A Review on Cardiac Biomarkers Detection for Heart Failure Prognosis
A B S T R A C T
Nowadays, problems in healthcare sector seem to be more crucial than ever. Among these, cardiovascular diseases (CVDs) play an important role, being for years a major threat to human health. In a bid to reduce the number of deaths due to heart diseases by taking precautions, science has turned to the development of devices able to offer early diagnosis of a heart failure. Recently, many biosensors have been developed based on cardiac biomarkers that can be detected in blood and predict the risk of heart failure. Of these, cardiac troponin is the best known, while C-reactive protein and myoglobin have also been used. Troponin is a complex of proteins found in the thin filaments of striated muscles and consists of three protein subunits, I, C and T. In case of myocardial malfunction, troponin is released into the blood. Consequently, the detection of elevated troponin levels at an early stage in human serum, is an indication of high risk of cardiovascular event enabling early medical intervention and treatment. In the present work, after the analysis of heart diseases, medical tests, troponin and its role in controlling myocardial health, there is an overview of the available biosensor platforms for cardiac biomarkers detection.
Keywords
Cardiovascular diseases, heart failure prediction, cardiac biomarker, troponin, biomolecule detection techniques, biosensors
Introduction
It is widely accepted that nowadays, healthcare sector is in a state of global turmoil. Science and technology are called to sustain medicine by consisting of a weapon to fight all life-threatening ailments and diseases. Cardiovascular diseases (CVDs) are the cause for about 18 million deaths each year, according to the World Health Organization. More than 75% of them occur in low- and middle-income countries, where access to health care is limited. In fact, it is predicted that by 2030, 7 out of 10 deaths worldwide will be the result of chronic diseases, with cardiovascular diseases accounting for the largest percentage of them [1, 2].
The risk factors for CVDs are numerous and unfortunately almost inevitable due to the modern lifestyle in urban areas. They are related to stress, fast pace of life, lack of physical exercise, poor diet, or drug abuse. The effects of these factors have an impact in human health causing high blood pressure, blood glucose, blood lipids or obesity and indicate a high risk of heart attack, heart failure and many other complications [3]. The prognosis for CVDs is crucial for both the healthcare sector and the global economy. However, early detection of vulnerable population groups and appropriate treatment can prevent premature death. In fact, a recent report by the US Centers for Disease Control and Prevention states that almost a quarter of all deaths caused by CVDs can be prevented if precautions are taken [4].
Therefore, there has been a growing demand for the development of portable, fast, easy-to-use and low-cost devices that are used to prevent cardiovascular diseases. In these circumstances, biosensors play an important role, as they enable early diagnosis with no need for a hospital visit associated with costly and time-consuming laboratory tests.
Troponins have been introduced into laboratory testing and the diagnosis of heart disease as highly specific and sensitive biological markers of myocardial damage. They form a protein complex located in the myocardium and consist of subunits I, C and T. The detection of elevated troponin levels in the blood is the first and basic sign of myocardial damage. In this case, further medical tests and treatments should follow in order to avoid the risk of patient's life [5, 6].
The present work, therefore, aspires to provide the essential information about CVDs, biological markers, medical examinations in order to review the available detection techniques and considerations to create more practical biosensors that could help people of high-risk groups.
Methods
Scientific terms such as “biosensors”, “biomolecule detection techniques”, “cardiac biomarkers” and “troponin” were searched in PubMed, Google Scholar, ScienceDirect. The appropriate articles were studied and a bibliographic research was done to select the information and form the present review.
Review of Literature
I Cardiovascular Diseases and Medical Tests
Among the cardiac disorders, many of them concern the heart itself, while others are related to pathology of the large vessels that are directly associated with it as well as its association with other pathological conditions. The most common CVDs are: [7]
i. Heart attack: Interruption of blood supply to the heart muscle, resulting from blocked arteries [8].
ii. Angina: Chest pain resulting from the heart not receiving enough oxygen, a common symptom of coronary heart disease [9].
iii. Coronary artery disease (CAD): Hardening of the arteries that supply blood to the heart. Acute coronary syndromes include: [10]
a. acute myocardial infarction (AMI) with ST-segment elevation (STEMI),
b. acute myocardial infarction (AMI) without ST elevation (NSTEMI),
c. unstable angina.
iv. Arrhythmia: Any change in the normal rhythm of the heartbeat [11].
v. Chronic or congestive heart failure: Inability of the heart to pump enough blood through the body to ensure a sufficient supply of oxygen [12].
vi. Hypertension (high blood pressure): Occurs when the blood pressure is constantly elevated, above the recommended limits [13].
vii. Stroke: Damage to nerve cells in the brain caused by lack of oxygen [14].
viii. Peripheral vascular disease (PVD): Hardening of the arteries leading away from the heart, usually in the legs, lower arms or arms [15].
