Technique for Spleen Resection Using Radiofrequency Ablation
A B S T R A C T
From modern positions, removal of the spleen is undesirable due to the risk of post-splenectomy syndrome development, where first of all, the clinical manifestations of the syndrome of suppressive post-splenectomy infections are taken into account, which justifies either performing organ-preserving surgical interventions on the spleen, or heterotopic autotransplantation of spleen tissue. Spleen resection is accompanied by significant blood loss, both during the intervention and in the postoperative period. The various methods of prevention of bleeding (clips, coagulation instruments, local hemostatics, etc.) are not always effective. Currently, there have been reports of the possibility of using radiofrequency ablation (RFA) in interventions on the spleen. The literature data and own 10 observations of spleen resection with the use of RFA in patients with focal organ lesions are presented. All described in literature technologies use the Rita® device for RF destruction and the corresponding attachment, which consists of 4 needle active electrodes. The spleen parenchyma is "burned" blindly, and the organ parenchyma is divided in the middle, between the treated injections. A new technique for performing spleen resection using radiofrequency ablation has been proposed at A.V. Vishnevsky National Medical Research Center of Surgery. The essence of our proposed method for minimizing blood loss during spleen resection is as follows: RFA-exposure is carried out along the zone of organ ischaemia under the control of ultrasound. For RFA, we used a Radionics Cool-Tip® Ablation System and a set of water-cooled electrodes (MEDTRONIC, USA). The features of the surgical intervention are noted. No bleeding was noted either intraoperatively or in the postoperative period in any case. A comparative analysis of the results of various methods of organ-saving surgical treatment is presented.
Keywords
Splenic local lesions, partial splenectomy, radiofrequency ablation, RFA, RFA-resection
Introduction
Until recently, tumor and cystic lesions of the spleen were considered relatively rare diseases, which were diagnosed only with large sizes or the development of complications. The widespread use of non-invasive research methods for screening and diagnostics today makes it possible to identify focal lesions of the spleen in the early stages of their development, which creates the impression of an increase in morbidity. As a rule, a small individual experience in the treatment of spleen tumors and cysts makes it difficult to choose a timely and adequate treatment method or the optimal amount of surgery. This is reflected in the contradictions existing on the pages of the press regarding the choice of the method of treatment, the principles of examination, the details of the operative technique, the criteria for monitoring the state of patients in the postoperative period.
For a long time, splenectomy had no alternative in spleen surgery; it is still dogma for a number of medical schools. The traditionally used midline laparotomy for access to the organ does not always allow for precise identification of the anatomical gates of the spleen (because of the size, fragility, as well as the adhesions in the left upper quadrant of the abdomen), which requires the surgeon to know and other ways to reach the organ (transverse, bicuspid, thoracophrenal, etc.). Intraoperative ultrasound can provide significant help in determining the course of the main vessels, especially with pronounced adhesions, the phenomena of pancreatitis. From modern positions, removal of an organ is undesirable due to the risk of post-splenectomy syndrome development, where first of all, the clinical manifestations of the syndrome of suppressive post-splenectomy infections are taken into account, which justifies either performing organ-preserving surgical interventions on the spleen or heterotopic autotransplantation of spleen tissue [1]. It should be admitted that in general practice, organ-saving spleen surgeries are not yet popular, not only for traumatic injuries but also for benign tumors and cysts of the spleen. Behind this lies the neglect of a wide range of vital functions of the spleen and the possible consequences of its loss.
Spleen resection is accompanied by significant blood loss, both during the intervention and in the postoperative period. The various methods of prevention of bleeding (clips, coagulation instruments, local hemostatics, etc.) are not always effective [1]. Since the beginning of the 90s of the last century, the use of minimally invasive technology – radiofrequency ablation (RFA) of liver tumors – has begun around the world. Initially used as a cytoreductive technique in inoperable patients, it gradually occupied a broader niche in surgical hepatology, including aspects of section processing during liver resection, as well as alternatives to the classical surgical treatment of operable patients with focal liver neoplasms with a high degree of operational risk. The heat source in this system is not the electrode, but the tissue, which is heated by the electric current passing through it. Compared to a thermal applicator, which itself emits thermal energy, the RF electrode provides greater depth and uniformity of the coagulation effect.
