Surgical Treatment of Tensile Effusion in Postoperative Cavity after Glioma Resection
A B S T R A C T
Objectives: To explore the clinical surgical methods for the treatment of tensile effusion in postoperative cavity after glioma resection.
Methods: Clinical data of 26 cases of tensile effusion in postoperative cavity after glioma resection were analyzed retrospectively. All 26 patients underwent surgical treatment, including 16 cases who underwent puncture and drainage (P&D), and 10 cases who underwent craniotomy decompression (CD). Among the cases of P&D, postoperative cavity in 7 cases were punctured through the burr hole for creating the skull flap for intraoperative resection of glioma, and postoperative cavities in 9 cases were punctured through a burr hole for the dura mater suspended under the skull flap. Glasgow Coma Scale (GCS) was used to evaluate the consciousness status of the two groups of patients before and after surgery. Moreover, time of operation, intraoperative blood loss, postoperative hospital duration, and postoperative complications were also adopted for evaluation of the merits of the two methods for treatment of tensile effusion in postoperative cavity after glioma resection.
Results: The consciousness of the patients in both groups improved within 24 hours after the operation, and there were no death cases. In the P&D group, the GCS score within 24 hours after surgery was (14.38±0.20), the time of operation time was (7.81±0.64) mins, intraoperative blood loss was (6.25±0.56) ml, and postoperative hospital duration was (6.69±0.54) days. There were 3 cases with postoperative complications (1 case of puncture tract hemorrhage, 1 case of subdural hematoma and 1 case of intracranial infection) in the P&D group. All of the puncture tract hemorrhage and subdural hematoma did not require surgical treatment, and the intracranial infection was cured after antibiotic treatment and lumbar puncture for cerebrospinal fluid replacement. In the CD group, the GCS score within 24 hours after surgery was (13.70±0.21), the time of operation was (124.10±8.96) mins, intraoperative blood loss was (260±30.55) ml, and postoperative hospital duration was (11.30±0.60) days. There were 2 cases with postoperative complications (both of them were with subdural hematoma), none of which required surgical treatment. The GCS score, time of operation, intraoperative blood loss and postoperative hospital duration in the P&D group were better than those in the CD group (P < 0.05). There was no significant difference in the incidence of postoperative complications between the two groups (P >0.05).
Conclusion: The P&D and CD for tensile effusion in postoperative cavity after glioma resection can both achieve ideal results, but P&D in line with the concept of micro-invasive neurosurgery was better than CD in terms of postoperative GCS score within 24 hours, duration of operative, intraoperative blood loss, and postoperative hospital duration.
Keywords
Glioma, tensile effusion, postoperative cavity, puncture and drainage (P&D), craniotomy decompression (CD)
Introduction
Glioma is the most common primary tumor of the adult central nervous system, accounting for 35%-60% of intracranial tumors. The incidence of glioma reaches 80,000-100,000/year and is still on the rise in recent years [1]. The invasive growth pattern, abundant angiogenesis and high invasiveness of glioma determine its malignant biological behavior and poor prognosis [2]. Current treatments for glioma include surgical resection (maximum safe resection of the tumor), postoperative radiotherapy with concomitant and adjuvant temozolomide chemotherapy [3]. Although the total resection rate of the glioma can be improved, and the space-occupying effect can be relieved better by the maximum safe resection of the glioma, the larger postoperative cavity will lead to the existence of effusion if it is not closed as soon as possible. In some cases, the effusion inside the cavity increases progressively, forming tensile effusion, which will form new space-occupying effect, pressing the brain tissue around the cavity, and causing the deterioration of central nervous function. Compared to other postoperative implications, such as intracranial bleeding, cerebral infarction, cerebral edema, etc., tensile effusion in postoperative cavity is relatively rare [4]. Once it occurs, it can not only cause headache and coma in patients, but also even death in patients by forming a cerebral hernia [5]. In view of the above situation, this study retrospectively analyzed the clinical data of 26 patients with tensile effusion in postoperative cavity in 487 patients underwent craniotomy resection of glioma in our hospital from March 2015 to December 2019, and the report is as follows.
Materials and Methods
I Clinical Materials
In this study, patients with tensile effusion in postoperative cavity after the resection of glioma were enrolled in the department of neurosurgery, the Xinqiao Hospital Affiliated with Third Military Medical University from March 2015 to December 2019. The inclusion criteria were: (1) Imaging data on admission and pathological diagnosis were obtained after craniotomy resection of patients were confirmed as supratentorial glioma; (2) All patients had received craniotomy for gross total or subtotal resection for glioma, and woke up within 2 hours postoperation; (3) All patients underwent enhanced MRI or CT and the Glasgow Coma Scale (GCS) ?13 within 24 hours after craniotomy; (4) The central nervous function of patients deteriorated within 2 weeks after glioma resection, and the GCS score was reduced to 9-12. The CT scan or MRI on the day of the disturbance of consciousness aggravation showed the dilation of postoperative cavity which was filled with effusion, and the midline structure deviation was more serious compared with the previous 24 hours after glioma resection; (5) No improvement was found after treatment with mannitol and dexamethasone. The exclusion criteria were: (1) Patients with obvious surgical contraindications; (2) Patients who already had indications of terminal brain herniation; (3) Patients who had glioma recurrence and underwent craniotomy resection again.
