Ten-Year Review of Pediatric Microbial Keratitis and Applicability of the 1-2-3- Rule
A B S T R A C T
Purpose: To describe the current spectrum of bacterial etiologies, clinical features, medical management and visual outcomes of pediatric community-acquired bacterial keratitis and to evaluate the usefulness of the 1-2-3-Rule in predicting vision loss.
Methods: A retrospective review was performed of consecutive cases of pediatric keratitis treated at a single institution over a 10-year period. The 1-2-3-Rule was applied retrospectively and analyzed to determine its accuracy in predicting vision loss.
Results: Seventy-seven cases were identified. Pseudomonas was the most common organism identified followed by coagulase-negative staphylococcus and staphylococcus aureus. Only one case of staphylococcus aureus keratitis was methicillin resistant. Most (n=66) of the 74 verbal patients regained 20/30 or better visual acuity and 5 had final visual acuity of 20/100 or worse. The 1-2-3-Rule yielded high sensitivity and negative predictive values in our pediatric patients.
Conclusions: Methicillin-resistant staphylococcus aureus (MRSA) is an infrequent pathogen in cases of pediatric community-acquired bacterial keratitis. Pseudomonas continues to be a common etiology. Most pediatric patients retain good visual acuity with treatment. When applied at a patient’s initial evaluation, the 1-2-3-Rule test may be helpful in predicting which pediatric patients will retain good visual acuity following treatment.
Keywords
Keratitis, corneal ulcer, pediatric, children
Introduction
Infectious keratitis is often a sight threatening disease and the risk for poor visual outcome may be higher in children due to 1) limited verbal communication and/or deliberate under-reporting of symptoms, 2) limited cooperation with eye examinations, corneal culture collection and topical medication instillation and 3) amblyopia due to media opacity or anisometropia [1-6]. Over 30,000 cases of infectious keratitis occur in the US per year but only 11-13% are pediatric cases [7-10]. The initial management of infectious keratitis may or may not include corneal culture and fortified antibiotic drops. The American Academy of Ophthalmology (AAO) management guidelines indicate that culture should be performed if a corneal ulcer is large (>2 mm), central, involves stromal melting, has atypical appearance or appears to be sight-threatening; however, indications such as “central” and “sight-threatening” are not defined [11]. Other severity grading systems vary regarding classifications of ulcer size, location, density, depth, scleral involvement and degree of anterior chamber reaction [4, 12-15].
The AAO guidelines recommend use of topical fortified vancomycin in settings in which MRSA “is likely to occur” since almost 80% of ocular MRSA isolates are resistant to fluoroquinolones [11-17]. Although MRSA is an increasingly common cause of keratitis among adults, there is little data regarding the frequency of MRSA keratitis in children in the US [1, 6, 11]. The primary aim of the present study is to describe the current spectrum of bacterial etiologies, clinical features, management and visual outcomes of pediatric community-acquired microbial keratitis presenting to a single US academic medical center. A second goal is to evaluate the potential utility of the 1-2-3-Rule test in predicting vision loss in pediatric cases of microbial keratitis [16].
Patients and Methods
This study was approved by the Institutional Review Board of Cincinnati Children’s Hospital Medical Center and conformed to the requirements of the United States Health Insurance Portability and Accountability Act. A retrospective chart review was performed of microbial keratitis cases including all patients aged ≤ 21 years presenting to the pediatric ophthalmology outpatient clinic of Cincinnati Children’s Hospital Medical Center during the period of November 1, 2009 to October 31, 2019. The study population was community-acquired non-viral microbial keratitis. Patients with hospitalization during the 30 days prior to keratitis, neurodevelopmental disability and chronic exposure keratitis were excluded since these patients were deemed more likely to have exposure to hospital flora or chronic topical antibiotic use. Demographics, clinical examination features, corneal culture results, outpatient and inpatient antibiotic treatment and visual outcome data were collected. The 1-2-3-Rule test was retrospectively applied to stratify cases as either “potentially sight threatening” (PST) or “rarely sight threatening” (RST) based on the documented initial examination findings [16]. The 1-2-3-Rule test criteria are listed in (Table 1). Sensitivity, specificity, positive predictive value and negative predictive value of the 1-2-3-Rule test were calculated using visual acuity outcomes in a series of pediatric patients with microbial keratitis.