Acute myocardial infarction, in particular, is the most common form of heart disease and the leading cause of morbidity and mortality worldwide. Despite continuous efforts to improve the pharmacological and invasive treatment of myocardial infarction, it is widely accepted that the condition for the success of any treatment is the timely recognition of its symptoms and the immediate start of treatment. In these cases, even a few hours can save lives [16].
The tests and techniques for detecting heart disease that are followed in each case depend on the condition of each patient. In addition to blood tests for biomarker values and chest x-rays, tests for diagnosing heart disease often include:
i. Electrocardiogram (ECG or ECG or ECG): The ECG is a quick and painless examination performed either at rest or during exercise and records the electrical potential produced by the myocardium during its contraction [17].
ii. Holter Monitor: The Holter monitor is a portable ECG device that continuously records heart rate, usually for 24 to 72 hours, and is used to detect problems that are not detected during an ECG examination [18].
iii. Echocardiogram: Non-invasive examination that uses sound waves to produce detailed images of the structure of the heart [19].
iv. Stress Test: During cardiac examinations and imaging, an increase in heart rate is caused by exercise or medication to check the response of the heart.
v. Cardiac computed tomography (CT) scan: the patient lies on a table inside a cylindrical machine and an X-ray tube inside the machine rotates around the body collecting images of the heart and chest [20].
vi. Cardiac MRI: Using a magnetic field and computer-generated radio waves, detailed images of the heart are produced [21].
It should be noticed that the development of increasingly sensitive and tissue-specific myocardial biomarkers and sensitive imaging techniques allows the detection of myocardial damage even at a very early stage [22].
II Cardiac Biomarkers and Troponin
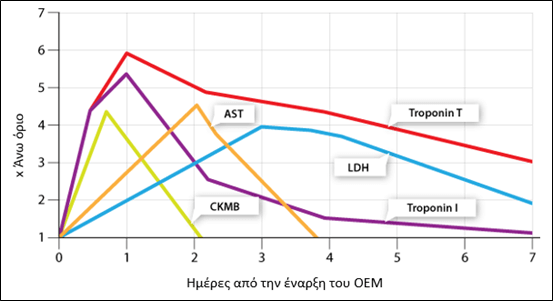
Cardiac enzymes have played a major role in the clinical treatment of patients with precardiac pain for many decades. In combination with some of the above techniques and the patient's history, they diagnose heart disease and help in monitoring the patient's course [23]. These enzymes are released into the circulation with the necrosis of myocardial fibers. Their levels in the blood increase within a few hours of the OEM invasion and gradually decrease over the next few days (Figure 1). The most common of them are: creatine kinase (CK-MB), cardiac I or T troponin (cTn), aspartic aminotransferase (AST) and lactic dehydrogenase (LDH). Hence, only cardiac troponin has been used to direct therapeutic interventions. Its concentration begins to increase rapidly within 3-4 hours after the onset of OEM, and remains high for up to 14 days, which is why its serum level is said to be more closely related to its degree of progression compared to other enzymes [24].
Thus, in recent years, troponins have been proposed in laboratory testing as highly specific and sensitive biological markers of myocardial damage [23]. Troponin is a protein complex found in the heart muscle. It is usually not found in the blood in high concentration. When the myocardial fibers are damaged, troponin is released into the bloodstream. As heart damage increases, more troponin is released into the bloodstream. High levels of troponin in the blood may indicate the presence or recent presence of a heart attack [25].