Table 1:Clinical data of 26 cases of tensile effusion in postoperative cavity after glioma resection.
|
|
|
Total |
P&D |
CD |
P value |
|
|
|
26 |
16 |
10 |
|
|
Sex |
Male |
14 |
8 |
6 |
0.926 |
|
Female |
12 |
8 |
4 |
||
|
Male/Female |
1.17 |
1 |
1.5 |
|
|
|
Age |
Range |
13-78 |
13-78 |
15-64 |
|
|
Mean age |
52.54±15.22 |
54.50±15.53 |
49.40±14.96 |
0.417 |
|
|
>50/≤50 |
15/11 |
10/6 |
5/5 |
0.826 |
|
|
Pathological diagnosis |
OD |
4 |
1 |
3 |
0.374 |
|
DA |
2 |
2 |
0 |
||
|
AA |
5 |
4 |
1 |
||
|
GBM |
15 |
9 |
6 |
||
|
Cavity site |
Frontal lobe |
11 |
6 |
5 |
0.305 |
|
Temporal lobe |
17 |
10 |
7 |
||
|
Parietal lobe |
7 |
3 |
4 |
||
|
Occipital lobe |
4 |
4 |
0 |
||
|
WHO grade |
II |
6 |
3 |
3 |
0.585 |
|
III |
5 |
4 |
1 |
||
|
IV |
15 |
9 |
6 |
||
|
GCS Score |
Preoperative |
— |
11.00±0.26 |
10.20±0.27 |
0.3146 |
|
Postoperative |
— |
14.38±0.20 |
13.70±0.21 |
0.0373 |
|
|
Time of operation (min) |
— |
7.81±0.64 |
124.10±8.96 |
<0.001 |
|
|
Intraoperative blood loss (min) |
— |
6.25±0.56 |
260.00±30.55 |
<0.001 |
|
|
Postoperative hospital duration (d) |
|
6.69±0.54 |
11.30±0.60 |
<0.001 |
|
|
Postoperative complications |
5 |
3 |
2 |
0.657 |
|
OD: oligodendrocytoma; GBM: glioblastoma; DA: diffuse astrocytoma; AA: anaplastic astrocytoma; P&D: puncture and drainage; CD: craniotomy depression; GCS: Glasgow Coma Scale.
Through screening, a total of 26 patients were included. In 26 patients, 16 cases underwent cavity puncture and drainage (P&D), and 10 cases underwent craniotomy decompression (CD). Among the cases for P&D, 7 cases were punctured through the burr hole for creating the skull flap for intraoperative resection of glioma; other 9 cases were punctured through a burr hole for the dura mater suspending under the skull flap. There were no statistically significant differences between the two groups in gender, age, pathological features of glioma, WHO classification, residual cavity location, and preoperative GCS score (P >, 0.05) (Table 1).
II Surgical Method
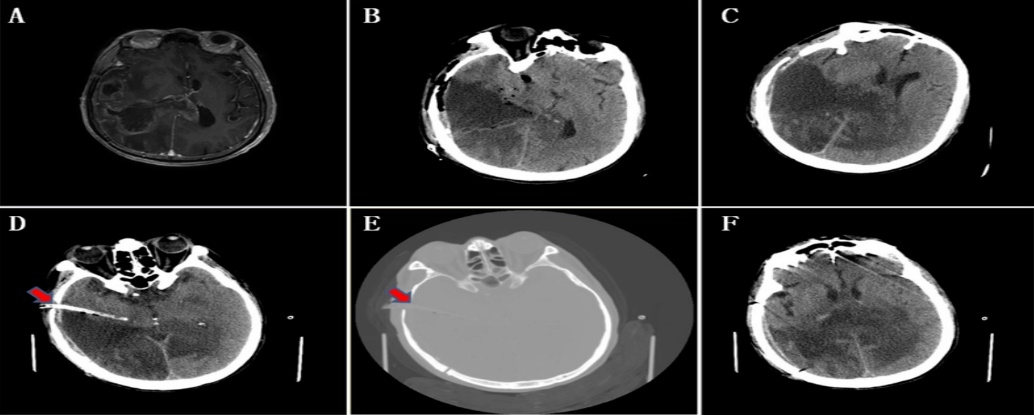
Both groups were performed by the same surgical team. All patients in the P&D group were operated under local anesthesia in the ward. The head was tilted to the contralateral side to expose the original surgical flap. The length of the drainage channel and the path drainage tube placed in were determined by the CT interpretation. If there was a bone hole which was made for milling a bone flap for the resection of glioma during the earlier craniotomy, where the projection of cavity was on, the drainage tube would cross the burr hole to puncture the cavity (Figure 1). If the burr hole was not on where the projection of the cavity was on, we could puncture through a smaller hole for the dura mater suspending under the skull flap (Figure 2). In (Figure 1), if the drainage tube was placed through the burr hole, the burr hole under the scalp should be palpated and marked.
