Bfp Adhesions and EspA Filament by Enteropathogenic E.Coli in Relation to Pathogenesis of Infantile Diarrhea in Children Under 5 Years in Mansoura University Children Hospital
A B S T R A C T
Background: Our study was applied aiming to detect the biofilm formation by the Enteropathogenic E. coli causing diarrhea and to investigate the relation between the Bfp adhesions and EspA filament containing strains with the severity of diarrhea in children under 5 years in Mansoura University Children Hospital, Egypt.
Methods: It was carried out by identification of EPEC, Serological agglutination test, Biofilm Formation under static condition at 3,6,9,24 hours.
Results: Out of 103 children with diarrhea, 50 E. coli were isolated, 16 (32%) were EPEC and 34 (68%) were non-EPEC. The CFU mean± SD of biofilm of 16 EPEC isolates at 4 time points 3, 6, 9, 24h are 108-1013, 69.5910±24.9310, 106-109, 20.2948±3.96778, 107-1011, 13.92×1010±3.376×1010, 109-1015, 6.402×1013± 2.496×1013. The relation of the biofilm formation and the severity of diarrhea in 16 EPEC isolates including 5 typical EPEC isolates and11 atypical EPEC reveals significant relation between the duration of the diarrhea and biofilm formation P=0.001**.
Conclusion: EPEC is capable of forming biofilms on plastic under static and flow through-continuous conditions which correlate to the severity of the diarrhea in children under 5 years.
Keywords
Bfp adhesions, EspA filament, enteropathogenic E. coli, infantile diarrhea
Introduction
EPEC was first known as a cause of infantile diarrhea in the 1940s’. It was associated with outbreaks in hospitals and nurseries. During this outbreak, antiserum was prepared to a strain of E. coli isolated from a patient with diarrhea and used this antiserum to show that the epidemic strains belonged to the same serogroup, later recognized as E. coli O111 [1]. Twelve sero-groups originally recognized by the World Health Organization as the classical EPEC were; O26, O55¸ O86, O111, O114, O119, O125, O126, O127, O128, O142 and O158 [2]. In the mid-twentieth century. In nursery outbreaks, mortality rates of EPEC 25–50% were reported in the United States and the United Kingdom. But the mortality rate of EPEC outbreaks in preterm neonates in Africa still can be as high as 30 %. EPEC has been categorized into two subgroups termed typical (tEPEC) and atypical (aEPEC). There was a decline in typical EPEC (tEPEC) prevalence paralleled by an increase in atypical EPEC (aEPEC) infections In the twenty-first century [3].
Typical EPEC strains are unique in that they express inducible bundle-forming pilus (BFP), composed mainly of BfpA, a highly conserved structural repeating protein subunit A, making it a suitable, highly specific, discriminatory virulence marker [4]. Infections caused by EPEC are difficult to be differtiated from those with other causes; symptoms include watery diarrhea, sometimes accompanied by low-grade fever and vomiting. If EPEC infection severe, vomiting may make oral rehydration difficult and life-threatening dehydration may occur. Furthermore, disease caused by EPEC may be protracted, resulting in weight loss, malnutrition and death. Symptoms can vary from one case to another according to the route by which the person was exposed, the dose of bacteria received, and other factors, including immune status [5].
Microcolony formation is the first step in biofilm development, and in EPEC it is mediated by several adhesins, including the bundle-forming pilus (BFP) and the EspA filament. EPEC forms biofilms on plastic under static conditions and a flowthrough. Under static conditions, this biofilm is arrested at the microcolony state and does not develop into a mature three-dimensional structure. On abiotic surfaces under flow through conditions, EPEC uses type 1 fimbriae for the initial adhesion. BFP is needed for microcolony development, and EspA might be also; development of a mature biofilm requires antigen 43 [6].
The ability to determine the microbial antibiotic susceptibility and to assess the effectiveness of other bactericidal treatments on bacteria in biofilm, depends on determining the number of viable bacteria in the biofilm, or at least the relative reduction in viable bacteria due to the treatment [7]. It was observed that EPEC infection is common in children before one-year age, this can be due to a decline in maternally acquired antibodies and the introduction of weaning foods that are potentially contaminated. In addition, crawling usually begins at this age and the risk of ingesting contaminated materials is high. Lower incidence of EPEC diarrhea in children after 2 years may be due to acquisition of antibodies reactive to EPEC virulence associated proteins by infants living in endemic areas [8]. The undertaken study aimed to investigate the biofilm formation by the Enteropathogenic E. coli causing diarrhea and to investigate the relation between the Bfp adhesions and EspA filament containing strains with the severity of diarrhea in children under 5 years in Mansoura University Children Hospital, Egypt.
Materials and Methods
The study was conducted over the period from September 2015 to April 2016.The study concluded 103 patients attended to Mansoura University Children Hospital (MUCH), Egypt. The age of the studied group ranged from 2-60 months (mean= 34.88 months, SD ± 17.469). There was 49 (47.6 %) male child and 54 (52.4%) female child. The inclusion criteria were that the patient should be under 60 months, have watery non-bloody diarrhea, have acute diarrhea at least for 48 hours but not more than 7days and have no history for antibiotic administration for 48 hours. Each patient was subjected to Complete Medical History of Diarrhea꞉ duration of the diarrhea, criteria of the stool꞉ colour, consistency, with bloody or mucous, previous attacks, vomiting꞉ duration of vomiting, amount and content of vomitus, previous attacks, abdominal pain, symptoms of infection꞉ fever, chills, myalgias, rash, rhinorrhoea, sore throat, cough, known immunocompromised status, appearance and behaviour꞉ include weight loss, level of thirst, level of alertness, increased malaise, or irritability, antibiotics, travel꞉ history of travel to endemic areas may make prompt consideration of organisms.