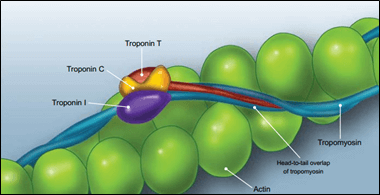
The troponin protein complex (cTn) plays a critical role in regulating myocyte contraction. This complex consists of three subunits that regulate the actin-myosin interaction, troponins C, T and I (Figure 2). Troponin-I (‘Inhibitory’) inhibits actin-myosin interaction. Troponin-C (‘Calcium-binding’) binds calcium thus regulating actin-myosin inhibition. Tropomyosin-binding (Tropomyosin-binding) binds tropomyosin. Troponins are found at regular intervals along actin filaments. In the presence of potential, the intracellular calcium concentration increases and calcium binds to troponin-C. This leads to a change in the modulation of troponin-I and tropomyosin, thus removing the inhibition of the actin-myosin interaction. This process also occurs in the skeletal muscle, except for the heart muscle, but not in the smooth muscle [5, 24].
Figure 1: Release of troponins T and I, CK-MB, AST, LDH [24].
Figure 2: Cardiac Troponin complex [24].
Troponins have isoforms found exclusively in myocardial cells (cTnT, cTnI and cTnC) but the homology between skeletal and cardiac troponin is 60% for troponin I, 90% for T and over 90% for C. This is the reason why troponin C is not used as a cardiac biomarker, while troponin I has a specificity advantage over T [23].
Lately, troponins have been widely used in clinical practice. However, it should be noted that elevated troponin levels are not sufficient to diagnose a heart condition. The doctor also takes into account the patient's other symptoms, as well as the results of other diagnostic tests, such as a physical exam and electrocardiogram, to make a diagnosis. Troponins can provide clinical assistance in the diagnosis of myocardial infarction, the prognosis and the evaluation of the effectiveness of treatment [23].
Troponins appear elevated in any case of myocardial damage (e.g., myocarditis, chemotoxicity from chemotherapy, heart failure, pulmonary embolism) and not only in CAD. It takes 4-6 hours from the onset of symptoms in order to sensitively diagnose myocardial damage based on troponin rise. Troponin levels remain high for 10-14 days [23]. Studies have shown that many heart diseases can lead to an increase in troponin due to myocardial injury, which makes the interpretation and specificity of a positive test more difficult, requiring further diagnosis and treatment [26]. However, it should be noted that the quality criteria of the troponin test in this study showed excellent sensitivity and excellent negative diagnostic value in all subgroups making it suitable to rule out acute myocardial infarction.
Finding elevated cTnT or cTnI values in patients with a clinical picture of unstable angina has prognostic value, as well. In patients with ST-elevation electrocardiogram, even when the cardiac enzyme creatine kinase CK-MB remains normal, elevated cTnT or cTnI values are associated with an increased risk of death, myocardial infarction, and ischaemic recurrence. In contrast, patients with angina pain who have normal troponin levels on two consecutive measurements 4-6 hours apart (and at least 6 hours after the onset of symptoms) are a low-risk group for relapses [23]. Several studies have already suggested that a single cTnI or cTnT measure is sufficient to safely exclude CAD and identify patients with low risk for adverse cardiac events during a 30-day follow-up [27, 28]. Studies data highlighted the role of cTn as a measure of cardiovascular health, after identifying a uniform, optimized concentration limit that safely identified the low-risk group. Thus, high-risk patients can be treated immediately while the exit of low-risk patients from the emergency department reduces overcrowding and costs for health centers.
III Cardiac Biomolecule Detection Techniques
Over the last decades, great progress has been made in the detection and measurement of substances, especially in the field of biosensor development. A biosensor is a device for identifying a biological target molecule and consists of two components: a biological recognition element and a signal conversion unit, the physicochemical converter, which produces the information. The biological recognition element determines the degree of selectivity of the biosensor, while the sensitivity of the biosensor is greatly influenced by the inverter. In this part, sensors based on optical, magnetic, capacitive and amperometric detection techniques for cardiac biomarkers are presented.
i Optical Biosensors
Optical detection and measurement techniques have the ability to accurately obtain all the clinical properties of individual microparticles rapidly. Optical biosensors offer significant benefits over conventional analytical techniques as they allow the immediate, real-time detection of many biological and chemical substances. Their advantages are high specialization and sensitivity, fast detection. However, each of their numerous applications has its own requirements concerning the concentration, the production accuracy, the reuse time or the system cleaning.