Figure 1: Puncture and drainage through the burr hole for creating the skull flap for intraoperative resection of glioma. A) MRI showed irregular long T1 abnormal signal in the right temporoparietal occipital and corpus callosum region, with a range of about 8.2cmx5.6cm and uneven enhancement. Edema band around the glioma, compression of the right ventricle, and displacement of the midline structure to the left was observed obviously. B) Yellow fluorescence guided resection was performed. On the 1st day of glioma resection, CT showed subtotal glioma resection, GCS was E3V5M6, and muscle strength of the left limb was level I. C) On the 10th day after the resection of glioma, CT showed the effusion in cavity was obvious, the midline structure was skewed to the left, the GCS was down to E2V5M6, and the muscle strength of the left limb was grade 0. D) Puncture and drainage through temporal bone hole. On the 11th day after tumor resection, CT showed that the cavity shrank significantly. GCS was back to E3V5M6, left limb strength was back to level I. E) The bone window at the puncture site (the red arrow is the bone hole left for the bone flap milling). F) The center line structure deviation has been recovery.
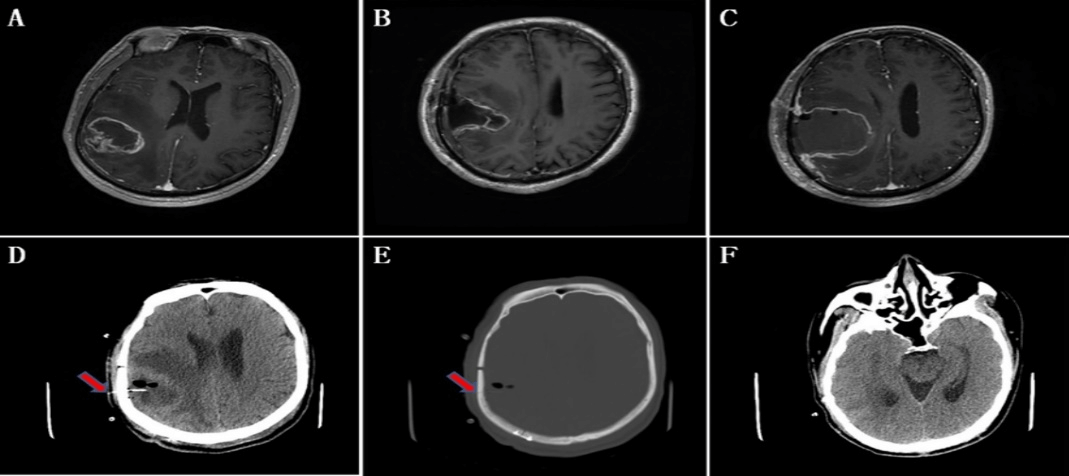
Figure 2: Puncture and drainage through the hole on the skull flap for the dura mater suspending. A) MRI showed mixed long T1 signal with obvious ring enhancement. The range is about 4.3cmx3.9cm. B) Intraoperative yellow fluorescence guided resection for glioma was performed. On the 1st day after glioma resection, MRI showed GTR of the tumor, leaving a triangular tumor cavity with the base facing to the skull, and annular enhancement on the cavity wall. GCS was E4V5M6, left limb strength level IV. C) On the 4th day after the resection of glioma, the disturbance of consciousness was aggravated, GCS was down to E2V5M6, and the muscle strength of the left limb was grade II. MRI revealed a dilated cavity filled with effusion and which lead to the midline structure left sided. D) and F) After the drainage of effusion by the intravenous indwelling needle through dura suspending hole on the skull scalp. CT on the 5th day after tumor resection showed that the cavity was narrowed, midline structure back to middle position, and the compression of brain ambient cisterns was released. GCS was back to E4V5M6, left limb strength was back to level IV. E) CT at the puncture site (the red arrow was the dura suspension hole on the skull flap).
If the burr hole was not easily felt by hand because it was covered by the temporal muscle or swollen scalp, the CT scan of the skull should be used to assist localization. In (Figure 2), if the catheter was placed through the smaller hole on the bone flap for dura suspension, CT assisted localization of the puncture site and marking it on the scalp was essential. After the marking of puncture site on the scalp, the puncture area was routinely sterilized, local infiltration anesthesia with lidocaine 2% with epinephrine (1:100,000) as a vasoconstrictor to minimize bleeding at the site of the incision was made, and the scalp incision about 1cm long was made with a sharp knife. In the cases of burr hole puncture, the scalp was separated to expose the dura, then the dura was cut open, and a drainage tube was placed into postoperative cavity of glioma. If puncture was performed through the smaller hole for dura suspension on the bone flap, the scalp was separated to expose the bone hole, and a 29G intravenous indwelling needle was inserted in. If the puncture is successful in P&D, we can see the effusion gushing out.