Complete clinical examination was done. Stool samples were collected in clean containers containing no disinfectant and were transported immediately in ice box to the laboratory for processing and examination. Fresh film was examined to exclude parasitic infestation. Direct film was stained with Gram stain. Each sample was inoculated on ordinary nutrient agar, blood agar, MacConkey’s, eosin methylene blue (EMB) as a selective media for E. coli and inoculated at 37°C for 24 hours. The observed colonies were examined by colonial morphology, film stained with gram stain and biochemical reactions; Oxidase test (OXOID Ltd., UK), Negative oxidase test colonies were selected, MICROBACT GNB 12/B/E, 24E (OXOID Ltd., UK) Oxidase negative samples were then tested by MICROBACT GNB 12/B/E, 24E according to the manufacturer instructions. On the next day, the microbact biochemical reactions were examined.
Serological agglutination test (2): E. coli strains then underwent serological agglutination test using polyvalent antisera for EPEC (O26, O55, O86, O111, O119, O125, O126, O127, O 128) (BIO -RAD). The E. coli colony from selective media (E.M.B), was suspended in 0.85% saline to give homogenous suspension. One drop of EPEC polyvalent antisera was placed at one end of clean microscopic slide and one drop of saline 0.85% NaCl was placed at the other end to be used as negative control. One loopful of the heated suspension was put onto each of the drops of antiserum and saline and mixed well for 1 minute. Strong agglutination within one minute was regarded as positive. The agglutination reaction was examined by naked eye and microscopically.
The ability of EPEC to form biofilms under static condition in 24 well cell culture was assessed by counting directly the CFU (Cm)2 on LBA media at four time points 3h, 6h, 9h, 24h, this method gave highly reproducible results. Dulbecco’s modified Eagle medium (DMEM; Invitrogen) and Luria Bertani Agar media (LBA; Invitrogen) were used. This experiment performed using Dulbecco’s modified Eagle medium (DMEM; Invitrogen). This medium was chosen because expression of the EPEC virulence genes has been extensively reported to be induced during growth in DMEM. E. coli colonies (5-10 colonies) were selected to be cultured in 1 ml DMEM in Eppendorf tube overnight in 37°C under static conditions. In the day after, the colour of DMEM were observed to be changed from red to yellow, the bacteria cultures were inoculated into fresh DMEM in a 1:100 dilution in 24-well cell culture plates (Falcon). The plates were incubated at 37°C in a CO2 atmosphere, using four different time points: 3, 6, 9 and 24 hours. The medium in plates was changed every 3 hours up to 24 hours. The biofilms were formed in 24 cell culture plate were rinsed three times with phosphate-buffered saline and disrupted with a solution of Triton X-100 (1%) in phosphate-buffered saline for 20 min. Serial dilutions were performed and plated on LBA for CFU counts.
Biofilm formation was quantified by direct calculating CFU ̸ (Cm)2 of bacteria on LBA media. The sample was considered positive when it formed growth on LBA media at the four time points ; 3h, 6h, 9h, 24h together, quantified by CFU ̸ (Cm)2, it was considered negative if it gave no growth in one or more of the four time points.
Statistical Analysis
Data were fed to the computer and analysed using IBM SPSS software package version 20.0. Qualitative data were described using number and percent. Quantitative data were described using range (minimum and maximum), mean, standard deviation and median. Significance of the obtained results was judged at the 5% level. The used tests were Chi-square test for categorical variables, to compare between different groups and Fisher’s Exact or Monte Carlo correction. Correction for chi-square when more than 20% of the cells have expected count less than 5.
Statistically significant꞉ means that the relationship between two or more variables is caused by something other than random chance, statistical hypothesis is used to determine whether the result of data set is statistically significant. This test provides a p-value, representing the probability that random chance could explain the results ; p-value of 5% or lower is considered to be statistically significant.
Results
The study concluded 103 patients attended to Mansoura University Children Hospital (MUCH) in the period from September 2015 to April 2016. Their ages ranged from 2-60 months with mean age ± SD 34.88 months ± 17.469. The diarrheal clinical manifestations of the children were described in (Table 1). Of the 103 clinically examined children, one (0.97%), 32(31.1%), 38(36.9%), 25(24.3%), 7 (6.8%) had diarrhea for 2, 3, 4, 5, 6 days respectively. The range was 2-6 days with Mean ± SD was 4.05 ± 0.93.
Of the total 103 clinically examined children, 41 (39.8%) had previous diarrheal attacks and 62 (60.2%) with no history of previous attacks of diarrhea, the range was 2- 48. Twenty-two children (21.4%), 34, (33%), 28 (27.2%), 103(100%) had fever, abdominal pain, vomiting, diarrhea respectively.