Currently, there have been reports of the possibility of using RFA in interventions on the spleen. All described technologies use the Rita® device for RF destruction and the corresponding attachment, which consists of 4 needle active electrodes. The spleen parenchyma is ‘burned’ blindly, and the organ parenchyma is divided in the middle, between the treated injections. The author of the method of applying the attachment to the Rita® N.A. Habib widely promotes its technique, both for liver resection and for spleen interventions, and this technique has followers [2, 3]. There is a publication of Italian authors on the possibility of using RFA for the purpose of hemostasis in trauma to the spleen, but it is indicated that this technique cannot be used in trauma of large vessels or the area of the gates of the organ [4]. The experience of British surgeons using the Rita® device in 7 patients with spleen cysts with a good hemostatic effect and satisfactory immediate results has been published [5]. The possibility of laparoscopic resection of the spleen using the Rita® apparatus for a nonparasitic cyst has been described [6].
There are known works devoted to the use of RFA for hypersplenism by traditional access, percutaneously or laparoscopically, as an alternative to performing splenectomy. The most common complication of such interventions is intra-abdominal bleeding [7-9]. In the Russian-language medical literature, such publications are still rare, and they all relate to the use of the Rita® apparatus and a nozzle with 4 needles-electrodes of the Habib type [10, 11]. Experts from A.N. Ryzhikh National Medical Research Center of Coloproctology (Moscow, Russia) described an attempt to resect the lower pole of the spleen for solitary metastasis of colorectal cancer using a Habib-Sealer 4x radiofrequency electrode. The spleen tissue was dissected with a scalpel along the treated plane. In the area of spleen resection near the hilum during transsection, a branch of the splenic artery was transected, which was the reason for performing splenectomy. The mistake was that the vessels of the spleen hilum area were not controlled [11].
A few months later, the same specialists, using the Habib-sealer technique, described resection of the lower pole of the spleen for a lesion with a diameter of 10 mm, which was interpreted at the preoperative stage as metastasis of colorectal cancer; according to morphological studies, it turned out to be a hemangioma [10]. The practice of using this technique has revealed the following drawback. In view of the uncontrolled depth of ablation along the entire length of the separation area, there is a risk of bleeding when the parenchyma is dissected with an instrument, which creates the risk of an uncontrolled intraoperative situation. In this situation, it becomes more justified to treat the complication as a trauma to the spleen, which justifies removing the entire organ. Thus, the technical problem we are solving was to minimize the risk of both intraoperative and postoperative bleeding when performing RFA of the spleen during its resection, due to the presence of lesions of various etiology. The achieved results are minimization of the risk of intraoperative blood loss and the development of postoperative complications caused by bleeding.
Objective
To develop a method of bloodless resection of the spleen using RFA in patients with focal lesions.
Materials and Methods
At A.V. Vishnevsky National Medical Research Center of Surgery, an experience in treating more than 420 patients with focal lesions of the spleen has accumulated. Of these, in 139 cases, resection methods of surgical treatment were used. Various methods of organ-preserving operations on the spleen were: resection of the spleen with traditional access (n = 51), resection using RFA (n = 10), laparoscopic interventions (LS resections) (n = 66), robot-assisted operations (n = 12) [12-15]. The experience we have accumulated indicates that the use of minimally invasive surgical technologies is not always possible; therefore, it is necessary to develop approaches to minimize blood loss during open interventions. The essence of our proposed method for minimizing blood loss during spleen resection involves RFA-exposure along the zone of organ ischaemia under the control of ultrasound. The method is carried out as follows:
At the preoperative stage, a complete examination is carried out, including ultrasound with duplex scanning, MSCT and/or MRI. With CT reconstruction, the volume of the forthcoming intervention, the optimal surgical access, the diligence of the organs, the passage of the main vessels is determined. After laparotomy and access to the area of the anatomical hilum of the spleen under ultrasound navigation, the course of the splenic artery and vein, the level of division of the main vessels into branches of the first order, the peculiarities of the passage of the short arteries of the stomach to the hilum and the upper pole of the spleen, the features of the blood supply to the lower pole of the spleen gastro-splenic artery). With the help of ultrasound, the localization of the pancreatic tail is determined, which is extremely important for severe adhesions, splenomegaly, and symptoms of chronic pancreatitis. The splenic artery and vein are taken on a soft (tape) tourniquet, which is not pinched but is a guarantee of control over the development of possible bleeding. According to intraoperative ultrasound, the localization of the pathological lesion in the spleen and the vessels feeding it is specified. The vessels feeding the lesion are gradually isolated, ligated or clipped, and intersected.