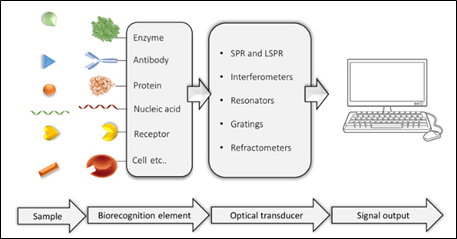
Optical detection is performed based on the interaction of the visual field with a bio-recognition element. Optical sensors are based on the change in phase, polarization or frequency of the incoming light and this change is related to the detection or measurement process. An optical biosensor, in other words, is a compact analytical device that contains a bio-recognition element integrated in an optical transducer system. The main purpose of the sensor is to generate a signal proportional to the concentration of a measured substance called an analyte. Various biological materials such as enzymes, antibodies, antigens, receptors, nucleic acids and tissues can be used as bio-recognition elements (Figure 3) [28]. Optical techniques can be divided into two categories: label-free and label-based. In the former, the detected signal is generated directly by the interaction of the analysed material with the inverter. Instead, the second category involves the use of a label and the optical signal is then generated by a colorimetric, fluorescent or luminous method [29]. Various optical biosensors have already been used to detect cardiac markers.
Figure 3: Schematic diagram of an optical biosensor [28].
An important application is that of Surface plasmon resonance biosensors (SPR). A plasmon is a quantum of oscillations in the plasma. The SPR effect, i.e., the surface resonance of plasmons, occurs on the surface of a conductive material (usually metal) at the interface of two media (e.g., glass and liquid) when it is illuminated by polarized light at a certain angle. This creates surface plasmons (and oscillations) and consequently a reduction in the intensity of the reflected light at a specific angle known as the resonant angle. By measuring the change in reflectance, angle or wavelengths over time, it is possible to obtain the sensor diagram. SPR devices provide direct, real-time and unlabeled changes in the refractive index on the sensor surface that are proportional to the biomolecule concentration. In a practical experiment one interaction component, e.g. the ligant is permanently attached to the surface of the chip and the other interacting component, e.g. the analyte or target substance, as it flows over the surface, binds to the ligant [29]. In 2007, Dutra and Kubo proposed an SPR sensor in which streptavidin was immobilized and used to bind anti-troponin T antibodies to detect human cardiac troponin T [30]. The system was able to measure cTnT without dilution of human serum and with good specificity.
Troponin I was also attempted to be measured by a fluorescence detection biosensor. In this type of sensor, the biological elements are labeled with fluorescent labels or dyes, and the presence of target molecules is determined when a change in fluorescence intensity or colour occurs. The colorimetric measurement is immediately visible, but the sensitivity and cost-effectiveness are much lower. In 2011, Song and colleagues used antibody-labeled fluoro-microspheres [31]. The cTnI target antigen was added to the cTnI capture antibody that was immobilized on the surface of the chip and then the antigen antibody was blocked. The immobilized cTnI was ligated to the antibody conjugated to the fluoro-microspheres, and the ligated fluoro-microspheres conjugates were measured directly using a conventional fluorescence microscope.
Recently, biosensors based on luminescence methods have also been developed for the detection of cardiac biomarkers and have been categorized into two types, chemiluminescence (CL) and electroluminescence (ECL). Electrochemiluminescence is the production of luminosity during the electrochemical reaction. In 2013, Li and colleagues reported the ability of a non-labeled ECL biosensor to detect cTnI by using gold nanoparticles that acted on bright light as antibody vectors [32]. Anti-cTnI antibodies, conjugated to the nanoparticles, were concentrated on a modified streptavidin-coated electrode. The molecules activated the electrode surface to generate ECL signals and were attached to the modified electrode to identify the cTnI target. The ECL reaction formed electron transport interactions and the intensity of the ECL was dependent on the cTnI concentration.