Table 1: Clinical manifestation of 103 clinically examined children.
|
Clinical Manifestation |
No Total No =103 |
% |
|
Duration of diarrhea (days) |
|
|
|
2 |
1 |
0.97 |
|
3 |
32 |
31.1 |
|
4 |
38 |
36.9 |
|
5 |
25 |
24.3 |
|
6 |
7 |
6.8 |
|
Previous Attacks |
|
|
|
Present |
41 |
39.8 |
|
Absent |
62 |
60.2 |
|
Associated Symptoms |
|
|
|
Fever |
22 |
21.4 |
|
Abdominal Pain |
34 |
33.0 |
|
Vomiting |
28 |
27.2 |
|
Diarrhea |
103 |
100.0 |
|
Macroscopic Examination |
|
|
|
Watery non bloody yellowish stool |
51 |
49.5 |
|
Brownish yellow watery non- bloody stool |
34 |
33.0 |
|
Greenish brown watery non bloody stool |
18 |
17.5 |
|
Parasitic Infestation |
|
|
|
Undigested food |
98 |
95.15 |
|
Ascaris lumbricoides |
1 |
0.9 |
|
Oxyuris vermicularis |
14 |
13.59 |
|
Entamoeba histolytica |
9 |
8.7 |
Table 2: Numbers and percentages of E.coli isolates recovered from 103 clinically examined children.
|
Bacterial species |
No Total No = 50 |
% |
|
E.coli only |
29 |
28.2 |
|
E.coli + Klebsiella oxytoca |
5 |
4.85 |
|
E.coli+ Pseudomonas aeruginosa |
10 |
9.7 |
|
E.coli+ Proteas mirabilis |
6 |
5.8 |
Table 3: Classification of 16 EPEC cases into typical and atypical according their clinical history.
|
|
Typical |
Atypical |
||
|
No (%) |
5(31.25%) |
11(68.75%) |
||
|
Age |
<12months |
>12months |
||
|
Duration of diarrhea |
4- 6 days |
1 - 2days |
||
|
|
Total No = 5 |
% |
Total No = 11 |
% |
|
Fever |
5 |
100 |
2 |
18 |
|
Abdominal pain |
5 |
100 |
4 |
36 |
|
Vomiting |
3 |
60 |
1 |
9.1 |
Out of all the 103 clinically examined children, 51(49.5%) showed yellowish watery non bloody stool in macroscopic examination, 34(33%) showed Brownish yellow watery non bloody stool, 18(17.5%) showed greenish brown watery non bloody stool. Ninety-eight (95.15%) samples were associated with undigested food, 1(0.97%), 14 (13.59%), 9 (8.7%) suffered from Ascaris lumbricoides, Oxyuris vermicularis, Entamoeba histolytica respectively. Only 50 child (48.5%) revealed growth of E. coli either in pure or mixed culture. The Numbers and percentages of E. coli isolates are recovered (Table 2). Of the 50 E. coli isolates, 29 (28.2%) were isolated in pure culture, 5 (4.85%) mixed with Klebsiella oxytoca, 10 (9.7%) mixed with Pseudomonas aeruginosa, 6 (5.8%) mixed with Proteas mirabilis. Also, 50 samples (100%) were associated with undigested food, 1(0.97%), 6 (5.8%), 4(3.9%) were associated with Ascaris lumbricoides, Oxyuris vermicularis, Entamoeba histolytica respectively.
The EPEC represented 16 cases from the E. coli isolates, 16 (32%) and 34 (68%) were non-EPEC. Of the EPEC isolates, 11 (68.75%) were near to the characters of atypical EPEC as their age were more than 12 months, and their symptoms and signs were less aggressive including the presence of fever in 2 (18%), abdominal pain in 4 (36%), vomiting in 1(9.1%) and duration of diarrhea was from 1 to 2 days, 5 (31.25%) cases were near to characters of typical EPEC as their age were less than 12 months and their symptoms and signs were more aggressive including the presence of fever in 5 (100%), abdominal pain in 5(100%), vomiting in 3 (60%) and duration of diarrhea was from 4 to 6 days as shown in (Table 3).
At 3 hours, the CFU (cm) 2 of biofilm formation of EPEC isolates and non EPEC isolates showed that all isolates of EPEC had range 108-1013, mean ± SD 69.59×1010±24.93×1010, 17(50%), 6(17.6%), 11 (32.4%) of 34 non EPEC isolates had a range, mean ± SD of 108-1012, 18.93×1010 ± 3.75×1010, 105 _107, 70×104± 5 ×104, 0, 0 respectively as cleared in (Table 4). But at 6 hours as shown in (Table 5), all isolates of EPEC had range 106-109, mean ± SD 20.29×108 ± 3.96×108, 17(50%), 6(17.6%), 11 (32.4%) of 34 non EPEC isolates had a range, mean ± SD of 106-109, 291.5×106±45.3×106, 103 _105, 70×102± 5 ×102, 0, 0 respectively (Table 5).
Table 4: CFU (cm)2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 3hour.
|
CFU (cm)2 |
EPEC Total No = 16 |
Non EPEC Total No = 34 |
||||||
|
No |
% |
Range |
Mean ± SD |
No |
% |
Range |
Mean ± SD |
|
|
> (10)7 |
16 |
100 |
108-1013 |
69.59×1010±24.93×1010 |
17 |
50 |
108-1012 |
18.93×1010±3.75×1010 |
|
>104 _ 107 |
0 |
0 |
0 |
0 |
6 |
17.6 |
105 _107 |
70×104± 5 ×104 |
|
0 (no growth) |
0 |
0 |
0 |
0 |
11 |
32.4 |
0 |
0 |
Table 5: CFU (cm)2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 6 hour.
