Ultrasonography-Guided Cannula Method for PMMA Injection in Aesthetic Gluteal Augmentation: A Technical Note
A B S T R A C T
Purpose: Revolumization with deep injections has become increasingly common, even in high-risk areas for intravascular infusion or nerve injury. This study aimed to assess the usefulness of the ultrasonography-guided cannula method for preventing side effects during the injection of polymethylmethacrylate (PMMA) fillers. Real-time ultrasonography was used to monitor the injection of PMMA as a filler in the gluteus for aesthetic purposes. This way, adjacent anatomical structures such as arteries, veins, and nerves could be seen during the whole procedure.
Materials and Methods: This study describes the technique used for ultrasound-guided injections of PMMA in the gluteal region of two patients. Patients 1 and 2 were injected with 300 mL and 220 mL of the filler, respectively. Injections were administered intramuscularly and subcutaneously into sites that had been marked before the procedure. While one professional performed the injections, another guided them with real-time ultrasound imaging, using the same equipment for both patients. The location and depth of blood vessels and nerves were determined before the procedure, and the insertion of the cannula was accompanied by ultrasound imaging.
Results: The PMMA filler, as well as the cannula, blood vessels, and nerves, was visualized using ultrasound. Some differences were observed between the cases. In the first patient, a small amount of PMMA from a previous procedure could be found (hyperechoic images), while the second one had had no filler injection before.
Conclusion: Based on these cases, ultrasonography can be seen as a complementary tool to monitor tissue filler injections, especially in the gluteal region, thus, avoiding possible vascular and nervous injuries. Real-time ultrasound makes the procedure safer, protecting regional structures from lesions. The ultrasonography-guided method succeeded in localizing the cannula, blood vessels, and the sciatic nerve, allowing for safe injection of PPMA filler into a deep gluteal region.
Keywords
Dermal fillers, aesthetics, PMMA, ultrasonography
Introduction
Doppler ultrasonography (US) is an imaging modality that has been widely used in the medical field to evaluate body structures. However, its use in the gluteal region is still unusual. Due to high-frequency, high-definition, and portable US devices, this imaging method has become more widespread among professionals of various specialties [1].
Nevertheless, despite having gained importance due to the large number of cosmetic procedures being performed today, the literature indicates that US is still not part of the relevant protocols, even though US could assist in pre-procedural diagnostic evaluation, follow-up, and post-procedure assessment of eventual complications [2]. With this in mind, this work sought to demonstrate the applicability of US as a highly useful auxiliary tool in aesthetic procedures performed in the gluteal region.
Methods
This is an observational, retrospective study of the case reports of two patients who underwent gluteal BioPlasty with PMMA associated with the use of ultrasound. This study was approved by the Universidade de Santa Cruz do Sul Research Ethics Committee under protocol number CAAE 60038022.3.0000.5343.
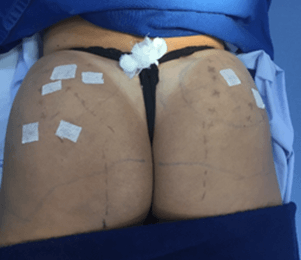
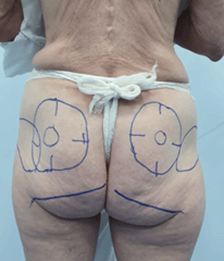
Two patients received ultrasound-guided injections of PMMA in the gluteal region. Patients 1 and 2 were injected intramuscularly and subcutaneously with 300 mL and 220 mL of the filler, respectively. With the patient in the prone position, real-time ultrasound was used to identify the subcutaneous tissue, fascial layers, veins, arteries, and nerves of the buttocks. The injection sites were marked (skin markings: ++=artery, -- = nerve, and deep structures) in the region before the procedure (Figures 1A, 1B, 1C, 3, 4, 5A and 5B). While one professional performed the injections with a blunt cannula, another guided them with real-time ultrasound imaging, using the same equipment for both patients. The location and depth of blood vessels and nerves were determined before the procedure, and the insertion of the cannula was done with real-time ultrasound guidance. During the procedure, the physician saw the images on a screen to follow the planes in which the cannula was being introduced and the PMMA injected.
The ultrasound images facilitate the identification of important structures before the procedure. During the procedure, the introduction of the cannula and PMMA injection are monitored in real-time with US. This technique makes the procedure safer by allowing the doctor who knows the anatomical points to prevent possible vascular-nervous injuries with real-time ultrasound during the procedure.
