Spinal Intraosseus Schwannoma: A Case Report
A B S T R A C T
Introduction: Schwannomas are the most frequent extramedullary intradural spinal tumors, accounting for approximately 24% of all nerve sheath tumors in adults. In a lower percentage of cases they may be extradural or transdural (1-19%). Intramedullary schwannomas and intraosseous schwannoma are quite rare tumors representing.
Case Description: A 74-year-old woman presented intense low back pain for a few months, associated with left cruralgia and dysesthesia spread to both lower limbs. Magnetic resonance and computed tomography examination demonstrated an extramedullary intradural osteolytic lesion located in the posterior part of the vertebral body and pedicles of L1. The patient underwent a first surgery 4 years earlier with complete removal of the tumor but after about two years she had a recurrence. She was then subjected to a new intervention of decompression, removal of the tumor and arthrodesis. The histological findings were compatible with a classical schwannoma characterized by intraosseous growth and multifocal bone infiltration.
Conclusion: We reported a rare case of intraosseous schwannoma presenting as a recurrence four years after radical surgical removal of a classic lumbar schwannoma. Only 25 cases of these tumors have been previously reported and none of them as an intraosseous recurrence.
Keywords
Intraosseous schwannoma, spinal schwannoma, neurosurgery
Introduction
Spinal Schwannomas are benign tumors originating from Schwann cells most frequently located extramedullary intradural and account for approximately 24% of all nerve sheath tumors in adults [1]. In a lower percentage of cases, they may be extradural or transdural (1-19%) [2]. Intramedullary schwannomas and intraosseous schwannoma (IOs) are quite rare tumors representing, respectively, less than 1% and less than 0,5% of all schwannomas. IOs usually present as osteolytic lesions that can mimic other aggressive tumors [3]. Relevant literature available about IOs is mostly represented by single case-reports of neoplasms [4]. Here we report the case of an IOs presenting as recurrence of a lumbar common schwannoma completely removed four years earlier.
Case Description
I History and Radiographic Evaluation
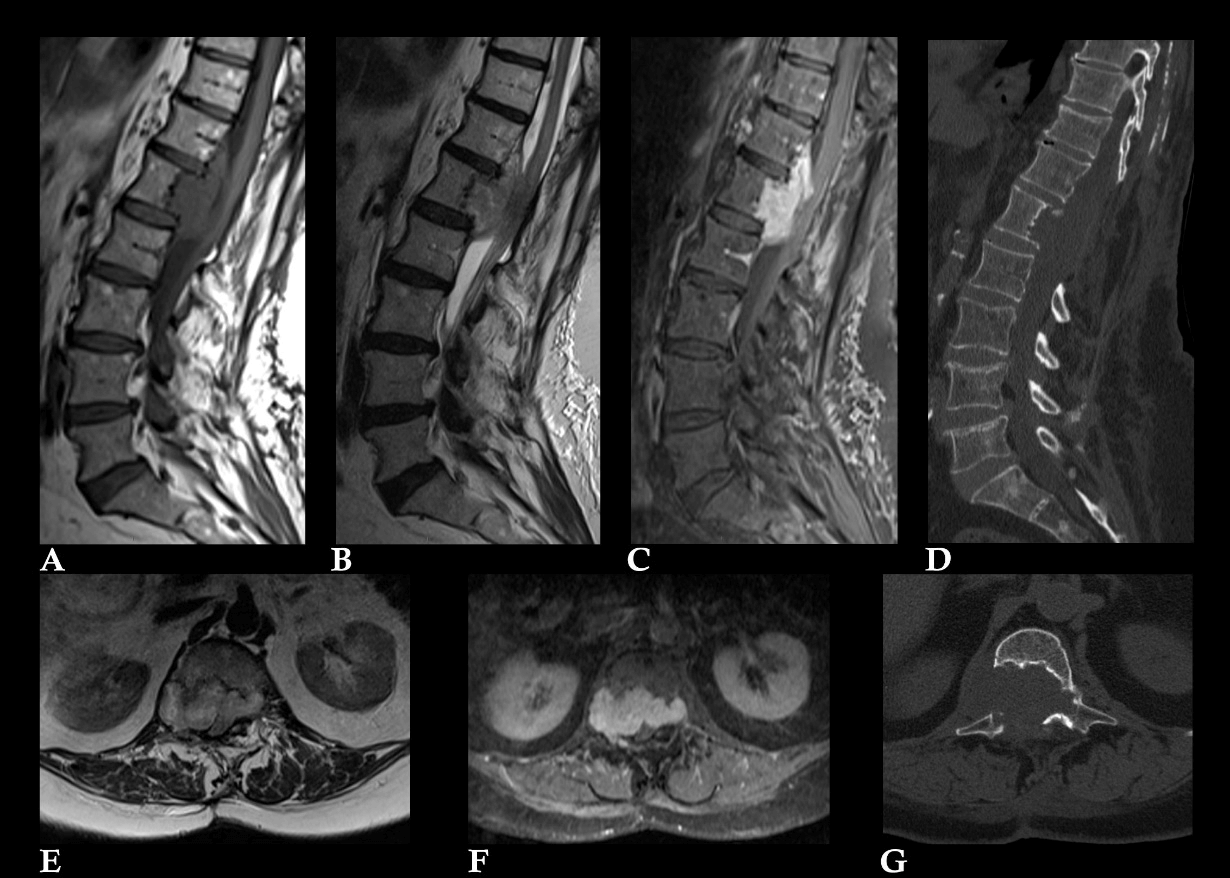