|
CFU (cm)2 |
EPEC Total No = 16 |
Non EPEC Total No = 34 |
||||||
|
No |
% |
Range |
Mean ± SD |
No |
% |
Range |
Mean ± SD |
|
|
> 105 |
16 |
100 |
106-109 |
20.29×108±3.96×108 |
17 |
50 |
106-109 |
291.5×106±45.3×106 |
|
> 102 _ 105 |
0 |
0 |
0 |
0 |
6 |
17.6 |
103_ 105 |
70×102 ±5.1×102 |
|
0 (no growth) |
0 |
0 |
0 |
0 |
11 |
32.4 |
0 |
0 |
Table 6: CFU (cm)2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 9 hour.
|
CFU (cm)2 |
EPEC Total No = 16 |
Non EPEC Total No = 34 |
||||||
|
No |
% |
Range |
Mean ± SD |
No |
% |
Range |
Mean ± SD |
|
|
> 106 |
16 |
100 |
107-1011 |
13.92×1010±3.376×1010 |
17 |
50 |
107-1011 |
20.29×109±3.69×109 |
|
>103 _ 106 |
0 |
0 |
0 |
0 |
6 |
17.6 |
104 _ 106 |
40×104± 5.196×104 |
|
0 (no growth) |
0 |
0 |
0 |
0 |
11 |
32.4 |
0 |
0 |
Table 7: CFU (cm)2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 24 hour.
|
CFU(cm)2 |
EPEC Total No = 16 |
Non EPEC Total No = 34 |
||||||
|
No |
% |
Range |
Mean ± SD |
No |
% |
Range |
Mean ± SD |
|
|
> (10)8 |
16 |
100 |
109-1015 |
6.402×1013±2.496×1013 |
17 |
50 |
109-1015 |
31.11×1012±4.41×1012 |
|
>(10)5 _ (10)8 |
0 |
0 |
0 |
0 |
6 |
17.6 |
106_ 108 |
40×105±5.19×105 |
|
0 (no growth) |
0 |
0 |
0 |
0 |
11 |
32.4 |
0 |
0 |
Table 8: Area Under the Roc Curve.
|
Test Result Variable(s) |
Area |
Std. Errora |
Asymptotic Sig.b |
Asymptotic 95% Confidence Interval |
|
|
Lower Bound |
Upper Bound |
||||
|
biofilm 3h |
0.730 |
0.093 |
0.026 |
0.548 |
0.913 |
|
biofilm 6h |
0.729 |
0.094 |
0.027 |
0.545 |
0.912 |
|
biofilm 9h |
0.672 |
0.102 |
0.097 |
0.471 |
0.873 |
|
biofilm 24h |
0.697 |
0.105 |
0.057 |
0.491 |
0.904 |
The CFU (cm) 2 of biofilm formation of EPEC isolates and non EPEC isolates at 9 hours was described in (Table 6). All isolates of EPEC had range 107-1011, mean ± SD 13.92×1010±3.376×1010, 17(50%), 6 (17.6%), 11 (32.4%) of 34 non EPEC isolates had a range, mean ± SD of 107-1011, 20.29×109±3.69×109, 104 _106, 40×104±5.196×104, 0, 0 respectively. Table 7 illustrates CFU (cm) 2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 24 hours. All 16 isolates of EPEC had range 109-1015, mean ± SD 6.402×1013±2.496×1013, 17(50%), 6(17.6%), 11 (32.4%) of 34 no EPEC isolates had a range, mean ± SD of 109-1015, 31.11×1012±4.41×1012, 105 _108, 40×105±5.19×105, 0, 0 respectively.
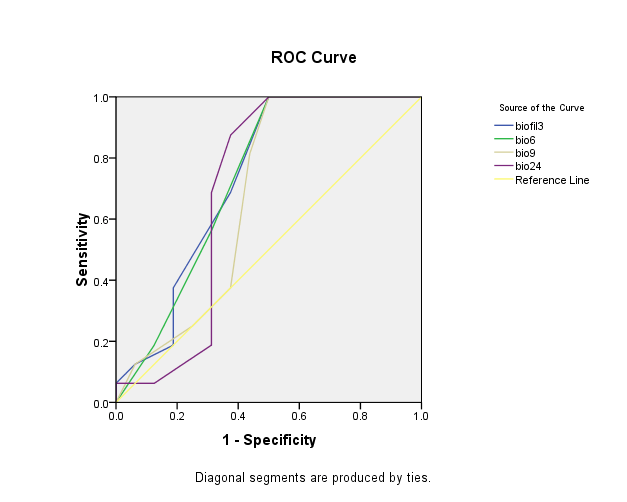
The Roc curve shows the specificity of EPEC isolates (Figure 1). Biofilm 3, biofilm 6, biofilm 9, biofilm 24 has at least one tie between the positive actual state group and the negative actual state group. Statistics may be biased (A) Under the nonparametric assumption, B) Null hypothesis: true area = 0.5). The point of the maximum specificity and sensitivity at 3h is 5.05× 107. The point of the maximum specificity and sensitivity at 6h is 5.05×106. The point of the maximum specificity and sensitivity at 9h is 5.5 ×106. The point of the maximum specificity and sensitivity at 24 h is 5.05 ×108 (Table 8).
Figure 1: Roc curve showing the specificity of EPEC isolates.
Studying the correlation between the biofilm formation and severity of diarrhea in the EPEC isolates, there was significant relation between the duration of the diarrhea and biofilm formation P=0.001** (Table 9).