Surface-enhanced Raman scattering biosensors (SERS), is a technique that amplifies the vibration spectra of a molecule by several orders of magnitude. Amplification occurs on a metal surface that has nanoscale roughness and the molecules adsorbed on this surface can be amplified. The typical metals used are gold and silver. Surface preparation can be provided by electrochemical curing, metal coating of a nanostructured substrate or deposition of metal nanoparticles. In 2014, Chon et al. developed a SERS sensor for cTnI and CK-MB detection [33].
ii Magnetic Biosensors
Recently, magnetic techniques for detecting, measuring and identifying substances have emerged as a promising new technology platform for various sensors. These methods are generally based on observing the magnetic field of a magnetically labeled biomolecule that interacts with a complementary biomolecule attached to a sensor. Magnetic biosensors have several advantages. They are distinguished for their stability over time which is extremely important for labeling tests on tissues and organs while at the same time they do not present noise effects [34].
Magnetic nanobeads-MNBs or magnetic nanoparticles-MNPs are commonly used in magnetic biosensors. The magnetic core gives super-paramagnetic properties to MNBs, which cause them to move along dynamic lines under the influence of an external magnetic field and disperse after its removal. The polymer shell imparts to MNBs various functions that allow them to be further modified with various desired biochemical materials for specific binding to biological targets. Magnetic nanospheres are characterized by excellent paramagnetic properties, highly specialized surface area, rapid reactions, good biological compatibility and these advantages are the reason why they have often been reported for biological labeling, signal amplification, or both in various biosensors [35].
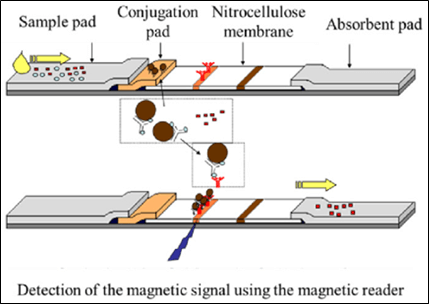
Magnetic Side Flow Sensors include a magnetic strip and a magnetic reader. A sample falls onto the sample pad of magnetic strip and flows through the coupling pad loaded with modified antibody capture MNBs. The targets bind to the MNBs to form the magnetic targets. The magnetic targets are then captured by detection antibodies that are immobilized on the test line, while the non-targets flow through the strip and are absorbed by the absorbent pad (Figure 4). The quantification is done with the magnetic reader consisting of an activation coil, a differential detection coil and a microcontroller. Magnetic side flow biosensors have been reported to detect viruses, bacteria and biomarkers as being distinguished for their rapid detection, easy operation and low cost [35]. It should be noticed that the sensitivity of a side flow test for the detection of troponin I by immobilizing antibodies to magnetic beads under optimized conditions has also been studied [36].
Figure 4: Magnetic Side Flow Sensor with conventional magnetic strip [35].
iii Capacitive Biosensors
Capacitance appears in the capacitor, an electrical element consisting of two separate plates, between which there is a dielectric material. In general, this concept describes how two conductive objects with a gap between them respond to a voltage difference applied to them. Capacitive sensors operation is based on changes in dielectric properties, charge distribution, dimension or shape, when the target (e.g., antigen / antibody complex) appears on the surface of an electrode [37]. Actually, in capacitive sensors, when a target molecule attaches to the receptor, the displacement of counter ions around the electrode leads to a change in capacitance. The greater the number of targets attached to the receptor, the greater the change in capacitance [38-40].
A key advantage of capacitive sensors over other detection approaches is their ability to detect different types of materials, without any wear. They are also highly sensitive and have been used successfully in the field of substance detection as biosensors for the detection of proteins, nucleotides, saccharides or small organic molecules. However, they are affected by environmental conditions and require electronic signal reading and processing circuits suitable for any application. Their special disadvantage compared to other types of direct biosensors is that the immobilization of the bio-recognition layer is more critical. If it is not sufficiently insulated, ions can move through the mattress causing the system to short circuit, leading to a reduction or absence of the signal [37].
One of the most common types of capacitive sensors used for substance detection, are the interdigitated electrodes (IDE), where the electrodes are in the form of a comb topology. These sensors are known as "interdigitated" or equivalent, "microstrip", "comb", "interdigital". The capacitance of an IDE sensor is given by the equation C = (η ∙ ε ∙ l ∙ t) / d, where η is the number of fingers, ε is the permeability, l is the length of the electrodes, t is the thickness of the electrodes and d is the distance between them. Of course, this capacitance calculation cannot be applied when nanoscale electrodes are used. Interdigitated capacitive sensor applications have been widely used in food inspection, humidity sensors, biosensor applications (direct detection of neurotransmitters, glucose, HIV antibodies), etc. as they stand out for their easy and low-cost construction but also for their high sensitivity [41].