Ventricular drainage tube or intravenous indwelling needle connected with antisiphon ventricular drainage bottle and drainage bag. The antisiphon drainage bottle was suspended 10 cm above the bilateral external auditory canal, and postoperative cavity was rinsed through the drainage tube with 10 ml sterile normal saline each time and 3 times a day. CT scan was performed on the day after puncture. The drainage time was determined according to the recovery of the patient's consciousness disorder and the CT examination. Usually, the tensile effusion was evacuated with the sign of drainage fluid gradually cleared up, the CT scan showed that postoperative cavity shrank significantly, and the midline structure shifted to the center line within 3-5 days.
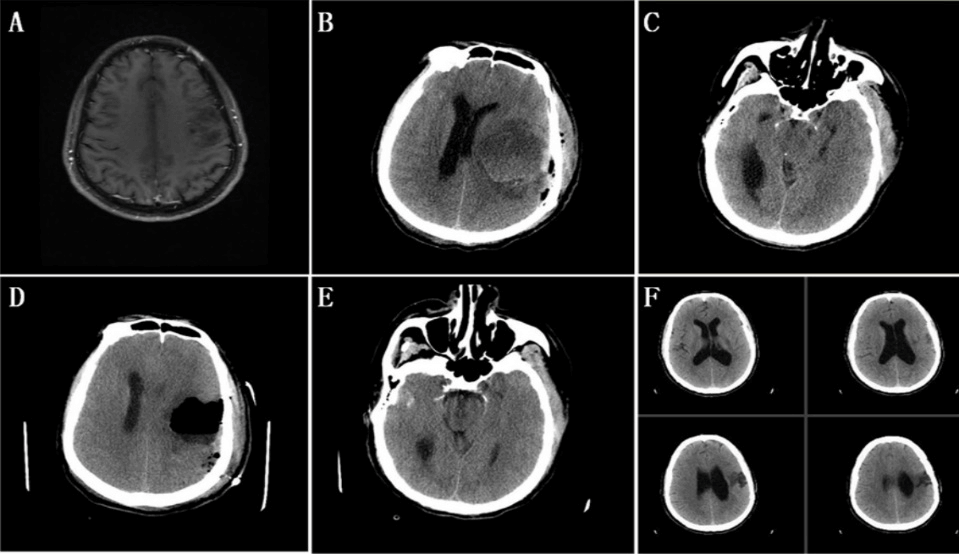
All patients in the CD group were operated under general anesthesia in the operating room. Routine disinfection was performed after exposing the original craniotomy flap. The flap was opened, and the bone was removed after the removal of the bone flap fixation material. Exploration revealed that dura tension was higher. When the dura was opened, we could see that cortical incision for early glioma resection often adhered to the dura or arachnoid. Reopened the adhesive cortical incision, cleaned the effusion and other foreign material (such as gelfoam or hemostatic yarn attached to the wall of the cavity) carefully in postoperative cavity, and to explore whether the bottom of the cavity communicated with the lateral ventricle and if so, the channel was closed by pressure with thin sheet gelfoam. After did that, postoperative cavity was rinsed with sterile normal saline until the residual fluid was clear and the brain tissue collapsed after the fluid was sucked out of postoperative cavity, then the cranium was closed routinely (Figure 3).
Figure 3: Craniotomy and depression for tensile effusion. A) MRI showed an abnormal signal in the left frontotemporal lobe, with a range of about 3.8cmx3.4cm, unclear boundary and long T1 signal without obvious enhancement. Following glioma resection under the intraoperative guidance of awake anaesthetic and yellow fluorescence, GCS was E4V4M6 and with partial motor aphasia. B) and C) On the 6th day after the operation of tumors, CT showed the cavity was filled with effusion was obvious, which lead to obvious deviation of the midline structure and compression of the brain ambient cisterns. GCS was down to E2V2M5. D) and E) Craniotomy and decompression under general anaesthesia was performed. On the 7th day after the glioma resection, the cavity was filled with effusion was significantly reduced on CT scan, the deviation of midline structure was recovered, and the depression of brain ambient cisterns was released. Consciousness was back to E3V5M6 in GCS. F) On the 13th day after the resection of glioma, the cavity on the CT scan was almost closed, without the midline shift.
III Intraoperative and Postoperative Evaluation
The intraoperative evaluation was conducted with 2 aspects for comparative statistical analysis: time of operation, blood loss volume during operation. Postoperative evaluation was reflected in 3 aspects: the GCS score within 24 hours after surgery, the postoperative hospital duration, and the incidence of postoperative complications.
IV Statistical Methods
SPSS 13.0 was used for statistical analysis, measurement data that were normally distributed are denoted by mean±standard deviation and were compared with the t-test. Enumeration data were tested by the Chi-Square test. P<0.05 was considered statistically significant.