In the event of bleeding, the turnstile is clamped on the main vessels. The operation continues on the ‘dry’ organ. A clear demarcation zone is defined. Next, the apparatus is used to perform RFA. For RFA, we used a Radionics Cool-Tip® Ablation System and a set of water-cooled electrodes (MEDTRONIC, USA). The apparatus consists of a generator (power 200 W, oscillation frequency 480 kHz), a peristaltic pump supplying a cooling liquid (sterile distilled water) in a closed loop, and a waveguide electrode. To carry out the intervention, needle electrodes 15-25 cm long with a working part of 10-20 mm were used. The use of active electrodes with a longer working part is unjustified due to the creation of a larger zone of necrosis. After the termination of RFA-exposure under ultrasound control, an incision is made to a depth of no more than 5-8 mm so that the depth of the remaining necrosis zone is 3 to 5 mm (in the cut). The ablation time for each immersion of the electrode was 20-40 sec. During this period, a zone of coagulation necrosis in the form of a cylinder was formed around the electrode. The next immersion of the electrode is made 1.0-1.5 cm higher than the previous one.
Similarly, (RFA-exposure followed by dissection of the parenchyma), the entire plane of the forthcoming division of the parenchyma is processed. The colour difference at the border of the ischaemic zones after such exposure became much more pronounced. In this case, bleeding from the parenchyma is not noted. The resected fragment of the spleen is removed along with the pathological lesion. The turnstiles are removed from the main vessels. After 30 minutes, ultrasonic testing is performed. Visual control for hemostasis is performed. The presence and characteristics of the blood supply to the organ stump are determined. The depth of the non-critical eschar is calculated. The usual depth of the scab is 3-5 mm, which, even with partial lysis, remains sufficient to prevent postoperative bleeding. The state of the distal pancreas is monitored (blood flow, the presence and development of parenchymal edema), which determines the features of the prevention and treatment of postoperative pancreatitis. A control silicone drainage is brought to the intervention zone, removed through a counter-opening, which will be removed after 24-48 hours. The operating wound is sutured tightly in layers. Performing the listed techniques allows to eliminate the risk of bleeding from the parenchyma. Reducing the risk of blood loss is due to the clinically significant depth of necrosis revealed by our studies. So, if the depth of necrosis on the remaining part of the spleen is less than 3 mm, then both intraoperative bleeding and the postoperative period may develop. If the depth of necrosis on the preserved parenchyma of the organ is more than 5, then its discharge in the postoperative period is possible, which will also lead to acute bleeding. Moreover, the possibility of obtaining a zone of necrosis from 3 to 5 mm is achieved due to the implementation of exposure under ultrasound guidance.
Besides, the minimization of the risk of bleeding was also due to the technique of inserting an electrode into the parenchyma, namely, the injection of a single needle-active electrode was carried out not immediately to the entire depth, but superficially, with its further immersion perpendicular to the longitudinal organ. In the immediate postoperative period, ultrasound control is performed on the 1st, 3rd, 7th day. The area of intervention is monitored: features of blood flow and subcapsular accumulations in the spleen, the presence of free fluid, fluid accumulations in the abdominal and pleural cavities. Using this technique, 10 patients were operated at the A.V. Vishnevsky National Medical Research Center of Surgery. Patients characteristics are shown in (Table 1).
Table 1: Main characteristics of patients who received RFA-resection of the spleen.
|
№ |
Gender |
Age, years |
Morphology, localization |
Intervention |
|
1 |
F |
51 |
echinococcal cyst with suppuration in the upper and middle lobes (12x14 cm) |
hemisplenectomy |
|
2 |
F |
26 |
sclerosed capillary hemangioma of the upper pole and hilum (6.5 cm in diameter) |
hemisplenectomy |
|
3 |
M |
55 |
abscess of the lower lobe of the spleen (5 cm in diameter) |
resection of the lower pole of the spleen |
|
4 |
M |
46 |
abscesses of the middle lobe of the spleen (the affected area is 6 cm in diameter); chronic pancreatitis, pancreatic tail pseudocyst |
hemisplenectomy, opening + excision of the walls of the pancreatic pseudocyst + suturing of the stomach defect (the upper pole remained, measuring 3x3 cm) |
|
5 |
F |
54 |
echinococcal cyst of the middle lobe of the spleen with suppuration (6 cm in diameter) |
hemisplenectomy |
|
6 |
M |
54 |
echinococcal cysts of the spleen (13x12 cm), liver (4.5 cm), left kidney (1.0 cm) |
resection of the lower lobe of the spleen, echinococcectomy from the liver and kidney |
|
7 |
F |
53 |
hamartoma of the upper lobe (10 cm in diameter) |
anatomical resection of the upper spleen |
|
8 |
F |
33 |
hamartoma of the hilum area (8x9 cm) |
hemisplenectomy (the lower pole remained, measuring 4x3 cm) |
|
9 |
F |
21 |
lymphangiomas of the upper and middle lobes (10 cm, 3 cm and 1 cm in diameter) |
resection of the superior pole of the spleen |
|
10 |
F |
49 |
hemlymphangioma of the upper and middle lobe (6 cm in diameter) |
resection of the superior pole of the spleen |
Results
No bleeding was noted either intraoperatively or in the postoperative period in any case. Only 1 (10%) patient who underwent RFA-resection had clinical manifestations of acalculous cholecystitis in the immediate postoperative period.