At this point, it should be mentioned that a sensor chip consisting of interdigitated capacitor arrays has been developed to detect cardiac troponin-I (cTn-I). The chip was fabricated on a borosilicate glass substrate using reverse image photolithography (for pattern transfer) and DC sputtering (for gold deposition). The IDC surface was activated using GPTMS to immobilize antibodies. Antibody-antigen binding to the IDC surface resulted in a change in IDC sensor capacity. This change is directly related to the concentration of antigen that binds to the surface and aids in quantification. Specific and sensitive detection of cTn-I in real human serum was achieved with anti-cTn-I antibody immobilized on the IDC surface with a detection range of 50pg-1500 pg / ml respectively [42].
iv Amperometric Biosensors
Amperometry is the detection of the presence of ions in a solution based on the change in electric current. Such applications usually require a working electrode (microelectrode), a reference electrode, an auxiliary electrode, a voltage source and a device for measuring current and voltage. The amperometric method is based on the measurements of the changes in current passing through the electrode system over time, relative to the potential applied to the operating electrode. More specifically, amperometric biosensors operate by generating current when a potential is applied between two electrodes. The analyte is involved in a redox reaction followed by the measurement of current in an electrochemical cell. It has been shown that almost all biochemicals can now be detected amperometrically by their enzyme-catalyzed electro-oxidation or electro-reduction or their involvement in a bio-affinity reaction that allows electro-oxidation or electro-reduction. The analyte being measured or the biomolecule involved is changing the oxidation state at the electrode and the electron transfer signal being measured is proportional to the amount of redox active substances at the electrode. In amperometry, changes in current are directly monitored as a function of time [43].
It should be noted that an amperometric sensor has also been used to detect cardiac troponin I, using carbon electrodes coated with a graphene oxide derivative as a switching element [44]. This carboxylic acid enriched nanomaterial allowed the easy and high immobilization of the capture molecules on the electrode surface. The bioassay interface was mounted by covalently linking an aptamer to the electrode surface. The detection approach was based on the recognition of cTnI by the aptamer. The sensor was used for large-scale detection from 1.0 pg/mL to 1.0 μg/ml with a detection limit of 0.6 pg/mL. It showed high specificity, reproducibility and stability while it was also useful for the quantification of cTnI in human serum samples.
Conclusion and Future Perspectives
Preventing heart disease and reducing the number of deaths due to it, is a key issue that has been of concern to the global community for years. It is, therefore, a challenge for engineering science and technology, to contribute to medicine providing new tools and devices that could play a crucial role in tackling such serious health problems. It is essential to develop smart and sensitive diagnostic methods able to detect cardiac biomarkers even at low concentrations in order to prevent CVDs. Early diagnosis leads to an increase in the human survival rate by saving patients’ lives. In this context, biosensors seem to be a great economical and smart solution, facilitating people's lives in many ways. In this review, an attempt was made to summarize the available biosensing platforms for cardiac biomarkers detection. It appeared that recently developed sensors can fulfill their demands but they need to be expanded to surpass their malfunctions and disadvantages in order to be widely used. Funding support mechanisms are needed to facilitate moving such devices, from research and experimental stage to the commercial applications and make them available to everyone, especially to people at high-risk groups.
Conflicts of Interest
None.