Results
There are 26 cases of tensile effusion in postoperative cavity. The incidence of it after 487 glioma craniotomy resections was 5.34%. Patients with tensile effusion in postoperative cavity underwent P&D or CD, and all showed significant improvement in consciousness within 24 hours after operation, and the GCS score rose to 13-15. The GCS score was higher in the P&D group (14.38±0.20 vs. 13.70±0.21, P < 0.05). The time of operation of the P&D group was (7.81±0.64) min, the blood loss volume during operation was (6.25±0.56) ml, and the postoperative hospital duration was (6.69±0.54) d. The time of operation of the CD group was (124.10±8.96) mins, blood loss volume during the operation was (260.00±30.55) ml, and postoperative hospital duration was (11.30±0.60) days. There were significant differences between the two groups in time of operation, blood loss volume during operation, and postoperative hospital duration (P < 0.001). In terms of complications, 1 case of puncture tract hemorrhage, 1 case of subdural hematoma and 1 case of intracranial infection occurred in the P&D group. Neither puncture tract hemorrhage nor subdural hematoma was treated surgically. Intracranial infection of 1 case was cured by antibiotic treatment and cerebrospinal fluid replacement through a lumbar puncture. Subdural hematoma occurred in the CD group in 2 cases, none of which was treated surgically. There was no significant difference in the incidence of postoperative complications between the two groups (P >0.05)..
Discussion
I The Causes of Tensile Effusion in the Postoperative Cavity after Resection of Glioma
Glioma, as the central nervous system tumor with the highest incidence, brings great harm to human health. Effusion after craniotomy resection in glioma usually occurs in subdural and subcutaneous space, and tensile effusion in postoperative cavity are rarely reported. Yu et al. and Luo et al. reported two cases, respectively [6, 7]. From March 2015 to December 2019, our department performed a total of 487 cases of intraoperative fluorescently guided resection for the initial diagnosis of glioma, tensile effusion in postoperative cavity occurred in 26 cases, with an incidence of 5.34%.
Combined with clinical observations and literature review, the causes of tensile effusion in postoperative cavity after resection of glioma were summarized as follows:
i. The formation of the ampulla-shaped cavity and the unidirectional flap - 'Maximum safe resection' and 'minimally invasive' as the widely accepted concept in recent years in the field of surgical treatment of glioma [8]. With the application of multiple assistive technologies (virtual reality technology, intraoperative navigation, intraoperative MRI, intraoperative fluorescence guide resection, conscious sedation for glioma resection), the rates of gross total resection (GTR) have been greatly increased, and the protection of brain parenchyma around glioma tissue is also getting better and better [9-13]. However, with the above progress, especially the continuous enlargement of the extent of glioma resection, the volume of residual cavity after resection of the glioma increases continuously. At the same time, with the widespread acceptance of the concept of brain parenchyma protection and micro invasion, the cerebral cortex incision became smaller and smaller. Especially, after the combination of neuronavigation and intraoperative yellow fluorescence guidance for the resection of glioma, the two phenomena of the increased volume of postoperative cavity and decreased the width of cortical incision were more prominent, which easily resulted in the formation of an ampulla-shaped cavity after glioma resection. In addition, the postoperative edema in the cerebral tissue and the gravity of the brain itself lead to the adhesion between the cortical ostomy and arachnoid or dura, which can form a unidirectional flap-like structure. Once the unidirectional flap was formed, cerebrospinal fluid (CSF) in the subarachnoid space can enter postoperative cavity, but with the change of body position or the increase of intracranial pressure such as cough, sneeze and deep breath, the flap will be closed, and CSF in the cavity cannot flow out, thus forming tensile effusion [5]. Such effusions are usually CSF in nature, and the CT scan shows that the density of the effusion is like that of CSF.
ii. The destruction of the blood-brain barrier (BBB) and the formation of new blood vessels on the wall of postoperative cavity - Glioma can damage the integrity and increase the permeability of the BBB by a variety of ways in the process of its development, such as, it can reduce the expression of the microcapsule protein caveolin-1/2 and upregulated the expression of tight junction proteins occludin and ZO-1 to disrupt the tight connections between the endothelial cells on the BBB [14-16]. Glioma also can improve the permeability of BBB by enhancing the expression of water channel protein AQP-4 [17]. In addition, the mechanical damage to the cavity wall during glioma resection can also destroy the local BBB and small blood vessels. The destruction and the increase of permeability of the BBB, and the neovascularization on the cavity wall caused by biochemical and mechanical injury factors will constantly increase the exudation of the cavity wall and resulted in the tensile effusion in postoperative cavity in glioma. The resulting effusion, in this case, was often mixed with blood components. So, on the CT scan, the density of effusion was higher than that of CSF.