All patients were under dispensary control for up to 5 years. In all cases, the scab along the edge of the resection was consistent. During long-term examination, including laboratory, instrumental examination methods, VAS, the SF-36 questionnaire system, no manifestations of post-splenectomy hyposplenism syndrome were noted. For comparison, the results of traditional resections of the spleen, RFA-resections, laparoscopic resections (LS resections), and robotic resections (RA resections) are presented. The main characteristics of patients who underwent resection surgery on the spleen for benign focal lesions are presented in (Table 2).
Table 2: The main characteristics of patients and methods of resection interventions.
|
Indicator |
Spleen resection with traditional approach (n = 51) |
RFA-resection of the spleen with traditional approach (n = 10) |
LS resection of the spleen (n = 66) |
RA spleen resection (n = 12) |
|
Age, years |
46,4+12,8 (16-74) |
36,2+16,4 (26-55) |
26,2+11,4 (18-56) |
29,6+12,4 (20-54) |
|
Male |
12 (23,5%) |
3 (30%) |
17 (25,8%) |
1 (8,3%) |
|
Female |
39 (76,5%) |
7 (70%) |
49 (74,2%) |
11 (91,7%) |
|
Morphological structure |
||||
|
true cyst |
12 (23,5%) |
- |
30 (45,4%) |
2 (16,7%) |
|
pseudocyst |
13 (25,5%) |
- |
20 (30,3%) |
5 (41,6%) |
|
echinococcal cyst |
12 (23,5%) |
3 (30%) |
2 (3,03%) |
- |
|
abscess |
4 (7,8%) |
2 (25%) |
- |
- |
|
lymphangioma |
6 (11,7%) |
1 (10%) |
13 (19,7%) |
5 (41,6%) |
|
hemangioma |
3 (5,7%) |
1 (10%) |
1 (1,5%) |
- |
|
hamartoma |
1 (1,9%) |
2 (20%) |
- |
- |
|
inflammatory pseudotumor |
1 (10%) |
- |
- |
- |
|
Localization |
||||
|
top pole |
20 (39,2%) |
1 (10%) |
26 (39,4%) |
7 (58,3%) |
|
bottom pole |
17 (33,3%) |
1 (10%) |
28 (42,4%) |
3 (25%) |
|
middle segments |
12 (23,5%) |
3 (30%) |
12 (18,2%) |
2 (16,3%) |
|
subtotal lesion |
2 (3,9%) |
5 (50%) |
- |
- |
|
Lesion diameter |
8,2+4,8 (4-25) |
9,4+ 3,6 (5-14) |
6,8+3,2 (2-12) |
8,46+2,4 (5-14) |
Traditional interventions, laparoscopic and robotic interventions were performed more often for fluid benign lesions (true and pseudocysts). In half of the observations, with experience, RFA-resections were performed either for tumor lesions or for complex abscesses of the spleen. Obviously, RFA-resections were more often used in cases of significant lesions (more than half of the organ or more, and even with subtotal lesions). Comparative results of spleen interventions are presented in (Table 3). With an approximately equal duration of the intervention, there is a statistically significant lower volume of blood loss and a lower number of postoperative complications. The duration of a bed-day with RFA resection is somewhat shorter than with traditional spleen interventions and corresponds to the same time as after robotic and laparoscopic interventions.
Table 3: The results of surgical treatment depending on the method of spleen resection.
|
Indicator |
Spleen resection with traditional approach (n = 51) |
RFA-resection of the spleen with traditional approach (n = 10) |
LS resection of the spleen (n = 66) |
RA spleen resection (n = 12) |
Significance level, p |
|
Duration of surgery, min. |
138+38,4 (110-220) |
130,2+42,0 (95-182) |
123,9+39,8 (60-180) |
120+57,8 (60-235) |
0,78 |
|
Blood loss, ml |
420,2+260 (60-1500) |
80+44,2 (40-200) |
215+268,8 (50-900) |
122,2+257 (10-800) |
0,035 |
|
Duration of the postoperative period, bed-days |
9,8+4,2 (8-24) |
7,8+4,8 (5-22) |
7,1+2,8 (2-13) |
7,8+4,8 (5-22) |
0,55 |
|
Clavien-Dindo complication rate: |
|||||
|
I-II |
6 (11,7%) |
1 (10%) |
0 |
0 |
- |
|
III-IV |
3 (5,8%) |
0 |
0 |
2 (16,7%) |
>0,3 |
|
Mortality |
0 |
0 |
0 |
0 |
|
Clinical Case
Female, 26-year-old, was admitted with complaints of pulling pains in the left hypochondrium, aggravated after physical exertion.