Results
Figures 1A, 1B, 1C, 2, 3, 4, 5A and 5B show the markings of arteries and nerves on the skin of the patients. In US, hard tissues interfere with the passage of US waves, producing a strong echo, as evidenced by hyperechoic margins. In contrast, soft tissues offer less resistance to waves and appear hypoechoic in intensity. In vivo, a region filled with PMMA appears as a hyperechoic delimited structure of high intensity or relatively high echogenicity (i.e., whiter). On the other hand, muscle and fat areas appear hypoechoic without defined limits. Such differences have already been established by magnetic resonance and ultrasound imaging in cases using hyaluronic acid as a dermal filler [3].
Figure 1: A-C) Ultrasound-guided marking to determine the course of arteries and the sciatic nerve. Patient 1.
Figure 2: Post-procedure. Patient 1.
Figure 3: Markings for the filler injections. Patient 2.
Figure 4: Ultrasound-guided marking to determine the course of arteries and the sciatic nerve. Patient 2.
Figure 5: A & B) Ultrasound-guided marking to determine the course and depth of arteries and the sciatic nerve. Patient 2.
The brightness of PMMA depends on how dense the product is, among other factors. Nevertheless, PMMA can be easily differentiated from anatomical structures and surrounding soft tissues, just as occurs with hyaluronic acid [4]. For locating and differentiating PMMA, it is also helpful for the operator to have a thorough knowledge of US techniques and the anatomy of the region.
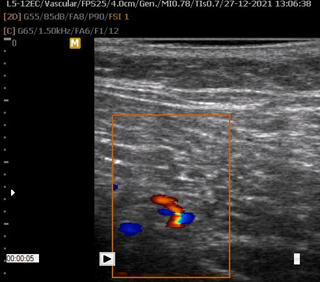
Regarding both cases in the present study, the US imaging performed before the procedure showed a useful location of arteries, veins, and nerves. The invasion of vital structures, such as blood vessels, was avoided thanks to the Doppler ultrasound-guided procedure, carried out using a linear probe of 5 to 12 Mhz. The color Doppler mode was used before and during the injection process to assess vascular structures. Ultrasound imaging of the gluteus area was performed to identify the PPMA, arteries, veins, sciatic nerve, and subcutaneous tissue (Figures 6-13).
Figure 6: PMMA in the subcutaneous tissue (hyperechogenic areas in the subcutaneous tissue). Aponeurosis of the gluteus maximus (hyperechogenic line in white). Patient 1.
Figure 7: Identification of arteries and their depths. Patient 2, who had no previous injection of PMMA.
Figure 8: Right sciatic nerve – hypoechoic tubular structure seen without Doppler flow under the muscle (hyperechoic lines interspersed with hypoechoic areas).
Figure 9: Left sciatic nerve – hypoechoic tubular structure seen without Doppler flow under the muscle (hyperechoic lines interspersed with hypoechoic areas).
Figure 10: Identification of arteries and their depths. Patient 2, who had had no previous injection of PMMA.
Figure 11: Identification of superior gluteal artery and vein, aponeuroses, subcutaneous tissue, and skin using Doppler US.
Figure 12: Identification of the superior gluteal artery and determination of its depth (1.04 cm deep to the skin) using Doppler US with a linear probe of 5 to 12 Mhz.
Figure 13: Identification of the inferior gluteal artery and determination of its depth (3.36 cm deep to the skin) using Doppler US.
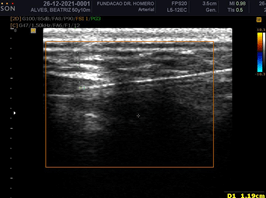
The position of the cannula and the injection of PMMA, as well as the volume of product injected into the area, could be observed (Figures 14 & 15). Well-defined hyperechoic pockets of PMMA were formed and proved compatible with both the cannula path and the frequency of injection by the clinician (Figures 16 & 17). An important observation was the absence of vascular and nervous compression by the filler, as evaluated by Doppler US.
Figure 14: Injection cannula and PMMA in the gluteus maximus. Hyperechoic area at the tip of the cannula at the time of injection. Patient 1.
Figure 15: PMMA injection cannula (hyperechoic line) and intramuscular hyperechoic areas of PMMA. Depth measuring of the cannula tip.
Figure 16: Patient with previous injection of PMMA. Doppler US is used to visualize the injection (distal to the cannula in red) in an area where there is no PMMA and to avoid the gluteal arteries (red areas in the middle third of the cannula). Patient 1.
Figure 17: Doppler US showing the injection of PMMA in an area without the filler. Patient 2.
Discussion
An exploratory study was carried out, providing case evidence of the usefulness of the ultrasonography-guided cannula method for preventing vascular and nervous trauma, which could be caused by filler injections. PMMA soft-tissue fillers are generally safe and popularly used as injectable cosmetic fillers for gluteal augmentation [5]. However, thus far, the use of US to guide these injections is not a common practice. An appropriate technique to guide the injections of PMMA was demonstrated.