iii. The high fluid osmotic pressure of the effusion the in cavity - In the conventional resection of glioma, normal saline was often used for flushing the residual cavity until the rinse fluid was clear after the glioma had been removed to ensure complete hemostasis and prevented massive postoperative bleeding in the surgical area, and after that, the cranial closure was performed. Although the fluid osmotic pressure of effusion in cavity was not high after normal saline irrigation, but with the disintegration of artificial absorbable materials such as gelfoam or hemostatic gauze, aseptic inflammatory reaction would be caused, which would increase the permeability of blood vessels on cavity wall and make a large number of proteins and inflammatory cells exude from the vessel to increase the osmotic pressure of fluid in cavity. In addition, Lee et al. believed that not only artificial absorbable materials but also the tiny blood clots and bits of brain tissue in the tumor cavity could also lead to aseptic inflammatory responses, it would also increase the osmotic pressure of fluid in the cavity [18]. These two factors would form the osmotic gradient between the hypertonic fluid in the cavity and the fluid with normal osmotic pressure in the surrounding brain tissues. The osmotic gradient would make the fluid in the glioma surrounding brain tissues continuously absorbed into the cavity, resulting in increased hydrops in the cavity until a balance of the fluid osmotic pressure between in the cavity and the surrounding brain tissues is reached [19].
II Advantages and Disadvantages of Traditional Surgical Treatment
At present, in the field of neurosurgery, there are few summaries and analyses of tensile effusion in postoperative cavity of glioma. Through reviewing the reports of several cases, we found that craniotomy was the main treatment plan to tensile effusion in postoperative cavity in various neurosurgical centers [6, 7]. The main operating procedures of craniotomy are to loosen the adhesion between the cortical incision and the arachnoid or dura, expand the cortical fistula, clear the effusion in the tumor cavity, deal with the hemorrhage of the small blood vessels on the cavity wall, close the communication between the ventricle and the cavity and the continuous drainage from the cavity.
Through the above measures, although it can be achieved to clear the effusion in the cavity and prevent its regeneration, so as to remove the space-occupying effect of the effusion in the cavity and relieve intracranial hypertension, its disadvantages are also very obvious. Firstly, the preparation and the duration of general anesthesia craniotomy are too long to remove the space-occupying effect of effusion in cavity as soon as possible, resulting in the continuous aggravation of neurological dysfunction. Secondly, obvious surgical trauma of CD will cause additional heavy blow to the patients after glioma resection, resulting in prolonged postoperative hospital duration, which will not only hinder the implementation of subsequent concurrent radiotherapy and chemotherapy but also lead to the occurrence of some postoperative complications, such as pendulous pneumonia, deep vein thrombosis, etc. Finally, CD and subsequent intensive treatment will greatly increase the medical cost and bring additional heavy burden to patients and society.
III P&D Shows the Concept of Micro-Invasion and Precise Treatment
At present, in the field of glioma treatment, postoperative radiotherapy with concomitant and adjuvant temozolomide chemotherapy (the standard Stupp scheme) is of great importance to improve the progression-free survival (PFS) and overall survival (OS) of patients after the maximum safe resection of tumors [3]. Once the tensile effusion in postoperative cavity occurs, it can aggravate the patient's nervous system damage and even cause brain hernia leading to death [5]. Therefore, it is of particular importance to remove tensile effusion and to start the follow-up concurrent chemoradiotherapy as soon as possible. In order to achieve the above objectives, we treated the tensile effusion in postoperative cavity by placing an indwelling catheter (ventricular drainage tube or intravenous indwelling needle) to fully drain the effusion in the cavity and rinse the cavity though indwelling catheter with normal saline and achieved good results.
By analyzing the case data of 16 patients undergoing P&D, we believe that P&D has the following advantages in the treatment of tensile effusion in postoperative cavity compared with CD. Firstly, P&D can release the accumulated effusion in the cavity in a short time by the operation under local anesthesia in the ward, remove the space-occupying effect of the effusion in the cavity, reduce the intracranial pressure, and facilitate the recovery of neurological dysfunction as soon as possible. Secondly, P&D is a minimally invasive operation with minimal damage to the brain parenchyma around the cavity, which, compared with CD, can protect the patient's nerve function and provide a structural basis for the rapid recovery of postoperative nerve function.
Thirdly, we can flush the cavity with normal saline though an indwelling catheter, which can evacuate the disintegrated artificial absorbable materials (gelfoam or hemostatic yarn), micro-blood clots, and brain tissue debris after P&D. On the one hand, the flushing can alleviate the inflammatory reaction caused by the above material, reduce the vascular permeability to prevent the vascular seepage on cavity wall; the other is to reduce the osmotic pressure of effusion in cavity, eliminate the osmotic pressure gradient between hyperosmotic fluid in tumor cavity and normal osmotic fluid in surrounding brain tissue, so it can reduce the accumulation of effusion in the cavity. Finally, with the evacuation of effusion, the retraction and collapse of local brain parenchyma will open the adhesion of the cortical incision with arachnoid and dura, open the 'unidirectional flap', and make the effusion join the cerebrospinal fluid circulation in the subarachnoid space, accelerating the absorption of effusion in cavity. Within 3-5 days after the P&D, with the regression of parenchymal edema around the cavity and the decrease of intracranial pressure, the cortical incision would continue to open even if the drainage tube was removed later, preventing the re-accumulation of effusion in the cavity.