I Anamnesis
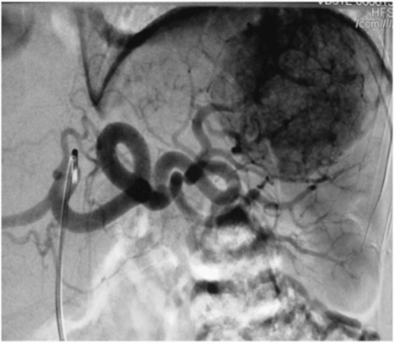
For the first time, the lesion of the spleen was detected in 2008. With dynamic observation, there is an increase in lesion size. In 2009, the patient was examined in another medical center, where, according to angiography (celiacography) (Figure 1), the presence of the lesion of the middle lobe and spleen hilum up to 6 cm was confirmed - hemangioma? Embolization of the vessels feeding the focus was performed at the same time. The patient abstained from the proposed splenectomy.
Figure 1: Celiacogram: hypervascular lesion of the middle lobe of the spleen.
II Ultrasound Examination
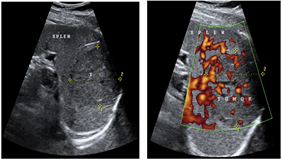
Figure 2 showed that the spleen was not enlarged, the area of the spleen was 38 cm2, the contours were smooth and clear. In the area of the upper pole of the spleen, a focal lesion of irregular rounded shape, with indistinct, relatively even contours, heterogeneous in structure, close in structure to the spleen parenchyma, slightly reduced echo density, measuring 65x54 mm, was determined. In the structure of the lesion, multiple vessels, both venous and arterial, were identified. The medial contour of the lesion was bent around the branches of the splenic artery and splenic veins. No enlarged lymph nodes were found. Conclusion: The hypervascular lesion of the spleen was more consistent with the hemangioma; there was an increase in the linear blood flow velocity in the splenic artery and an increase in the diameter of the splenic vein.
Figure 2: Hypervascular lesion of the spleen: ultrasound images in A) B-mode and in B) the Power Doppler mapping mode.
III Multispiral Computed Tomography
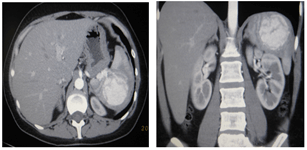
The abdominal organs (Figure 3) revealed a mass, measuring 6.4x6.7 cm, in the upper pole of the spleen. The lesion accumulated a contrast agent in the arterial phase of the study; in the venous and delayed phase, it became isodental. Conclusion: Spleen hemangioma.
Figure 3: Spleen hemangioma, MSC-tomograms in the arterial phase of the study, A) axial and B) frontal scans.
IV Magnetic Resonance Imaging
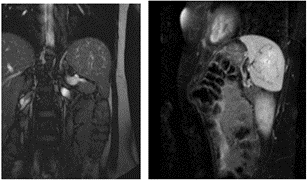
Figure 4 revealed that the spleen is of normal shape and location, measuring 11.5x5.5x8.4 cm. In its structure, at the level of the hilum, a rounded lesion with a diameter of up to 6.0 cm was determined with clear, bumpy contours. The structure of the lesion appeared to be heterogeneous due to the presence of many small vessels along the periphery, some of which spread to the center of the lesion, forming there an irregularly shaped zone. With contrast enhancement, this lesion accumulated contrast agent already in the arterial phase. Further, in the venous and delayed phases, the lesion was gradually freed from the contrast agent, remaining hyperintense to the organ parenchyma even 10 minutes after the administration of the contrast agent. Conclusion: Spleen hemangioma.
Figure 4: Spleen hemangioma: MRI scan in B-TFE mode in A) coronary projection and B) SPIR in sagittal projection.
