The human body harbors several microbial cells as all of our somatic and germ cells combined. The collective genome of our microbiota – referred to as the microbiome, encodes approximately 100 fold more genes than the human genome. The majority of the microbiota are bacteria that reside within our gastrointestinal tract; however, viruses, archaea, and eukaryotes such as yeast and protozoans also inhabit the GI tract and other body sites.
Cancer is a leading cause of mortality and morbidity, with ~1.7 million newly diagnosed cancer cases and ~600,000 cancer deaths every year in USA alone. The human body nurtures numerous microbiota that influences cancer susceptibility, in part, via their gigantic metabolic capacity and profound influences on the host immune system. Microorganisms drive tumorigenesis in 15-20% of cancer cases. Metagenomic sequencing studies of microbiome found that a large number of malignancies are associated with an altered composition of commensal microbiota/dysbiosis. However, association studies fail to distinguish whether modifications in microbiota are causes/effects of cancer. Studies indicate that microbiota can alter cancer progression and susceptibility by diverse mechanisms such as inducing DNA damage, modulating inflammation, and producing metabolites involved in oncogenesis and tumor suppression.
The high-impact report states that cancer is primarily stochastic or “bad luck” that occurs due to gathering spontaneous mutations during DNA replication in cells and tissues where stem cells undergo a relatively large number of cell divisions. Although, it is believed that the environment significantly influences the risk of cancer. The microbiota that inhabits our GI tract and other anatomical sites is considered an environmental factor that we are constantly exposed throughout our lives.
Microbes Drive Certain Cancers
Helicobacter pylori infections are strongly linked to gastric adenocarcinoma and are mediated by inflammation with H. pylori-induced gastritis which is considered a cancer precursor. However, H. pylori protect against esophageal adenocarcinoma and Barrett’s esophagus possibly by affecting stomach pH and ameliorating acid reflux. H. pylori are found to cause cancer by triggering chronic inflammation inside the host and, in turn, damages the DNA. Due to its cancerous nature, H. pylori is in the line of being exterminated from human populations. This illustrates that the relationship between so-called “pathogenic microbes” and the human host can be considerably more complicated than initially assumed.
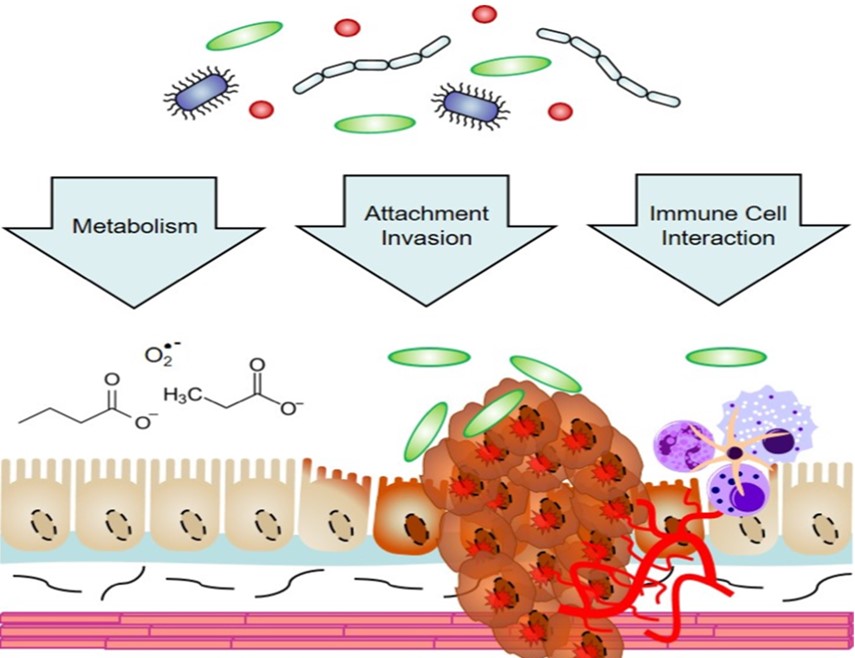
Current data analysis has shown that short-chain fatty acids such as acetate, butyrate, and propionate function in the suppression of inflammation and development of cancer, whereas other microbial metabolites, such as secondary bile acids, also promote carcinogenesis. Unlike viruses, which constitutively express active viral mimics of cellular proto-oncogenes; tumor initiation and progression are associated with microbial dysbiosis, a multifactorial event and arises following “multiple hits”. All individuals infected with oncogenic microorganisms do not develop cancer. Microbe genetic heterogeneity and the host, in addition to environmental factors – determine the prevalence of cancer and severity. H. pylori strains containing the CagA virulence factor that efficiently triggers gastric cancer and gastritis. Host genetics that influences the immune cell response is another significant determinant of whether an infected individual will develop cancer. In addition, external factors such as lifestyle and diet such as consuming alcohol, tobacco.
Research suspects a different mechanism in which bacteria may have the capacity to directly alter/damage DNA and modify the genes that can be turned on and off in normal cells. An enzyme named APOBEC3G found in the study with E. coli is known to trigger mutations that occur as benign tumor cells transform into cancerous malignancies that spread throughout the body and appear to cause harmful mutation in genes during DNA replication. Emerging evidence suggests that microbiota can be manipulated to improve cancer treatment and can also be used to synthesize personalized medicines. By designing small molecules that target microbial enzymes or incorporating probiotics as adjuvants in checkpoint immunotherapy, microbiota can be harnessed to improve cancer care.