IV Clinical Analysis of P&D and CD
This retrospective analysis showed that both P&D and CD could relieve the space-occupying effect of tensile effusion in postoperative cavity and prevent its recurrence, and the GCS score of patients within 24 hours after P&D was better than CD (14.38±0.20 vs. 13.70±0.21, P<0.05) . On the one hand, P&D is performed under local anesthesia in the ward, the time of operation is short, so the nerve function damage caused by the space-occupying effect of the tensile effusion can be relieved more quickly. On the other hand, P&D is characterized by micro-invasion, with minimal damage to the brain parenchyma surrounding the cavity, providing a structural basis for the rapid recovery of postoperative nerve function. The micro-invasion of P&D is fully reflected in less intraoperative bleeding (6.25±0.56ml vs. 260.00±30.55ml, P<0.001) and postoperative hospital duration (6.69±0.54d vs. 11.30±0.60d, P<0.001).
In terms of the incidence of complications, there was no statistical difference between the P&D and CD. But it is worth noting that the inability of intracranial hemostasis under direct vision in P&D may lead to postoperative bleeding in puncture passage and on cavity wall. In addition, the need for placing a drainage tube postoperative for several days of P&D may lead to intracranial infection. The risks of intracranial hemorrhage and infection can be reduced by puncturing cavity to the nearest approach to the scalp, inserting the appropriate length of the drainage tube, strengthening the care of the drainage tube, and removing the tube as soon as possible.
Conclusion
Tensile effusion in postoperative cavity is a rare complication after glioma resection, which will seriously affect the treatment and prognosis of patients. P&D and CD can both safely and effectively treat postoperative tensile effusion in the cavity in glioma. Compared with CD, P&D has obvious advantages in terms of postoperative GCS score, duration operation, intraoperative bleeding, and postoperative hospital duration. P&D can better reflect the concepts of microinvasion and precise treatment for glioma. The shortcomings of this study due to the limited number of samples and the lack of long-term prognostic data of the patients in two groups. The conclusions of this study are yet to be confirmed by large-sample, multicenter and prospective relevant studies.
Ethical Approval
This study was approved by the Medical Ethical Committee of Xinqiao Hospital Affiliated with Third Military Medical University.
Consent
Written informed consent was obtained from all patients.
Competing Interests
None.
Funding
This work was partly supported by grants from the National Natural Science Foundation of China (NSFC 81972360) and the Joint Research Foundation from Chongqing Science and Technology Bureau and Chongqing Municipal Health Commission (2018ZDXM011).
Acknowledgements
We thank Mrs. Qing-Rui Li from the Institute of Pathology and Southwest Cancer Center, Southwest Hospital, Third Military Medical University, Chongqing, China for her work on sample collection.
Article Info
Article Type
Research ArticlePublication history
Received: Tue 18, Feb 2020Accepted: Mon 09, Mar 2020
Published: Mon 16, Mar 2020
Copyright
© 2023 Sheng-Qing Lv. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2020.02.16
Author Info
Guo-Hao Huang Lin Yang Sheng-Qing Lv Xun Qin Yan Xiang Yao Li Yu-Chun Pei Zuofeng Zhang
Corresponding Author
Sheng-Qing LvDepartment of Neurosurgery, Xinqiao Hospital, Third Military Medical University (TMMU), Chongqing City, P.R. China
Figures & Tables
Table 1:Clinical data of 26 cases of tensile effusion in postoperative cavity after glioma resection.
|
|
|
Total |
P&D |
CD |
P value |
|
|
|
26 |
16 |
10 |
|
|
Sex |
Male |
14 |
8 |
6 |
0.926 |
|
Female |
12 |
8 |
4 |
||
|
Male/Female |
1.17 |
1 |
1.5 |
|
|
|
Age |
Range |
13-78 |
13-78 |
15-64 |
|
|
Mean age |
52.54±15.22 |
54.50±15.53 |
49.40±14.96 |
0.417 |
|
|
>50/≤50 |
15/11 |
10/6 |
5/5 |
0.826 |
|
|
Pathological diagnosis |
OD |
4 |
1 |
3 |
0.374 |
|
DA |
2 |
2 |
0 |
||
|
AA |
5 |
4 |
1 |
||
|
GBM |
15 |
9 |
6 |
||
|
Cavity site |
Frontal lobe |
11 |
6 |
5 |
0.305 |
|
Temporal lobe |
17 |
10 |
7 |
||
|
Parietal lobe |
7 |
3 |
4 |
||
|
Occipital lobe |
4 |
4 |
0 |
||
|
WHO grade |
II |
6 |
3 |
3 |
0.585 |
|
III |
5 |
4 |
1 |
||
|
IV |
15 |
9 |
6 |
||
|
GCS Score |
Preoperative |
— |
11.00±0.26 |
10.20±0.27 |
0.3146 |
|
Postoperative |
— |
14.38±0.20 |
13.70±0.21 |
0.0373 |
|
|
Time of operation (min) |
— |
7.81±0.64 |
124.10±8.96 |
<0.001 |
|
|
Intraoperative blood loss (min) |
— |
6.25±0.56 |
260.00±30.55 |
<0.001 |
|
|
Postoperative hospital duration (d) |
|
6.69±0.54 |
11.30±0.60 |
<0.001 |
|
|
Postoperative complications |
5 |
3 |
2 |
0.657 |
|
OD: oligodendrocytoma; GBM: glioblastoma; DA: diffuse astrocytoma; AA: anaplastic astrocytoma; P&D: puncture and drainage; CD: craniotomy depression; GCS: Glasgow Coma Scale.