Article Info
Article Type
Review ArticlePublication history
Received: Mon 06, Jun 2022Accepted: Mon 27, Jun 2022
Published: Sat 30, Jul 2022
Copyright
© 2023 Vasilopoulou Angeliki. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.JICOA.2022.02.02
Author Info
Vasilopoulou Angeliki Georgas A Hristoforou E
Corresponding Author
Vasilopoulou AngelikiSchool of Electrical and Computer Engineering, National Technical University of Athens, Athens, Greece
Figures & Tables
References
1. WHO CVD Risk Chart
Working Group (2019) World Health Organization cardiovascular disease risk
charts: revised models to estimate risk in 21 global regions. Lancet Glob
Health 7: e1332-e1345. [Crossref]
2. Roth GA, Mensah GA,
Johnson CO, Addolorato G, Ammirati E et al. (2020) Global Burden of
Cardiovascular Diseases and Risk Factors, 1990-2019: Update From the GBD 2019
Study. J Am Coll Cardiol 76: 2982-3021. [Crossref]
3. Dahlöf B (2010)
Cardiovascular disease risk factors: epidemiology and risk assessment. Am J
Cardiol 105: 3A-9A. [Crossref]
4. Johnson NB, Hayes
LD, Brown K, Hoo EC, Ethier KA et al. (2014) CDC National Health Report:
leading causes of morbidity and mortality and associated behavioral risk and
protective factors--United States, 2005-2013. MMWR Suppl 63: 3-27. [Crossref]
5. Wettersten N,
Maisel A (2015) Role of Cardiac Troponin Levels in Acute Heart Failure. Card
Fail Rev 1: 102-106. [Crossref]
6. Mair J, Lindahl B,
Hammarsten O, Müller C, Giannitsis E et al. (2018) How is cardiac troponin
released from injured myocardium? Eur Heart J Acute Cardiovasc Care 7:
553-560. [Crossref]
7. Flora GD, Nayak MK
(2019) A Brief Review of Cardiovascular Diseases, Associated Risk Factors and
Current Treatment Regimes. Curr Pharm Des 25: 4063-4084. [Crossref]
8. Fang J, Luncheon C,
Ayala C, Odom E, Loustalot F (2019) Awareness of Heart Attack Symptoms and
Response Among Adults — United States, 2008, 2014, and 2017. MMWR Morb
Mortal Wkly Rep 68: 101-106. [Crossref]
9. Lenfant C (2010)
Chest pain of cardiac and noncardiac origin. Metabolism 59: S41-S46. [Crossref]
10. Camaro C, de Boer
MJ (2015) STEMI or non-STEMI: that is the question. Neth Heart J 23:
243-244. [Crossref]
11. Fu DG (2015)
Cardiac Arrhythmias: Diagnosis, Symptoms, and Treatments. Cell Biochem
Biophys 73: 291-296. [Crossref]
12. A M, D B, S V et
al. (2022) Congestive Heart Failure. StatPearls. [Crossref]
13. Oparil S, Acelajado
MC, Bakris GL, Berlowitz DR, Cífková R et al. (2018) Hypertension. Nat Rev
Dis Primers 4: 18014. [Crossref]
14. Hankey GJ (2017)
Stroke. Lancet 389: 641-654. [Crossref]
15. Nguyen M, Cohen J
(2022) Peripheral Vascular Disease. Pain Med An Essent Rev 495-496.
16. Maxwell S (1999)
Emergency management of acute myocardial infarction. Br J Clin Pharmacol
48: 284-298. [Crossref]
17. Y S, L C (2021)
Electrocardiogram. StatPearls. [Crossref]
18. A M, AM I (2022) Holter
Monitor. StatPearls. [Crossref]
19. Perera P, Lobo V,
Williams SR, Gharahbaghian L (2014) Cardiac echocardiography. Crit Care Clin
30: 47-92. [Crossref]
20. Gajulapalli RD,
Aneja A, Rovner A (2012) Cardiac stress testing for the diagnosis and
management of coronary artery disease: a reference for the primary care
physician. South Med J 105: 93-99. [Crossref]
21. Heart disease -
Diagnosis and treatment - Mayo Clinic. 2021.
22. Thygesen K, Alpert
JS, Jaffe AS, Simoons ML, Chaitman BR et al. (2012) Third universal definition
of myocardial infarction. Eur Heart J 33: 2551-2567. [Crossref]
23. Ohtsuki I, Morimoto
S (2022) Troponin. Encycl Biol Chem Second Ed. 445-449.
24. The role of
troponin testing in primary care - Best tests 2009. 2021.
25. Troponin Test:
MedlinePlus Medical Test.