V Surgery
Surgical intervention was performed in the following scope: hemisplenectomy (anatomical resection of the upper and middle lobes of the spleen). Laparotomy was performed from the L-shaped Kocher approach. It was noted that the spleen, measuring 16x10x6 cm, was in pronounced planar adhesions. By palpation and visualization, in the region of the upper pole with spread to the spleen hilum, an intraparenchymal tumor of a dense consistency, 6x6 cm in size, was determined. Further revision revealed the presence of significantly dilated veins in the region of the spleen hilum. The upper pole of the spleen was highlighted, with the transection of the short gastric arteries. Control ultrasound examination was performed. The splenic artery with a diameter of up to 2 cm in the area of the hilum was divided into three first-order arteries (three-lobe structure of the spleen). In the area of the hilum of the spleen, vessels were identified going to the upper pole and middle third, were pinched, sutured and crossed. The vessels feeding the lower lobe of the organ were preserved; for control, they were taken to the turnstile. In order to preserve the blood supply from the basin of the gastroepiploic artery, the lower pole was not isolated.
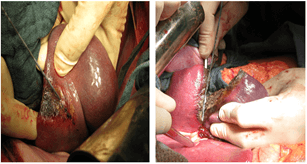
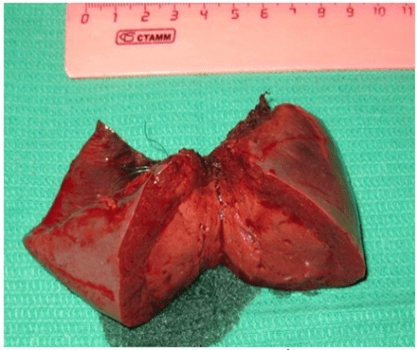
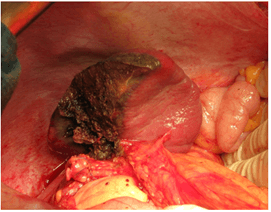
A radiofrequency action was performed along the emerging demarcation line under the control of a hand placed behind the spleen using a Cool-Tip apparatus (2 cm needle working part) (Figure 5A). The ablation time for each immersion of the electrode was 20-40 sec. During this period, a zone of coagulation necrosis was formed around the electrode in the form of a cylinder 1 cm in diameter and up to 2 cm in height. Then the parenchyma was dissected using a scalpel and scissors (Figure 5B). The next immersion of the electrode was made 1.5 cm higher than the previous one. The entire parenchyma dividing plane was processed in a similar way. The difference in colour between the lobes after such treatment became much more pronounced. As a result, together with the lesion, the upper and middle lobes of the spleen were removed (Figure 6). There was no bleeding from the parenchyma. The remaining lower lobe, 8x3x6 cm in size, was recognized as viable (Figure 7). The wound surface was not additionally processed. The operation ended with drainage of the left subphrenic space.
Figure 5: Stages of the operation: A) radio frequency exposure along the line of demarcation; B) dissection of the spleen parenchyma.
Figure 6: The removed part of the spleen with the lesion.
Figure 7: Preserved lower lobe of the spleen.
VI Morphological Examination
The microscopic picture was more consistent with sclerosed capillary hemangioma, a site of chronic inflammation with the presence of giant cells of the type of foreign bodies (a consequence of previously performed embolization) was also revealed. The postoperative period of the patient, given as a clinical example, was uneventful. The patient was discharged on the 7th day. At control examinations, moderate hypertrophy of the spleen stump was noted. After 3 years, a 5-mm avascular zone is preserved along the edge of the spleen resection. A year and a half after the operation, the woman gave birth to a healthy baby naturally. The use of RFA-resection, according to our data, is justified both with significant lesions of the spleen and with the localization of a pathological lesion in one of the poles of the spleen. Good results have been obtained both in the treatment of tumor lesions of significant volume and in spleen abscesses.
Conclusion
In our experience, the diagnosis of a benign focal lesion of the spleen was established based on the results of a comprehensive preoperative examination. The features of the blood supply to the spleen were identified. Therefore, the planned organ-saving interventions on the spleen were reasonable. Removing the spleen from our patients could significantly worsen their condition in the future. Often, there were changes in the spleen area due to earlier embolization and due to the inflammatory process inside or in the immediate vicinity of the spleen. The use of RFA made it possible to carry out the pesticide interventions practically bloodlessly. To carry out ablation of the spleen using any appropriate device, not only knowledge of the specific features of intra- and extraorganic blood flow is required, but also preliminary mobilization of the removed part of the spleen. It is preferable to ensure in advance control over the great vessels of the spleen in order to prevent possible bleeding. The use of control silicone drains when performing interventions on the spleen is considered mandatory. The presented method of performing bloodless resection of the spleen can be successfully applied in surgical practice.