Table 9: The biofilm formation and severity of diarrhea in 16 EPEC isolates including 5 typical EPEC isolates and11 atypical EPEC.
|
|
EPEC |
|
|
|
Characters of diarrhea |
Atypical N=11 |
Typical N=5 |
Test of significance |
|
Duration (days) |
2.0(1.0-2.0) |
5.0(4.0-6.0) |
Z=3.2 P=0.001** |
|
Previous Attack/weeks ago |
6.0(3.0-16.0) |
8.5(2.0-12.0) |
Z=0.16 P=0.8 |
|
Frequency/day |
4.0(2.0-6.0) |
4.0(3.0-6.0) |
Z=0.29 P=0.77 |
|
Characters of stool |
|||
|
Color |
|||
|
0(0.0) |
2(40.0) |
χ2=5.28 p=0.07 |
|
9(81.8) |
2(40.0) |
|
|
2(18.2) |
1(20.0) |
|
|
Blood |
0(0.0) |
0(0.0) |
|
|
Mucous |
2(18.2) |
1(20.0) |
FET p1.0 |
|
Consistency (watery)
|
11(100.0) |
5(100.0) |
|
Discussion
This study aimed to determine the percentage of EPEC strains among the isolated diarrhea samples from children under 5 years using serotyping slide agglutination test by polyvalent antisera and the ability of EPEC to form biofilm in comparison to non EPEC. This study was enrolled over 103 children attended to Mansoura University Children Hospital (MUCH). Out of them, only one case (1%) had diarrhea for 2 days, 32 cases (31.1%) had diarrhea for 3 days, 38 cases (36.9%) had diarrhea for 4 days, 25 cases (24.3%) had diarrhea for 5 days and 7 cases (6.8%) had diarrhea for 6 days The duration of diarrhea ranged from 2 to 6 days, mean (4.05 ± 0.93).
There was associated fever in 22 cases (21.4%), abdominal pain in 34 cases (33%) and vomiting in 28 cases (27.2%). There were 41 patients (39.8%) with previous diarrhea attacks and 62 patients (60.2%) with no history of previous diarrhea attacks. In a study by Nguyen et al. EPEC was associated with vomiting in 50% of patients, was generally not accompanied by fever, abdominal pain, or dehydration, and was not characterized by faecal blood indicating that it was not inflammatory in nature [9].
According to fresh film results, 98 (95.15%) samples contained undigested food, one case (0.9%) suffered from ascaris, 14 (13.59%) cases suffered from oxyuris, 9 (8.7%) cases suffered from amaebiasis. There were 50 (48.5%) E. coli isolates, 25 (24.3%) Klebsiella Oxytoca isolates, 11 (10.7%) Klebsiella pneumonia isolates, 6 (5.8%) Serratia liqueficiens isolates, 4 (3.9%) Proteas mirabilis isolates, 4 (3.9%) Pseudomenous aerogenesa isolates, 1 (1%) Enterobacter gergoviaeisolates, 1 (1%) Pleasemoneos shigelloide isolates and 1 (1%) Providincia staurtii isolates. In a cross-sectional study for 300 patients with acute diarrhea by Kabir et al., Enteropathogens were detected in 160 (53.5%) cases [10]. E. coli was detected in 18%, Shigella spp. in 2.4%, Salmonella spp. in 1.6%, E. Histolytica in 1.5% and G. Lamblia in 1.0% cases. Nguyen et al. studied etiologic agent of diarrhea in 134 children, EPEC were 31% of these children, followed by Salmonella sp. (10%), Campylobacter spp. (9%), Giardia sp. (6%) and Cryptosporidium sp. (2%) [9].
In this study, the serological test revealed that 32% of isolated E. coli were EPEC and 68% were non-EPEC. EPEC represented 15.5% of all cases of this study (16/103), while non EPEC represented 33% (34/103). In a study conducted by Behiry et al. the total percentages of E. coli isolates were 51.5%; of which, 46% were non EPEC which is more than our results non EPEC 33% and 5.2% (7%) were EPEC which is less than our result EPEC (16/103) cases (15.5%) [11].
A study of 500 E. coli isolates from children with diarrhea by multiplex PCR using fliC gene to detect that 8.2% isolated E. coli were EPEC [12]. Ochoa et al. studied diarrheagenic E. coli prevalence in Peru using 8,000 E. coli strains previously isolated from 8 different studies in children mainly younger than 36 months [8]. EPEC prevalence in diarrhea samples was 8.5% which is less than our result 32%. In contrast, a study conducted in Al Hilali et al. isolated only 3.4% of E. coli as EPEC with polyvalent antisera [13].
According to biofilm formation, all EPEC samples were positive biofilm formation, 16 non EPEC samples were tested, 8 samples were positive biofilm formation, 8 samples were negative biofilm formation. According to biofilm formation results after 3 hours, all EPEC samples (n=16, 50% of tested samples) gave growth > (10)7 CFU (cm)2 and were positive biofilm formation, 8 EPEC samples gave growth > (10)7 CFU (cm)2 and were positive biofilm formation, 3 non EPEC samples (9.4%) gave growth > (10)4 CFU(cm)2 and were negative biofilm formation, 5 samples (15.6%) gave no growth and were negative biofilm formation.
According to biofilm formation results after 6 hours, all EPEC samples (50.0% of samples were tested for biofilm formation) had shown growth > (10)5 CFU (cm)2 and were positive biofilm formation, 8 non EPEC samples (25%) had shown growth > (10)5 CFU (cm)2 and were positive biofilm formation, 3 samples (9.4%) gave growth > (10)2 CFU(cm)2 and were negative biofilm formation, 5 samples (15.6%) give no growth and negative biofilm formation. According to biofilm formation results after 9 hours, all EPEC samples (50.0% of samples were tested for biofilm formation) had shown growth > (10)6 CFU (cm)2 and were positive biofilm formation, 8 non EPEC samples (25%) had shown growth > (10)6 CFU (cm)2 and were positive biofilm formation, 3 samples (9.4%) gave growth > (10)3 CFU(cm)2 and were negative biofilm formation, 5 samples (15.6%) give no growth and negative biofilm formation.