Table 1: The 1-2-3-Rule for Corneal Ulcer Severity Classification [16].
|
Potentially Sight Threatening (PST) – ANY ONE of the following must be present:
|
|
Rarely Sight Threatening (RST) – ALL of the following must be present:
|
Results
A total of 77 patients (44 males, 33 females) were identified. Mean age was 14.3 years (range 2 months to 21 years). Right eyes were affected in 42 patients and left eyes in 35 patients. No patients had bilateral corneal ulcers. Clinical features of corneal ulcers at presentation are listed in (Table 2). Recent contact lens use was positive in 68 patients. No contact lenses were worn in 9 patients and of these 9 patients, all but one was under the age of 12 years. In the patients without contact lens wear, other risk factors were present: trauma (n=4) and severe blepharitis (n=2).
Corneal cultures were performed in 61 of the 77 cases and were found to be positive in 36 cases (Table 3). Pseudomonas was the most common isolate (n=16) and, in all cases, was sensitive to ceftazidime, gentamicin, piperacillin/tazobactam and tobramycin. All patients with Pseudomonas keratitis were contact lens wearers. All cases of Pseudomonas keratitis were treated with topical fortified tobramycin and either topical ceftazidime, moxifloxacin or ciprofloxacin.
Table 2: Ulcer Characteristics at Initial Presentation.
|
Characteristic |
Number of Patients |
|
Size |
|
|
< 1mm |
47 |
|
1mm to < 2mm |
8 |
|
≥ 2mm |
22 |
|
Location |
|
|
Central (≤ 3 mm from center) |
21 |
|
Paracentral (> 3 mm from center) |
32 |
|
Peripheral (limbal) |
24 |
|
Anterior Chamber Reaction |
|
|
< 1+ cells |
51 |
|
≥ 1+ cells |
26 |
Table 3: Positive Corneal Culture Results.
|
Organism |
Positive Culture Cases Number of cases (% of total cases) |
|
Pseudomonas species |
16 (21) |
|
Coagulase negative Staphylococcus |
9 (12) |
|
Methicillin-sensitive Staphylococcus aureus |
3 (3.9) |
|
Methicillin-resistant Staphylococcus aureus |
1 (1.3) |
|
Streptococcus species |
2 (2.6) |
|
Serratia marcescens |
3 (3.9) |
|
Enterococcus casseliflavus |
1 (1.3) |
|
Moraxella catarrhalis |
1 (1.3) |
|
Mycobacterium species |
1 (1.3) |
|
Stenotrophomonas maltophilia |
1 (1.3) |
|
Total |
36 (47) |
*some patients had cultures that yielded more than one organism.
MRSA was identified in 1 case. The MRSA organism isolated was resistant to erythromycin and nafcillin but sensitive to clindamycin, gentamicin, trimethoprim/sulfamethoxazole, tetracycline and vancomycin. Fluoroquinolone sensitivity testing was not performed in accordance with the hospital clinical laboratory protocol. Management of infectious keratitis cases was provided by multiple ophthalmologists and therefore treatment regimens varied. Topical fortified antibacterial eye drops were used in 53 cases and included vancomycin 25 mg/mL, ceftazidime 50 mg/mL, tobramycin 14 mg/mL and/or bacitracin 2000 units/mL. A nonfortified commercially available antibiotic (ciprofloxacin 0.3%, moxifloxacin 0.5% or tobramycin 0.3%) alone was used in 24 patients. Of the 53 patients treated with fortified antibiotics, 21 were admitted to the hospital for administration of hourly eye drops.