A 74-year-old woman presented intense low back pain for a few months, associated with left cruralgia and dysesthesia in both lower limbs. Magnetic resonance (MRI) and computed tomography (CT) examinations demonstrated an extramedullary osteolytic lesion (Figure 1) located in the posterior part of the vertebral body and pedicles of L1 characterized by an endocanalar component with bilateral intra-foraminal and right extra-foraminal involvement, extending cranially along the posterior wall of T12. At MRI the neoplastic tissue was isointense on T1-images relative to muscle, inhomogeneously hyperintense on T2-images with intense enhancement following contrast material injection.
Figure 1: Preoperative MR and CT showing an extramedullary intradural lesion located in the posterior part of L1, A) isointense on T1-images, B) & E) dishomogeneously hyperintense on T2-images with C) & F) intense enhancement. D) & G) CT images demonstrate osteolysis of the posterior part of the vertebral body and peduncles of L1 with a prominent extra-osseous endocanalar and bilateral foraminal component.
Four years earlier the patient underwent complete removal of a schwannoma arising at L1. Moderate proximal paraparesis and urinary incontinence were already present after the first procedure. No recurrence was detected at the MRI control one year after surgery. Considering the anamnesis, the patient underwent surgery without a prior biopsy of the lesion.
II Operative Description and Pathological Findings
Neurophysiological monitoring was used. After radioscopic control, scar lysis was performed with caudal and cranial extension. Lamina and articular processes were exposed from T11 to L3 with evidence of erosion of the right pedicle and transverse process of L1. Under radioscopic guidance, eight screws have been inserted (T11-T12-L2-L3). Left L1 arterectomy was also performed and the tumor was debulked from the left. The anterior part of the tumor was removed with aspiration and pituitary curettes until the residual anterior body of L1 was displayed. At the end of resection, the dural sac appeared well decompressed. The histological findings were compatible with a usual schwannoma characterized by intraosseous growth that showed multifocal bone infiltration (Figure 2A). The neoplasm had some cellular areas (Figure 2B) with some neoplastic elements characterized by greater cytokaryological atypia which can be attributed to elements with regressive/ancient atypia. The mitotic index was 1 X 10 HPF. Scattered lymphoid cells and siderophage were also noted. Immunohistochemical stainings revealed widespread positivity for S100 (Figure 2C) and SOX10 and retained expression of p16. Negative immunohistochemical for Melan A, EGFR, p53 (only a few positive cells). Ki67 labeling index was not evenly distributed and reached 10% in some areas.