Article Info
Article Type
Research ArticlePublication history
Received: Mon 01, Feb 2021Accepted: Mon 22, Feb 2021
Published: Wed 10, Mar 2021
Copyright
© 2023 Ionkin Dmitry. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2021.03.06
Author Info
Ionkin Dmitry Stepanova Yulia Aleksandrovna Alimurzaeva Maksalina Zakaryaevna Vishnevsky Vladimir Aleksandrovich
Corresponding Author
Ionkin DmitryLiver and Pancreas Surgery Department, A.V. Vishnevsky National Medical Research Center of Surgery, Russia
Figures & Tables
Table 1: Main characteristics of patients who received RFA-resection of the spleen.
|
№ |
Gender |
Age, years |
Morphology, localization |
Intervention |
|
1 |
F |
51 |
echinococcal cyst with suppuration in the upper and middle lobes (12x14 cm) |
hemisplenectomy |
|
2 |
F |
26 |
sclerosed capillary hemangioma of the upper pole and hilum (6.5 cm in diameter) |
hemisplenectomy |
|
3 |
M |
55 |
abscess of the lower lobe of the spleen (5 cm in diameter) |
resection of the lower pole of the spleen |
|
4 |
M |
46 |
abscesses of the middle lobe of the spleen (the affected area is 6 cm in diameter); chronic pancreatitis, pancreatic tail pseudocyst |
hemisplenectomy, opening + excision of the walls of the pancreatic pseudocyst + suturing of the stomach defect (the upper pole remained, measuring 3x3 cm) |
|
5 |
F |
54 |
echinococcal cyst of the middle lobe of the spleen with suppuration (6 cm in diameter) |
hemisplenectomy |
|
6 |
M |
54 |
echinococcal cysts of the spleen (13x12 cm), liver (4.5 cm), left kidney (1.0 cm) |
resection of the lower lobe of the spleen, echinococcectomy from the liver and kidney |
|
7 |
F |
53 |
hamartoma of the upper lobe (10 cm in diameter) |
anatomical resection of the upper spleen |
|
8 |
F |
33 |
hamartoma of the hilum area (8x9 cm) |
hemisplenectomy (the lower pole remained, measuring 4x3 cm) |
|
9 |
F |
21 |
lymphangiomas of the upper and middle lobes (10 cm, 3 cm and 1 cm in diameter) |
resection of the superior pole of the spleen |
|
10 |
F |
49 |
hemlymphangioma of the upper and middle lobe (6 cm in diameter) |
resection of the superior pole of the spleen |
Table 2: The main characteristics of patients and methods of resection interventions.
|
Indicator |
Spleen resection with traditional approach (n = 51) |
RFA-resection of the spleen with traditional approach (n = 10) |
LS resection of the spleen (n = 66) |
RA spleen resection (n = 12) |
|
Age, years |
46,4+12,8 (16-74) |
36,2+16,4 (26-55) |
26,2+11,4 (18-56) |
29,6+12,4 (20-54) |
|
Male |
12 (23,5%) |
3 (30%) |
17 (25,8%) |
1 (8,3%) |
|
Female |
39 (76,5%) |
7 (70%) |
49 (74,2%) |
11 (91,7%) |
|
Morphological structure |
||||
|
true cyst |
12 (23,5%) |
- |
30 (45,4%) |
2 (16,7%) |
|
pseudocyst |
13 (25,5%) |
- |
20 (30,3%) |
5 (41,6%) |
|
echinococcal cyst |
12 (23,5%) |
3 (30%) |
2 (3,03%) |
- |
|
abscess |
4 (7,8%) |
2 (25%) |
- |
- |
|
lymphangioma |
6 (11,7%) |
1 (10%) |
13 (19,7%) |
5 (41,6%) |
|
hemangioma |
3 (5,7%) |
1 (10%) |
1 (1,5%) |
- |
|
hamartoma |
1 (1,9%) |
2 (20%) |
- |
- |
|
inflammatory pseudotumor |
1 (10%) |
- |
- |
- |
|
Localization |
||||
|
top pole |
20 (39,2%) |
1 (10%) |
26 (39,4%) |
7 (58,3%) |
|
bottom pole |
17 (33,3%) |
1 (10%) |
28 (42,4%) |
3 (25%) |
|
middle segments |
12 (23,5%) |
3 (30%) |
12 (18,2%) |
2 (16,3%) |
|
subtotal lesion |
2 (3,9%) |
5 (50%) |
- |
- |
|
Lesion diameter |
8,2+4,8 (4-25) |
9,4+ 3,6 (5-14) |
6,8+3,2 (2-12) |
8,46+2,4 (5-14) |
Table 3: The results of surgical treatment depending on the method of spleen resection.