According to biofilm formation results after 24 hours, all EPEC samples (50.0% of samples were tested for biofilm formation) had shown growth > (10)8 CFU (cm)2 and were positive biofilm formation, 9 non EPEC samples (28.8%) had shown growth > (10)8 CFU (cm)2 and were positive biofilm formation, 2 non EPEC samples (9.4%) gave growth > (10)5 CFU(cm)2 and were negative biofilm formation, 5 samples (15.6%) give no growth and negative biofilm formation. Moreira et al. tested the ability of the EPEC to form biofilms by performing CFU counts [6]. EPEC formed a biofilm with a biomass of 107 CFU/cm2 by 3h, 105 CFU/cm2 by 6 h, 106 CFU/cm2 at 12 h, 108 CFU/cm2 by 24 h and this method we based on our research. E. coli strain Nissle 1917, serotype O6: K5: H1, is a colonizer of the human gut even in the presence of a natural resident bacterial flora. Hancock et al. studied its ability to form biofilm in comparison to other ETEC and EPEC strains [14]. Nissle 1917 displayed a growth rate that was 48 % higher than that of the K-12, in all cases, Nissle 1917 contributed to > 76 % of the final biofilm population.
In Weiss-Muszkat et al. study, strain O55:H7 formed a biofilm on an abiotic surface [15]. Detection and quantification of biofilms on polystyrene were performed using 96-well polystyrene microtiter plates. Tested strains (O55:H10 (EPEC), O55:H7 (EPEC), O7:H4 (ETEC), O153:H8 (ETEC), O126:H27 (EAEC), O26:H119 (EHEC), O157:H7 (EHEC) and O127 (EPEC) were grown overnight at 37°C 6 in LB. The optical density of the culture was adjusted to OD600 of 1.0 (corresponding to ca. 108 cfu/ml). The plates were dried, and each well was stained crystal violet. Finally, the amount of extracted CV was determined by measuring the OD595 using ELISA plate reader. It was observed that EPEC isolate of serotype O55:H7 formed more robust biofilm than the other tested strains.
In study by Nascimento et al., 126 EPEC strains isolated from 92 children with diarrhea and 34 asymptomatic controls were examined for biofilm formation [16]. The frequency of biofilm producers was higher among isolates from patients compared to isolates from controls (34.8% vs 14.7%¬¬), while in study all EPEC cases (n=16,100%) formed biofilm and only 50% (n=8) of controls (n=16), which were non EPEC, formed biofilm. Culler et al. studied 91 aEPEC strains, isolated from faeces of children with diarrhea, they were analysed by the crystal violet (CV) assay on an abiotic surface after 24 hours of incubation [17]. Indirect detection of biofilm formation was based on OD595 readings and analysis of mean values of assays was performed in triplicate, 55 strains (60.4%) were considered biofilm forming strains which is more than our results, whereas the other 36 (39.6%) were considered non biofilm forming strains.
Sheikh et al. studied the biofilm formation in EAEC (32 strains), EPEC (10 strains), ETEC (10 strains), EHEC (10 strains), EIEC (10 strains) [18]. The ability of EAEC O42 to form biofilm at was assessed in different growth media, biofilms grown in a microtitre dish were quantified by a visual scoring system, biofilm formation was maximal in high-glucose DMEM cell culture Medium. He found that 87% of the EAEC strains formed a biofilm in high-glucose DMEM after 24 hours, as assessed by light microscopy, while no biofilm formed in EPEC, ETEC, EHEC and EIEC, while in this study all EPEC were positive biofilm formation assessed by quantification of biomass by CFU which is highly reproducible and accurate method.
Conclusion
EPEC is capable of forming biofilms on plastic under static and flow through-continuous conditions. Structural appendages, such as Bundle forming pilli (Bfp) adhesins and EspA filaments, which are important for microcolony formation, are also involved in bacterial aggregation during biofilm development. There is a relation between the biofilm formation of the EPEC and the severity of the diarrhea in children under 5 years.
Funding
None.
Conflicts of Interest
None.
Article Info
Article Type
Research ArticlePublication history
Received: Thu 26, Mar 2020Accepted: Wed 08, Apr 2020
Published: Mon 13, Apr 2020
Copyright
© 2023 Ghada Barakat. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.CMR.2020.01.03
Author Info
Ghada Barakat Mohamed Elkenawy Shorouq Eletribi
Corresponding Author
Ghada BarakatMedical Microbiology and Immunology Department, Faculty of Medicine, Mansoura University, Mansoura, Egypt
Figures & Tables
Table 1: Clinical manifestation of 103 clinically examined children.