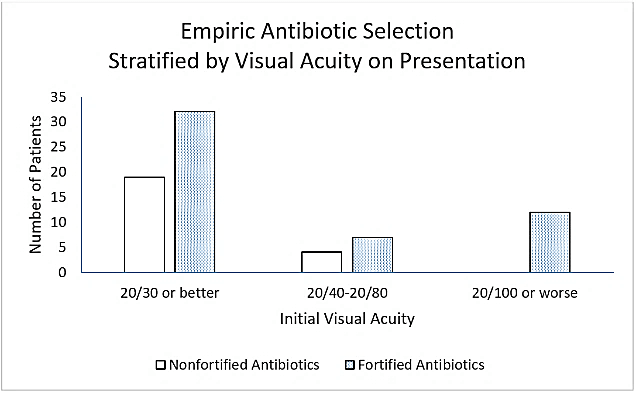
Figure 1 shows the number of patients treated with fortified and nonfortified antibiotics across 3 levels of initial visual acuity. Fortified antibiotics were used more often than nonfortified antibiotics for all categories of initial visual acuity. In 7 patients, empiric topical antifungals were used in conjunction with fortified antibacterial but none of these patients were found to be culture-positive for fungus.
Figures 1: Treatment with fortified and nonfortified antibiotics stratified by initial visual acuity.
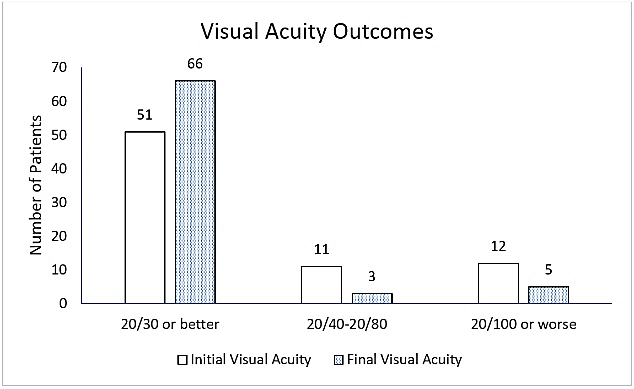
Figures 2: Initial and final visual acuity in eyes with corneal ulcers.
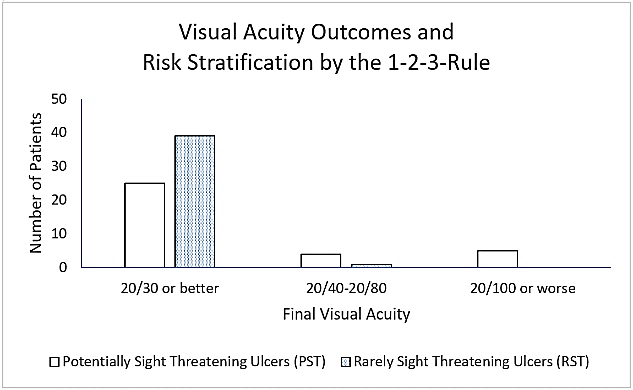
Figures 3: Risk of vision loss stratified using the 1-2-3-Rule and resulting final visual acuity.
Figure 2 shows initial and final visual acuities of all verbal patients (n=74). Five patients had final visual acuity of 20/100 or worse. All 5 of these patients were contact lens wearers, had large (≥ 5.5 mm) central Pseudomonas ulcers and were later referred to a cornea specialist for non-emergent optical penetrating keratoplasty. The results of the 1-2-3 Rule test were as follows. In 37 cases, the initial examination findings met the definition of “potentially sight threatening” (PST) and 40 cases met the definition of “rarely sight threatening” (RST) as defined by the 1-2-3-Rule. All patients classified as RST had final visual acuity of 20/30 or better except for one 3-year old patient who had final visual acuity of 20/50 that was attributed to amblyopia rather than corneal scar.
Figure 3 shows the final optotype visual acuities for 74 verbal patients that were used for analysis of the 1-2-3-Rule. Three pre-verbal patients all had PST ulcers but were not included in the 1-2-3-Rule analysis since optotype acuity was not recorded. Sensitivity and specificity of the 1-2-3-Rule were 90% and 61%, respectively. Positive and negative predictive values were 26% and 98%, respectively.