|
Indicator |
Spleen resection with traditional approach (n = 51) |
RFA-resection of the spleen with traditional approach (n = 10) |
LS resection of the spleen (n = 66) |
RA spleen resection (n = 12) |
Significance level, p |
|
Duration of surgery, min. |
138+38,4 (110-220) |
130,2+42,0 (95-182) |
123,9+39,8 (60-180) |
120+57,8 (60-235) |
0,78 |
|
Blood loss, ml |
420,2+260 (60-1500) |
80+44,2 (40-200) |
215+268,8 (50-900) |
122,2+257 (10-800) |
0,035 |
|
Duration of the postoperative period, bed-days |
9,8+4,2 (8-24) |
7,8+4,8 (5-22) |
7,1+2,8 (2-13) |
7,8+4,8 (5-22) |
0,55 |
|
Clavien-Dindo complication rate: |
|||||
|
I-II |
6 (11,7%) |
1 (10%) |
0 |
0 |
- |
|
III-IV |
3 (5,8%) |
0 |
0 |
2 (16,7%) |
>0,3 |
|
Mortality |
0 |
0 |
0 |
0 |
|
References
1. Kubyshkin VA, Ionkin DA (2007) Tumors and cysts of the spleen. Moscow: Medpraktika 287.
2. Habib NA, Spalding D, Navarra G, Nicholls J (2003) How we do a bloodless partial splenectomy. Am J Surg 186: 164-166. [Crossref]
3. Poos HP, Daryanani D, Klaase JM (2009) Partial Splenectomy for Splenic Cyst using a Bipolar Radiofrequency Device. Gastroenterology Res 2: 242-244. [Crossref]
4. Stella M, Percivale A, Pasqualini M, Pittaluga M, Gandolfo N et al. (2005) Conservative management of a spleen trauma using radiofrequency. Ann Ital Chir 76: 559-561. [Crossref]
5. Bong JJ, Kumar R, Spalding D (2011) A novel technique of partial splenectomy using radiofrequency ablation. J Gastrointest Surg 15: 371-372. [Crossref]
6. Gumbs AA, Bouhanna P, Bar Zakai B, Briennon X, Gayet B (2008) Laparoscopic partial splenectomy using radiofrequency ablation. J Laparoendosc Adv Surg Tech A 18: 611-613. [Crossref]
7. Boer TD, Habib S (2015) Big spleens and hypersplenism: fix it or foget
it? Liver Int 2: 1492-1498. [Crossref]
8. Feng K, Ma K, Liu Q, Wu Q, Dong J et al. (2011) Randomized clinical trial of splenic radiofrequency ablation versus splenectomy for severe hypersplenism. Br J Surg 98: 354-361. [Crossref]
9. Liu Q, Ma K, Song Y, Zhou N, He Z (2008) Two-year follow-up of splenic radiofrequency ablation in patients with cirrhotic hypersplenism: does increased hepatic arterial flow induce liver regeneration? Surgery 143: 509-518. [Crossref]
10. Ponomarenko AA, Rybakov EG, Khomyakov EA, Mainovskaya OA, Trubacheva Yu L (2014) Technical aspects of spleen resection using the Habib-sealer 4-x radio frequency electrode (clinical observation). Coloproctology 4: 62-67.
11. Rybakov EG, Ponomarenko AA, Khomyakov EA, Mainovskaya OA (2014) Metastatic lesion of the spleen in colorectal cancer. Surgery. Khirurgiia (Mosk) 9: 92-95. [Crossref]
12. Ionkin DA, Ikramov RZ, Andreenkov SS, Stepanova Yu A, Zhavoronkova OI et al. (2011) Spleen resection using radiofrequency ablation. Ann Surg Hepatol 4: 108-113.
13. Ionkin DA, Stepanova Yu A, Ikramov RZ, Alimurzaeva MZ (2018) Method of bloodless resection of the spleen. Patent for invention No. 2675355.
14. Kubyshkin VA, Tsvirkun VV, Ionkin DA (1996) Laparoscopic treatment of nonparasitic spleen cysts. Endosc Surg 3: 18-20.
15. Berelavichus SV, Smirnov AV, Ionkin DA, Kriger AG, Dugarova RS (2015) Robot-assisted and laparoscopic spleen resections for nonparasitic cysts. Khirurgiia (Mosk) 7: 41-48. [Crossref]