Figure 2: Hematoxylin and eosin staining shows A) spindle cells infiltrating the bone, magnification 10x and B) some more cellular areas with a single mitotic figure, magnification 40x; C) S100 immunostaining shows diffuse nuclear and cytoplasmatic positivity in neoplastic cells, magnification 40x.
III Post-Operative Course and Follow Up
After surgery, the patient presented a distal paresis of the lower left limb, with 1/5 MRC of the anterior tibial nerve and lateral peroneal nerve. A CT scan after surgery showed the correct positioning of the screws. Fourteen days after the surgery the patient was transferred to rehabilitation and showed progressive neurological recovery.
Discussion
There are numerous variants of spinal schwannomas: multiple schwannomas, schwannoma with cystic degeneration, schwannoma with hemorrhage, melanotic schwannoma, intraosseous schwannoma, schwannoma growing into the foramen transversarium, large or giant schwannoma and infarcted schwannoma [5]. Even if there is little consensus on the definition of IOs, they are defined as tumors originally arising from the abutting nerve root and causing bone invasion [6]. IOs represent an extremely rare localization of schwannomas (0.7%) and arise in differential diagnosis with other intraosseous spinal lesions, such as metastasis, hemangiomas or primitive malignant tumors of the bone. What appears most likely of the different pathogenetic mechanism is that IOs is probably an intraosseous invasion of extraosseous nerve sheath tumor [6]. IOs are usually described as osteolytic lesions and the presence of neural foramen widening and vertebral body scalloping can be helpful in the diagnostic process. On MR imaging, schwannomas are typically isointense or hypointense relative to muscle on T1-weighted images and hyperintense on T2-weighted images frequently with inhomogeneous signal due to cysts formation, dense cellularity or hemorrhage [5]. Because of their benign nature, schwannomas can really increase in size before causing symptoms through mass effect. Generally, they are firm, solitary, and well capsulated. The first classification of benign spinal schwannomas, including giant and invasive lesions, proposed by Sridhar et al. in 2001 was later modified by Park et al. in 2009 [7, 8].
The latter authors proposed two additional classes: type VI (entirely intravertebral portion without intracanal component) and type VII (intraspinal tumor with erosion into vertebral bodies and extension into nerve root foramen). For cervical benign masses, the most used classification is PUTH classification [9]. IOs belong to class V, both in Sridhar and PUTH classification. The most frequent locations of spinal IOs are sacral and cervical segments, while only a few cases have been described at the thoracolumbar level. Searching the literature, a total of 35 papers on intraosseous spinal schwannomas was found. Excluding cervical and sacral localizations and malignant nerve sheath tumors, a series of 26 patients were collected and all characteristics are summarized in (Table 1). None of these included papers described the IOs as a form of recurrence of a commonly intradural extramedullary schwannoma. As shown in (Table 1), there is a slight predominance of females (15/26 patients) with a prevalence of clinical onset at less than 60 years. Only one patient was more than 70-year-old at the time of surgery. The typical presentation symptoms reported are lower back pain and lower limb weakness as in the reported patient. Surgical excision is the preferred treatment option. Those tumors are usually well encapsulated without invasion of the surrounding structures, making complete resection possible.
As shown in (Table 1) gross total resection was possible in all cases except one, with no recurrence at an average follow-up of 1 year. The present case had been operated with complete excision of lumbar schwannoma at the same level four years earlier, with no recurrence at one year follow up. The recurrence of the lesion was detected 4 years after surgery because symptoms appear. The presence of osteolytic pattern was suspected of a malignant transformation of the previous schwannoma, but the histological examination confirmed the benign nature of the tumor with a proliferation index reaching 10% in some areas. The problem of recurrence is still currently quite discussed in the literature. The rate of schwannoma recurrence following surgical resection has been reported to vary between 4% and 6% in previous surgical series. Recurrence usually occurs several years after surgery and appears to be associated with subtotal tumor removal [10]. Some recent papers have shown that the pre-operative size of the conventional spinal schwannoma and intralesional resection are the main risk factors for local post-operative recurrence [11]. Other authors argue that the probability of recurrence is associated with melanocytic and cellular schwannomas histologic variants [11]. Ancient schwannomas were originally described in 1951 by Ackerman and Taylor [12]. This term is applied to those tumors that are long standing and show evidence of degeneration, accounting for 0.8% of soft tissue tumors. Their histology findings are those seen in schwannomas, with the additional features of degeneration and mild nuclear atypia that can pose diagnostic dilemmas from the malignant counterpart [13].