Discussion
Fluoroquinolone resistant MRSA keratitis in adults is increasingly common [17]. The AAO PPP guidelines state, “Due to the increasing resistance of MRSA to topical fluoroquinolones, combination therapy including vancomycin should be considered in a setting in which this etiological organism is likely to occur [11].” Over the past 10 years, there has been little published on the topic of pediatric microbial keratitis case in the US [6]. In a study of pediatric microbial keratitis in South Florida, only 2 of 107 cases involved MRSA [6]. Studies performed outside of the United States have demonstrated mixed results. In Vancouver, Noureddin et al. found no cases MRSA keratitis in their series of 17 cases of pediatric microbial keratitis [5]. A study performed in Taiwan reported 9 cases of MRSA in a series 68 cases of pediatric keratitis [18]. In Hong Kong, Young et al. found MRSA in 1 of 18 pediatric keratitis cases [19].
In our study performed in the mid-western United States, we found MRSA in 1 of 61 cultured cases. We acknowledge that 16 patients in our series were not cultured and that it is possible that MRSA was present in some of those cases. Only 2 of the 16 patients without cultures received empiric vancomycin and all 16 of those patients achieved resolution and 20/20 visual acuity. Given the current AAO guidelines and the low rate of MRSA in pediatric community-acquired microbial keratitis in our study, we believe that initial treatment does not need to routinely include vancomycin in our region. Some difficulties in initiating fortified vancomycin may include limited compounding pharmacy availability, cost, corneal toxicity and limited shelf life [20]. Despite our results, we understand that providers may tend to use as initial treatment due to their awareness of MRSA found in adult cases, risk of amblyopia due to a prolonged infiltrate or scar, medical-legal concerns, limited patient cooperation with evaluation and treatment and/or limited access to cornea specialists who treat children.
Pseudomonas was the most common bacterial etiology among our pediatric patients and contact lens use was a common risk factor. These findings are similar to other studies performed in the United States, Canada and Asia [5, 6, 18, 19, 21]. Although all Pseudomonas cases in our series were double covered with fortified tobramycin plus another antipseudomonal agent, 25% of patients went on to have poor visual outcomes due to corneal scar requiring optical penetrating keratoplasty for visual rehabilitation.
The AAO guidelines state that most corneal ulcers are managed without cultures [11]. The AAO guideline recommendation is to perform corneal cultures in cases in which an ulcer is central, large, chronic, unresponsive to antibiotic therapy or extends to the mid-stroma [11]. Some cornea specialists recommend corneal culture for all patients with corneal ulcers; however, surveys indicate that cornea specialists’ culture only 58% of their cases and non-cornea specialists’ culture only 22% [16, 17, 20]. Approximately 18% of ophthalmologists do not have eye culture supplies in their clinics [20]. Multiple grading scales have been proposed to increase the objectivity of the clinical evaluation of corneal ulcer and determine severity [4, 12, 14, 15]. The AAO guidelines do not define terms such as “central” and “sight-threatening” to aid the ophthalmologist in deciding whether or not to culture at initial presentation [11]. For adult patients, the 1-2-3-Rule provides more precise definitions of infiltrate size and location which may aid in the decision of whether or not to perform corneal culture at initial presentation [16].
In the present study, we applied the 1-2-3-Rule to pediatric cases and found high sensitivity to detect potentially sight threatening ulcers and a high negative predictive value of the 1-2-3-Rule. A negative predictive value of 98% suggests that an RST ulcer with treatment carries only a 2% chance of vision loss. The low positive predictive value of the 1-2-3-Rule suggests that many of the pediatric patients who meet criteria of PST on initial exam, may have no vision loss after treatment despite an initially guarded prognosis. While acknowledging this limitation, we believe that the 1-2-3-Rule may be of value in deciding whether or not corneal culture and fortified antibiotics are needed at the time of initial presentation or whether a more conservative approach with close follow-up is adequate. A prospective study would help delineate its usefulness.