|
Clinical Manifestation |
No Total No =103 |
% |
|
Duration of diarrhea (days) |
|
|
|
2 |
1 |
0.97 |
|
3 |
32 |
31.1 |
|
4 |
38 |
36.9 |
|
5 |
25 |
24.3 |
|
6 |
7 |
6.8 |
|
Previous Attacks |
|
|
|
Present |
41 |
39.8 |
|
Absent |
62 |
60.2 |
|
Associated Symptoms |
|
|
|
Fever |
22 |
21.4 |
|
Abdominal Pain |
34 |
33.0 |
|
Vomiting |
28 |
27.2 |
|
Diarrhea |
103 |
100.0 |
|
Macroscopic Examination |
|
|
|
Watery non bloody yellowish stool |
51 |
49.5 |
|
Brownish yellow watery non- bloody stool |
34 |
33.0 |
|
Greenish brown watery non bloody stool |
18 |
17.5 |
|
Parasitic Infestation |
|
|
|
Undigested food |
98 |
95.15 |
|
Ascaris lumbricoides |
1 |
0.9 |
|
Oxyuris vermicularis |
14 |
13.59 |
|
Entamoeba histolytica |
9 |
8.7 |
Table 2: Numbers and percentages of E.coli isolates recovered from 103 clinically examined children.
|
Bacterial species |
No Total No = 50 |
% |
|
E.coli only |
29 |
28.2 |
|
E.coli + Klebsiella oxytoca |
5 |
4.85 |
|
E.coli+ Pseudomonas aeruginosa |
10 |
9.7 |
|
E.coli+ Proteas mirabilis |
6 |
5.8 |
Table 3: Classification of 16 EPEC cases into typical and atypical according their clinical history.
|
|
Typical |
Atypical |
||
|
No (%) |
5(31.25%) |
11(68.75%) |
||
|
Age |
<12months |
>12months |
||
|
Duration of diarrhea |
4- 6 days |
1 - 2days |
||
|
|
Total No = 5 |
% |
Total No = 11 |
% |
|
Fever |
5 |
100 |
2 |
18 |
|
Abdominal pain |
5 |
100 |
4 |
36 |
|
Vomiting |
3 |
60 |
1 |
9.1 |
Table 4: CFU (cm)2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 3hour.
|
CFU (cm)2 |
EPEC Total No = 16 |
Non EPEC Total No = 34 |
||||||
|
No |
% |
Range |
Mean ± SD |
No |
% |
Range |
Mean ± SD |
|
|
> (10)7 |
16 |
100 |
108-1013 |
69.59×1010±24.93×1010 |
17 |
50 |
108-1012 |
18.93×1010±3.75×1010 |
|
>104 _ 107 |
0 |
0 |
0 |
0 |
6 |
17.6 |
105 _107 |
70×104± 5 ×104 |
|
0 (no growth) |
0 |
0 |
0 |
0 |
11 |
32.4 |
0 |
0 |
Table 5: CFU (cm)2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 6 hour.
|
CFU (cm)2 |
EPEC Total No = 16 |
Non EPEC Total No = 34 |
||||||
|
No |
% |
Range |
Mean ± SD |
No |
% |
Range |
Mean ± SD |
|
|
> 105 |
16 |
100 |
106-109 |
20.29×108±3.96×108 |
17 |
50 |
106-109 |
291.5×106±45.3×106 |
|
> 102 _ 105 |
0 |
0 |
0 |
0 |
6 |
17.6 |
103_ 105 |
70×102 ±5.1×102 |
|
0 (no growth) |
0 |
0 |
0 |
0 |
11 |
32.4 |
0 |
0 |
Table 6: CFU (cm)2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 9 hour.
|
CFU (cm)2 |
EPEC Total No = 16 |
Non EPEC Total No = 34 |
||||||
|
No |
% |
Range |
Mean ± SD |
No |
% |
Range |
Mean ± SD |
|
|
> 106 |
16 |
100 |
107-1011 |
13.92×1010±3.376×1010 |
17 |
50 |
107-1011 |
20.29×109±3.69×109 |
|
>103 _ 106 |
0 |
0 |
0 |
0 |
6 |
17.6 |
104 _ 106 |
40×104± 5.196×104 |
|
0 (no growth) |
0 |
0 |
0 |
0 |
11 |
32.4 |
0 |
0 |
Table 7: CFU (cm)2 of biofilm formation of 16 EPEC isolates and 34 non EPEC isolates at 24 hour.
|
CFU(cm)2 |
EPEC Total No = 16 |
Non EPEC Total No = 34 |
||||||
|
No |
% |
Range |
Mean ± SD |
No |
% |
Range |
Mean ± SD |
|
|
> (10)8 |
16 |
100 |
109-1015 |
6.402×1013±2.496×1013 |
17 |
50 |
109-1015 |
31.11×1012±4.41×1012 |
|
>(10)5 _ (10)8 |
0 |
0 |
0 |
0 |
6 |
17.6 |
106_ 108 |
40×105±5.19×105 |
|
0 (no growth) |
0 |
0 |
0 |
0 |
11 |
32.4 |
0 |
0 |
Table 8: Area Under the Roc Curve.
|
Test Result Variable(s) |
Area |
Std. Errora |
Asymptotic Sig.b |
Asymptotic 95% Confidence Interval |
|
|
Lower Bound |
Upper Bound |
||||
|
biofilm 3h |
0.730 |
0.093 |
0.026 |
0.548 |
0.913 |
|
biofilm 6h |
0.729 |
0.094 |
0.027 |
0.545 |
0.912 |
|
biofilm 9h |
0.672 |
0.102 |
0.097 |
0.471 |
0.873 |
|
biofilm 24h |
0.697 |
0.105 |
0.057 |
0.491 |
0.904 |
Table 9: The biofilm formation and severity of diarrhea in 16 EPEC isolates including 5 typical EPEC isolates and11 atypical EPEC.