Table 1: Previous reports of thoraco-lumbar intra-osseous schwannoma.
|
Author |
Gender/age |
Clinical presentation |
Level |
Treatment |
Hystological analysis |
|
Dickson, 1971 [15] |
Female, 51 y |
Pain in bilateral lower limbs |
L3 |
Surgery |
Schwannoma |
|
Nooraie H, 1997 [16] |
Male, 46 y |
Burst fracture |
L1 |
Surgery |
Schwannoma |
|
Chang CJ, 1998 [17] |
Male, 58 y |
Numbness and pain in lower extremities |
L4 |
Surgery |
Schwannoma |
|
Ramasamy, 2000 [18] |
Male, 37 y |
Back pain and weakness and numbness of lower extremities |
T12 |
Surgery |
Schwannoma with proliferation |
|
Inoaka, 2001 [27]
|
Female, 9 y
Female, 39 y |
Complete nontender distention of the back
Lumbar pain |
T10
L5 |
Surgery
Surgery |
Schwannoma
Schwannoma |
|
Gupta SP, 2005 [19] |
Female, 30y |
Backache and progressively increasing weakness in both lower limbs |
L2 |
Surgical resection |
|
|
Choudry Q, 2007 [20] |
Male, 18 y |
Back pain and deteriorating locomotor function |
D12 |
Surgical resection |
Intraosseous Schwannoma |
|
Cetinkal A, 2009 [6] |
Female, 55 y |
Progressive back pain and numbness |
D12 |
Surgical resection |
Schawannoma |
|
Park SC, 2009 [8] |
Female, 48 y |
Back pain and bilateral weakness of ankle dorsiflexion |
L4 |
Surgery |
Ancient Schwannoma |
|
Kojima M, 2011 [21] |
Male, 60 y |
Back pain and gait disturbance |
D9 |
Surgery |
|
|
Youn B, 2012 [22] |
Male, 65 y |
Lower back pain |
L2 |
Surgical resection |
Ancient Schwannoma |
|
Metoui L, 2013 [23] |
Female, 68 y |
Lumbalgia and cruralgia |
L3 |
Surgery + RT |
Malignant Schwannoma |
|
Song D, 2014 [3]
|
Male, 44 y |
Back pain and right lower extremity pain |
L5 |
Surgery |
Schwannoma |
|
Zhang F, 2015 [24] |
Male, 71 y
Female, 54 y |
Right lumbocrural pain myelopathy symptoms
Myelopathy symptoms |
L4
D9 |
Surgery
Surgery |
|
|
Wang YQ, 2018 [25] |
Male, 50 y
Female, 49 y
Female, 44 y
Female, 33 y
Female, 55 y
Female, 36 y
Male, 64 y
Male, 68 y |
Asymptomatic
Weakness, numbness, local pain
Numbness
Local pain, weakness
Local pain
Local pain, numbness
Asymptomatic
Local pain, numbness, weakness
|
L5
T9-T11
T2-T4
T7
T12
L5
T8
T5-T6 |
Subtotal resection Surgery (complete resection)
Surgery
Surgery
Surgery
Surgery
Surgery
Surgery |
Schwannoma
Schwannoma
Schwannoma
Schwannoma
Schwannoma
Schwannoma
Schwannoma
Schwannoma |
|
Jia S, 2019 [26] |
Female, 64 y |
Back pain |
D7/D8 + mobile schwannoma D5-D6 |
Surgical resection |
|
The microscopic picture of a typical schwannoma shows two components. The Antoni A pattern describes focal cellular areas with whorled nerve sheath cells. Antoni B pattern has hypocellular areas. In ancient schwannomas there is a relative loss of Antoni A and pronounced degenerative changes are attributed to the vascular insufficiency during growth of the tumor and the tumor size has been correlated with progressive degenerative features. They are characterized by cystic necrosis, stromal edema, xanthomatous change, fibrosis, perivascular hyalinization, calcification and degenerative nuclei with pleomorphism and hyperchromasia [14]. The presence of this component in our case allowed us to hypothesize that, despite complete resection after first surgery four years earlier, at the time of appearance of the recurrence, characteristics of bone invasion were found. As seen in (Table 1) the mean time of follow up for intraosseous schwannoma is 1 year, not sufficiently long to see any changes in biological aggression.