In the present study, one patient developed amblyopia due to a temporary period of occlusion of the visual axis by a corneal opacity. It is noteworthy that AAO guidelines and other AAO publications do not describe deprivational and anisometropic amblyopia as potential complications of microbial keratitis in children [11, 17, 22]. We believe that amblyopia should be included in such educational publications to remind providers to diagnose and treat amblyopia during or after prolonged microbial keratitis in children.
We recognize that the retrospective nature of the present study limits the generalizability of the findings, but the study offers some insights into a common pediatric eye disease that is not widely studied in the US. Prospective, region-specific data would be most applicable to clinical care but at this type of data is not available in many parts of the world.
Conclusion
Pseudomonas was the most common isolate in cases of community-acquired pediatric microbial keratitis. MRSA was very uncommon although fortified vancomycin was commonly prescribed empirically. The vast majority patients with microbial keratitis did well with medical management; however large, central, Pseudomonas ulcers required surgery despite aggressive double coverage with fortified antipseudomonal agents. The 1-2-3-Rule may be helpful in identifying pediatric patients with corneal ulcers that are “rarely sight threatening” and therefore have a good visual prognosis with treatment.
Funding
None.
Acknowledgment
Supported in part by a Challenge Grant from Research to Prevent Blindness, Inc., to the University of Cincinnati Department of Ophthalmology (Karl C. Golnik, MD, Chairman).
Article Info
Article Type
Review ArticlePublication history
Received: Sat 11, Apr 2020Accepted: Fri 24, Apr 2020
Published: Wed 29, Apr 2020
Copyright
© 2023 W. Walker Motley. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2020.04.11
Author Info
W. Walker Motley Wejdan Al-Thawabieh
Corresponding Author
W. Walker MotleyCincinnati Children’s Hospital Medical Center, University of Cincinnati, Abrahamson Pediatric Eye Institute, Cincinnati, Ohio, USA
Figures & Tables
Table 1: The 1-2-3-Rule for Corneal Ulcer Severity Classification [16].
|
Potentially Sight Threatening (PST) – ANY ONE of the following must be present:
|
|
Rarely Sight Threatening (RST) – ALL of the following must be present:
|
Table 2: Ulcer Characteristics at Initial Presentation.
|
Characteristic |
Number of Patients |
|
Size |
|
|
< 1mm |
47 |
|
1mm to < 2mm |
8 |
|
≥ 2mm |
22 |
|
Location |
|
|
Central (≤ 3 mm from center) |
21 |
|
Paracentral (> 3 mm from center) |
32 |
|
Peripheral (limbal) |
24 |
|
Anterior Chamber Reaction |
|
|
< 1+ cells |
51 |
|
≥ 1+ cells |
26 |
Table 3: Positive Corneal Culture Results.
|
Organism |
Positive Culture Cases Number of cases (% of total cases) |
|
Pseudomonas species |
16 (21) |
|
Coagulase negative Staphylococcus |
9 (12) |
|
Methicillin-sensitive Staphylococcus aureus |
3 (3.9) |
|
Methicillin-resistant Staphylococcus aureus |
1 (1.3) |
|
Streptococcus species |
2 (2.6) |
|
Serratia marcescens |
3 (3.9) |
|
Enterococcus casseliflavus |
1 (1.3) |
|
Moraxella catarrhalis |
1 (1.3) |
|
Mycobacterium species |
1 (1.3) |
|
Stenotrophomonas maltophilia |
1 (1.3) |
|
Total |
36 (47) |
*some patients had cultures that yielded more than one organism.