Although knowledge of the anatomy of the injection regions can minimize risks, the depth and distribution of the gluteus artery branches and nerves can sometimes vary, as occurs in facial anatomy [6, 7]. In the gluteal area, the combination of larger volumes of tissue filler and deeper injections has become increasingly common to achieve revolumization. Therefore, the anatomical structures involved must be identified [8].
In the cases presented in this article, US was used to make an imaging diagnosis and to aid in surgical procedures. The US images allowed for the precise visualization of the PMMA injection site, as well as the adjacent structures, as seen by Wortsman, who identified these structures at the injection site (Figures 16 & 17) [9].
The image of the filler was well-defined and bright, with no interference from other structures. As observed in the study by Greene and Sidle, the material was easily differentiated from the adjacent anatomical structures (Figures 14 & 15) [10]. PMMA was injected between the muscle bundles and subcutaneous tissue or intramuscularly, when necessary, to augment the patients’ buttocks.
The limitations of this exploratory study were that the cases of only two patients were studied, which are not enough to demonstrate the technique, and that the cost and duration of the procedures were not taken into consideration. This study showed the potential benefits of the ultrasound-guided cannula method, reinforcing its importance. Under ultrasonography guidance, blood vessels, nerves, and the tip of the cannula can be easily visualized, thereby reducing the risk of injuring these structures.
Conclusion
Based on the description of the technique presented in this study, ultrasonography can be used as a complementary tool to monitor tissue filler injections, especially in the gluteal region, and to avoid possible vascular and nervous injuries. Real-time ultrasound observation makes the procedure safer, protecting regional structures from lesions. The ultrasound-guided method was successful in detecting the cannula, blood vessels, and the sciatic nerve, thus, allowing safe injection of PMMA filler into a deep gluteal region.
Acknowledgment
We thank Augusta Ketzer Menezes for the English assistance.
Article Info
Article Type
Technical NotePublication history
Received: Tue 29, Nov 2022Accepted: Mon 26, Dec 2022
Published: Wed 18, Jan 2023
Copyright
© 2023 Honorio Sampaio Menezes. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2022.12.02
Author Info
Honorio Sampaio Menezes Ozorio Sampaio Menezes
Corresponding Author
Honorio Sampaio MenezesHospital Dr Homero Lima Menezes, State of Rio Grande do Sul, Brazil
Figures & Tables












References
1. Schelke LW, Elzen HJVD, Erkamp PPM, Neumann HAM (2010) Use of ultrasound
to provide overall information on facial fillers and surrounding tissue. Dermatol
Surg 36: 1843-1851. [Crossref]
2. Cansancao AL, Green AC, Vidigal RA, Rodriguez RL, Richard A D'Amico RA
(2018) Real-Time Ultrasound-Assisted Gluteal Fat Grafting. Plast Reconstr
Surg 142: 372-376. [Crossref]
3. Josse G, Haftek M, Gensanne D, Turlier V, Mas A et al. (2010) Follow up
study of dermal hyaluronic acid injection by high frequency ultrasound and
magnetic resonance imaging. J Dermatol Sci 57: 214-216. [Crossref]
4. Goh AS, Kohn JC, Rootman DB, Lin JL, Goldberg RA (2014) Hyaluronic acid
gel distribution pattern in periocular area with high-resolution ultrasound
imaging. Aesthet Surg J 34: 510-515. [Crossref]
5. Chacur R, Menezes
HS, da Silva Chacur NMB, Alves DD, Mafaldo RC et al. (2019) Gluteal
Augmentation with Polymethyl Methacrylate: A 10-year Cohort Study. Plast
Recons Surg Glob Open 7: e2193. [Crossref]
6.
Scholten HJ, Pourtaherian A, Mihajlovic N, Korsten
HHM, Bouwman RA (2017)
Improving needle tip identification during
ultrasound-guided procedures in anaesthetic practice. Anaesthesia 72: 889-904. [Crossref]
7. Rocha LPC, de Carvalho Rocha
T, de Cássia Carvalho Rocha S, Henrique PV, Manzi FR et al. (2020) Ultrasonography for long-term evaluation of hyaluronic acid filler
in the face: A technical report of 180 days of follow-up. Imaging Sci Dent 50:
175-180. [Crossref]
8. Ghavami A, Villanueva NL, Amirlak B (2018) Gluteal Ligamentous Anatomy
and Its Implication in Safe Buttock Augmentation. Plast Reconstr Surg
142: 363-371. [Crossref]
9. Wortsman X (2015) Identification and Complications of Cosmetic Fillers: Sonography First. J Ultrasound Med 34: 1163-1172. [Crossref]
10. Greene JJ, Sidle DM (2015) The Hyaluronic Acid Fillers: Current Understanding of the Tissue Device Interface. Facial Plast Surg Clin North Am 23: 423-432. [Crossref]