References
- Xiang Y, Zhu XP, Zhao JN, Huang GH, Tang JH et al. (2018) Blood-Brain Barrier Disruption, Sodium Fluorescein, And Fluorescence-Guided Surgery Of Gliomas. Br J Neurosurg 32: 141-148. [Crossref]
- Schwartzbaum JA, Fisher JL, Aldape KD, Wrensch M (2006) Epidemiology and molecular pathology of glioma. Nat Clin Pract Neurol 2: 494-503. [Crossref]
- Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ et al. (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10: 459-566. [Crossref]
- Hao S, Xue Z, Li D, Xiao X, Tang J et al. (2015) Analysis the reasons of emergency reoperation after skull base surgery: report of 14 cases. Chin J Neurosurg 31: 329-331.
- Rambarki O, Rajesh A (2014) Dreaded complications of mistaken identity - Hygroma vs effusion following decompressive craniotomy. J Neurosci Rural Pract 5: 305-307. [Crossref]
- Yu J, Xiong W, Chen Y (2010) Two cases of postoperative tension luminal effusion of intracranial tumors were reoperated: a case report. Chin J Surg 48: 560.
- Luo S, Ye F, Deng P, Liu X (2019) 2 cases of wound cavity tensile effusion after craniotomy and literature review. Chin J Clin Neurosurg 24: 173-175.
- Sanai N, Polley MY, McDermott MW, Parsa AT, Berger MS (2011) An extent of resection threshold for newly diagnosed glioblastomas. J Neurosurg 115: 3-8. [Crossref]
- Mazerand E, Le Renard M, Hue S, Lemée JM, Klinger E et al. (2017) Intraoperative Subcortical Electrical Mapping of the Optic Tract in Awake Surgery Using a Virtual Reality Headset. World Neurosurg 97: 424-430. [Crossref]
- Jenkinson MD, Barone DG, Bryant A, Vale L, Bulbeck H et al. (2018) Intraoperative imaging technology to maximise extent of resection for glioma. Cochrane Database Syst Rev 1: CD012788. [Crossref]
- Kubben PL, ter Meulen KJ, Schijns OE, ter Laak Poort MP, van Overbeeke JJ et al. (2011) Intraoperative MRI-guided resection of glioblastoma multiforme: a systematic review. Lancet Oncol 12: 1062-1070. [Crossref]
- Lin Yang, Yan Xiang, Guo Hao Huang, Hong Yao Lyu, Ke Jie Mou et al. (2018) Intraoperative fluorescence-guided resection of high-grade glioma: A systematic review. Glioma 1: 189-195.
- Peruzzi P, Bergese SD, Viloria A, Puente EG, Abdel Rasoul M et al. (2011) A retrospective cohort-matched comparison of conscious sedation versus general anesthesia for supratentorial glioma resection. Clinical article. J Neurosurg 114: 633-639. [Crossref]
- Eser Ocak P, Ocak U, Tang J, Zhang JH (2019) The role of caveolin-1 in tumors of the brain - functional and clinical implications. Cell Oncol (Dordr) 42: 423-447. [Crossref]
- Zhang C, Pan Y, Cai R, Guo S, Zhang X et al. (2020) Salvianolic acid A increases the accumulation of doxorubicin in brain tumors through Caveolae endocytosis. Neuropharmacology 167: 107980. [Crossref]
- Cai H, Liu W, Xue Y, Shang X, Liu J et al. (2015) Roundabout 4 regulates blood-tumor barrier permeability through the modulation of ZO-1, Occludin, and Claudin-5 expression. J Neuropathol Exp Neurol 74: 25-37. [Crossref]
- Yang L, Wang X, Zhen S, Zhang S, Kang D et al. (2012) Aquaporin-4 upregulated expression in glioma tissue is a reaction to glioma-associated edema induced by vascular endothelial growth factor. Oncol Rep 28: 1633-1638. [Crossref]
- Lee KS (2015) History of Chronic Subdural Hematoma. Korean J Neurotrauma 11: 27. [Crossref]
- Paredes I, Cicuendez M, Delgado MA, Martinez Pérez R, Munarriz PM et al. (2011) Normal pressure subdural hygroma with mass effect as a complication of decompressive craniectomy. Surg Neurol Int 2: 88. [Crossref]