References
- Cruz OA, Sabir SM, Capo H, Alfonso EC (1993) Microbial keratitis in childhood. Ophthalmology 100: 192-196. [Crossref]
- Clinch TE, Palmon FE, Robinson MJ, Cohen EJ, Barron BA et al. (1994) Microbial keratitis in children. Am J Ophthalmol 117: 65-71. [Crossref]
- Kunimoto DY, Sharma S, Reddy MK, Gopinathan U, Jyothi J et al. (1998) Microbial keratitis in children. Ophthalmology 105: 252-257. [Crossref]
- Parmar P, Salman A, Kalavathy CM, Kaliamurthy J, Thomas PA et al. (2006) Microbial keratitis at extremes of age. Cornea 25: 153-158. [Crossref]
- Noureddin GS, Sasaki S, Butler AL, Tilley P, Roscoe D et al. (2016) Paediatric infectious keratitis at tertiary referral centres in Vancouver, Canada. Br J Ophthalmol 100: 1714-1718. [Crossref]
- Rossetto JD, Cavuoto KM, Osigian CJ, Chang TCP, Miller D et al. (2017) Paediatric infectious keratitis: a case series of 107 children presenting to a tertiary referral centre. Br J Ophthalmol 101: 1488-1492. [Crossref]
- Ormerod LD, Murphree AL, Gomez DS, Schanzlin DJ, Smith RE (1986) Microbial keratitis in children. Ophthalmology 93: 449-455. [Crossref]
- Pepose JS, Wilhelmus KR (1992) Divergent approaches to the management of corneal ulcers. Am J Ophthalmol 114: 630-632. [Crossref]
- Joslin CE, Tu EY, McMahon TT, Passaro DJ, Stayner LT et al. (2006) Epidemiological characteristics of a Chicago-area Acanthamoeba keratitis outbreak. Am J Ophthalmol 142: 212-217. [Crossref]
- Hsiao CH, Yeung L, Ma DH, Chen YF, Lin HC et al. (2007) Pediatric microbial keratitis in Taiwanese children: a review of hospital cases. Arch Ophthalmol 125: 603-609. [Crossref]
- American Academy of Ophthalmology Cornea/External Disease Panel (2018) Preferred Practice Pattern® Guidelines. Bacterial Keratitis. San Francisco, CA: American Academy of Ophthalmology.
- Harrison SM (1975) Grading corneal ulcers. Ann Ophthalmol 7: 537-539. [Crossref]
- Jones DB (1981) Decision-making in the management of microbial keratitis. Ophthalmology 88: 814-820. [Crossref]
- Sweeney DF, Jalbert I, Covey M, Sankaridurg PR, Vajdic C et al. (2003) Clinical characterization of corneal infiltrative events observed with soft contact lens wear. Cornea 22: 435-442. [Crossref]
- Mah Sadorra JH, Yavuz SG, Najjar DM, Laibson PR, Rapuano CJ et al. (2005) Trends in contact lens-related corneal ulcers. Cornea 24: 51-58. [Crossref]
- Vital MC, Belloso M, Prager TC, Lanier JD (2007) Classifying the Severity of Corneal Ulcers by Using the ‘‘1, 2, 3’’ Rule. Cornea 26: 16-20. [Crossref]
- Austin A, Lietman T, Rose Nussbaumer J (2017) Update of the Management of Infectious Keratitis. Ophthalmology 127: 1678-1689. [Crossref]
- Lee YS, Tan HY, Yeh LK, Lin HC, Ma DH et al. (2014) Pediatric microbial keratitis in Taiwan: clinical and microbiological profiles, 1998-2002 versus 2008-2012. Am J Ophthalmol 157: 1090-1096. [Crossref]
- Young AL, Leung KS, Tsim N, Hui M, Jhanji V (2013) Risk factors, microbiological profile, and treatment outcomes of pediatric microbial keratitis in a tertiary care hospital in Hong Kong. Am J Ophthalmol 156: 1040-1044. [Crossref]
- Park J, Lee KM, Zhou H, Rabin M, Jwo K et al. (2015) Community practice patterns for bacterial corneal ulcer evaluation and treatment. Eye Contact Lens 41: 12-18. [Crossref]
- Al Otaibi AG (2012) Non-viral microbial keratitis in children. Saudi J Ophthalmol 26: 191-197. [Crossref]
- Weiner G (2012) Confronting Corneal Ulcers. Eyenet 44-42.