|
|
EPEC |
|
|
|
Characters of diarrhea |
Atypical N=11 |
Typical N=5 |
Test of significance |
|
Duration (days) |
2.0(1.0-2.0) |
5.0(4.0-6.0) |
Z=3.2 P=0.001** |
|
Previous Attack/weeks ago |
6.0(3.0-16.0) |
8.5(2.0-12.0) |
Z=0.16 P=0.8 |
|
Frequency/day |
4.0(2.0-6.0) |
4.0(3.0-6.0) |
Z=0.29 P=0.77 |
|
Characters of stool |
|||
|
Color |
|||
|
0(0.0) |
2(40.0) |
χ2=5.28 p=0.07 |
|
9(81.8) |
2(40.0) |
|
|
2(18.2) |
1(20.0) |
|
|
Blood |
0(0.0) |
0(0.0) |
|
|
Mucous |
2(18.2) |
1(20.0) |
FET p1.0 |
|
Consistency (watery)
|
11(100.0) |
5(100.0) |
|
References
- Vieira MA, Dias RCB, Dos Santos LF, Rall VLM, Gomes TAT et al. (2019) Diversity of strategies used by atypical enteropathogenic Escherichia coli toinduce attaching and effacing lesion in epithelial cells. J Med Microbiol 68: 940-951. [Crossref]
- Al Mamun A, Mily A, Sarker P, Tiash S, Navarro A et al. (2013) Treatment with phenylbutyrate in a pre-clinical trial reduces diarrhea due to enteropathogenic Escherichia coli: link to cathelicidin induction. Microbes Infect 15: 939-950. [Crossref]
- Mellor GE, Fegan N, Duffy LL, McMILLAN KE, Jordan D et al. (2016) National Survey of Shiga Toxin-Producing Escherichia coli Serotypes O26, O45, O103, O111, O121, O145, and O157 in Australian Beef Cattle Feces. J Food Prot 79: 1868-1874. [Crossref]
- de Souza Campos Fernandes RC, Quintana Flores VM, Sousa de Macedo Z, Medina Acosta E (2003) Coproantibodies to the enteropathogenic Escherichia coli vaccine candidates BfpA and EspB in breastfed and artificially fed children. Vaccine 21꞉ 1725-1731. [Crossref]
- World Health Organization. (2008) WHO Foodborne disease outbreaks: Guidelines for investigation and control. World Health Organization; Geneva 74.
- Moreira CG, Palmer K, Whiteley M, Sircili MP, Trabulsi LR et al. (2006) Bundle-forming pili and EspA are involved in biofilm formation by enteropathogenic Escherichia coli. J bacteriol 188: 3952-3961. [Crossref]
- Rasko DA, Webster DR, Sahl JW, Bashir A, Boisen N et al. (2011) Origins of the E.coli strain causing an outbreak of hemloytic-uremic syndrome in Germany. New Engl J Med 365: 709-717. [Crossref]
- Ochoa TJ, Ecker L, Barletta F, Mispireta ML, Gil AI et al. (2009) Age-related susceptibility to infection with diarrheagenic Escherichia coli among infants from Periurban areas in Lima, Peru. Clin Infect Dis 49: 1694-1702. [Crossref]
- Nguyen TV, Le PV, Le CH, Weintraub A (2005) Antibiotic resistance in diarrheagenic Escherichia coli and Shigella strains isolated from children in Hanoi, Vietnam. Antimicrob Agents Chemother 49: 816-819. [Crossref]
- Kabir MR, Hossain MA, Paul SK, Mahmud C, Ahmad S et al. (2012) Enteropathogens associated with acute diarrhea in a tertiary hospital of Bangladesh. Mymensingh Med J 21: 618-623. [Crossref]
- Behiry IK, Abada EA, Ahmed EA, Rania Labeeb RS (2011) Enteropathogenic Escherichia coli Associated with Diarrhea in children in Cairo, Egypt. ScientificWorldJournal 11: 2613-2619. [Crossref]
- Jafari A, Aslani MM, Bouzari S (2012) Escherichia coli: a brief review of diarrheagenic pathotypes and their role in diarrheal diseases in Iran. Iran J Microbiol 4: 102-117. [Crossref]
- Al Hilali SA, Almohana AM (2011) Occurrence and molecular characterization of enteropathogenic Escherichia coli serotypes isolated from children with diarrhea in Najaf, Iraq. Indian J Med Microbiol 29: 383-388. [Crossref]
- Hancock V, Dahl M, Klemm P (2010) Probiotic Escherichia coli strain Nissle 1917 outcompetes intestinal pathogens during biofilm formation. J Med Microbiol 59: 392-399. [Crossref]
- Weiss Muszkat M, Shakh D, Zhou Y, Pinto R, Belausov E et al. (2010) Biofilm formation by and multicellular behavior of Escherichia coli O55:H7, an atypical enteropathogenic strain. Appl Environ Microbiol 76: 1545-1554. [Crossref]
- Nascimento HH, Silva LE, Souza RT, Silva NP, Scaletsky IC (2014) Phenotypic and genotypic characteristics associated with biofilm formation in clinical isolates of atypical enteropathogenic Escherichia coli (aEPEC) strains. BMC Microbiol 14: 184. [Crossref]
- Culler HF, Couto SCF, Higa JS, Ruiz RM, Yang MJ et al. (2018) Role of SdiA on Biofilm Formation by Atypical Enteropathogenic Escherichia coli. Genes (Basel) 9: E253. [Crossref]
- Sheikh J, Hicks S, Dall'Agnol M, Phillips AD, Nataro JP (2001) Roles for Fis and YafK in biofilm formation by enteroaggregative Escherichia coli. Mol Microbiol 41: 983-997. [Crossref]