26. Al Mawed M, Mendrok
HC, John F, Högel R, Tebbe U (2020) [Troponin test in clinical daily practice -
Differences in the quality criteria for the diagnosis of acute myocardial
Infarction considering clinical leading symptoms: A cross-sectional study]. Dtsch
Med Wochenschr 145: e24-e31. [Crossref]
27. Pickering, Than MP,
Cullen L, Aldous S, Avest ET et al. (2017) Rapid Rule-out of Acute Myocardial
Infarction With a Single High-Sensitivity Cardiac Troponin T Measurement Below
the Limit of Detection: A Collaborative Meta-analysis. Ann Intern Med
166: 715-724. [Crossref]
28. Sandoval Y, Smith
SW, Love SA, Sexter A, Schulz K et al. (2017) Single High-Sensitivity Cardiac Troponin
I to Rule Out Acute Myocardial Infarction. Am J Med 130:
1076.e1-1083.e1. [Crossref]
29. Damborský P, Švitel
J, Katrlík J (2016) Optical biosensors. Essays Biochem 60: 91-100. [Crossref]
30. Dutra RF, Kubota LT
(2007) An SPR immunosensor for human cardiac troponin T using specific binding
avidin to biotin at carboxymethyldextran-modified gold chip. Clin Chim Acta
376: 114-120. [Crossref]
31. Song SY, Han YD,
Kim K, SS Y, Yoon HC (2011) A fluoro-microbead guiding chip for simple and
quantifiable immunoassay of cardiac troponin I (cTnI). Biosens Bioelectron
26: 3818-3824. [Crossref]
32. Li F, Yu Y, Cui H,
Yang D, Bian Z (2013) Label-free electrochemiluminescence immunosensor for
cardiac troponin I using luminol functionalized gold nanoparticles as a sensing
platform. Analyst 138: 1844-1850. [Crossref]
33. Abdolrahim M,
Rabiee M, Alhosseini SN, Tahriri M, Yazdanpanah S (2015) Development of optical
biosensor technologies for cardiac troponin recognition. Anal Biochem
485: 1-10. [Crossref]
34. Nabaei V,
Chandrawati R, Heidari H (2018) Magnetic biosensors: Modelling and simulation. Biosens
Bioelectron 103: 69-86. [Crossref]
35. Wang L, Lin J
(2020) Recent Advances on Magnetic Nanobead Based Biosensors: from Separation
to Detection. TrAC - Trends in Analytical Chemistry 128: 115915.
36. Ryu Y, Jin Z, Kang
MS, Kim HS (2011) Increase in the detection sensitivity of a lateral flow assay
for a cardiac marker by oriented immobilization of antibody. BioChip J
5: 193-198.
37. Berggren C,
Bjarnason B, Johansson G (2001) Capacitive Biosensors. Electroanalysis
13: 173-180.
38. Ertürk G,
Mattiasson B (2017) Capacitive Biosensors and Molecularly Imprinted Electrodes.
Sensors (Basel) 17: 390. [Crossref]
39. Bekić SS, Kuzminac
I, Bjedov S, Ajduković J, Savić M et al. (2022) Use of Fluorescent Yeast-Based
Biosensors for Evaluation of the Binding Affinities of New Steroid Hormone and
Bile Acid Derivatives for Select Steroid Receptors. Eng Proc 16: 4.
40. Georgas A, Lampas
E, Houhoula DP, Skoufias A, Patsilinakos S et al. (2022) ACE2-based capacitance
sensor for rapid native SARS-CoV-2 detection in biological fluids and its
correlation with real-time PCR. Biosens Bioelectron 202: 114021. [Crossref]
41. Chetpattananondh K,
Tapoanoi T, Phukpattaranont P, Jindapetch N (2014) A self-calibration water
level measurement using an interdigital capacitive sensor. Sensors Actuators
A Phys 209: 175-182.
42. Mahalingam D,
Gurbuz Y, Qureshi A, Niazi JH (2015) Design, fabrication and performance
evaluation of interdigital capacitive sensor for detection of Cardiac Troponin-I
and Human Epidermal Growth Factor Receptor 2.
43. Sadeghi SJ (2013) Amperometric Biosensors. Encycl Biophys 61-67.
44. Villalonga A, Estabiel I, Pérez-Calabuig AM, Mayol B, Parrado C et al. (2021) Amperometric aptasensor with sandwich-type architecture for troponin I based on carboxyethylsilanetriol-modified graphene oxide coated electrodes. Biosens Bioelectron 183: 113203. [Crossref]
