
6.14 |
2.14 |
6.84 |
- |
||
References
- Schaefer G (1976) Female genital tuberculosis. Clin Obstet Gynecol 19: 223-239. [Crossref]
- Stead WW, Kerby GR Schlueter DP, Jordahl CW (1968) The clinical spectrum of primary tuberculosis in adults: confusion with reinfection in the pathogenesis of chronic tuberculosis. Ann Intern Med 68: 731-745. [Crossref]
- Holick MF (2006) High prevalence of vitamin dinadequacy and implication for health. Mayo clin Proc 81: 353-373. [Crossref]
- Wilkinson RJ, Llewwlyn M, Toossi Z, Patel P, Pasvil G et al. (2000) Influence of vitamin D deficiency and vitamin D receptors polymorphisms on tuberculosis among Gujrati Asian in West London;a case control study. Lancet 355: 618-621. [Crossref]
- Talat N, Perry S, Parsonnet J, Dawood G, Hussain R (2010) Vitamin D deficiency and tuberculosis progression. Emerg Infect Dis 16: 853-855. [Crossref]
- Nnoaham KE, Clarke A (2008) Low serum vitamin D level and tuberculosis:a systemic review and meta-analysis. Int J Epidemiol 37: 113-119. [Crossref]
- Friis H, Range N, Pedersen ML , Molgaard C, Changalucha J et al. (2008) Hypovitaminosis D is common among pulmonary tuberculosis patients in Tanzania but is not explained by the acute phase response. J Nutr 138: 2474-2480. [Crossref]
- Fischer PR, Thacher TD, Pettifor JM (2008) Pediatric vitamin D and calcium nutration in developing countries. Rev Endocr Metab Disord 9: 181-192. [Crossref]
- Vin Tangpricha (2014) The role of vitamin D in tuberculosis. J clin trans endocrinol 167-169.
- Salahuddin N, Ali F, Hasan Z, Rao N, Aqeel M et al. (2013) Vitamin D accelerates clinical recovery from tuberculosis: results of the SUCCINCT study(Supplementary Cholecalciferol in recovery from tuberculosis). A randomized placebo-controlled clinical trial of vitamin D supplementation in patients with pulmonary tuberculosis. BMC infectious Diseases 13: 22. [Crossref]
- Martineau A R, Wilkinson K A, Newton SM, Floto RA, Norman AW et al. (2007) IFN-gamma and TNF-independent Vitamin D inducible human suppression of mycobacteria: the role of cathelicidin LL-37. J Immunol 178: 7190-7198. [Crossref]
- Malhotra N , Sharma V, Bahadur A, Sharma JB, Roy KK et al. (2012) The effect of tuberculosis on ovarian reserve among women undergoing IVF in India. Int J Gynecol Obst 117: 40-44. [Crossref]
- Bhattacharya B, Karak K, Ghosal AG, Roy A, Das S et al. (2003) Development of a new sensitive and efficient multiplex polymerase chain reaction (PCR) for identification and differentiation of different mycobacterial species. Trop Med Int Health 8: 150-157. [Crossref]
- Datta A, Chowdhury S, Chowdhury RG, Bhattyacharya B and Chatterjee S (2019) Effect of Female Genital Tubercular Involvement on Anti-Mullerian Hormone Secretion in Latent Genital Tuberculosis Affected Women. Hearld J Reprod Med Gynaecol Obst 4: 108.
- Kearns Malcolm D, Tangpricha Vin (2014) The role vitamin D in tuberculosis. J Clin Transl Endocrinol 1: 167-169. [Crossref]
- Trullen JA, Lu lu, Pulido D, Bhakta S, Boix E (2017) Host antimicrobial peptides: The promise of new treatment stratigies against tuberculosis. Front Immunol 8: 1499. [Crossref]