In our case, the intra-osseous involvement of tumor recurrence was probably due to the presence of a cellular component despite the complete tumor resection initially achieved. As seen in (Table 1) the mean follow-up time of IOs is 1 year, an interval not sufficiently long to see any changes in biological aggression.
Conclusion
We reported an extremely rare case of IOs presenting as a recurrence four years after radical surgical removal of a classical lumbar schwannoma. Only 25 cases of IOs have been previously reported, none of them characterized by recurrence. Intra-osseous invasion in our patient could be explained by the presence of a cellular component of the lesion within the bone as evidenced by the pathological examination.
Article Info
Article Type
Case ReportPublication history
Received: Sat 19, Dec 2020Accepted: Tue 29, Dec 2020
Published: Fri 15, Jan 2021
Copyright
© 2023 Giorgio Spatola. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2021.01.06
Author Info
Giorgio Spatola Migliorati Karol Liserre Barbara Morassi Mauro Giudice Lodoviga Fazio Marco Vivaldi Oscar Franzin Alberto
Corresponding Author
Giorgio SpatolaDepartment of Neurosurgery, Fondazione Poliambulanza, Brescia, Italy
Figures & Tables
Table 1: Previous reports of thoraco-lumbar intra-osseous schwannoma.
|
Author |
Gender/age |
Clinical presentation |
Level |
Treatment |
Hystological analysis |
|
Dickson, 1971 [15] |
Female, 51 y |
Pain in bilateral lower limbs |
L3 |
Surgery |
Schwannoma |
|
Nooraie H, 1997 [16] |
Male, 46 y |
Burst fracture |
L1 |
Surgery |
Schwannoma |
|
Chang CJ, 1998 [17] |
Male, 58 y |
Numbness and pain in lower extremities |
L4 |
Surgery |
Schwannoma |
|
Ramasamy, 2000 [18] |
Male, 37 y |
Back pain and weakness and numbness of lower extremities |
T12 |
Surgery |
Schwannoma with proliferation |
|
Inoaka, 2001 [27]
|
Female, 9 y
Female, 39 y |
Complete nontender distention of the back
Lumbar pain |
T10
L5 |
Surgery
Surgery |
Schwannoma
Schwannoma |
|
Gupta SP, 2005 [19] |
Female, 30y |
Backache and progressively increasing weakness in both lower limbs |
L2 |
Surgical resection |
|
|
Choudry Q, 2007 [20] |
Male, 18 y |
Back pain and deteriorating locomotor function |
D12 |
Surgical resection |
Intraosseous Schwannoma |
|
Cetinkal A, 2009 [6] |
Female, 55 y |
Progressive back pain and numbness |
D12 |
Surgical resection |
Schawannoma |
|
Park SC, 2009 [8] |
Female, 48 y |
Back pain and bilateral weakness of ankle dorsiflexion |
L4 |
Surgery |
Ancient Schwannoma |
|
Kojima M, 2011 [21] |
Male, 60 y |
Back pain and gait disturbance |
D9 |
Surgery |
|
|
Youn B, 2012 [22] |
Male, 65 y |
Lower back pain |
L2 |
Surgical resection |
Ancient Schwannoma |
|
Metoui L, 2013 [23] |
Female, 68 y |
Lumbalgia and cruralgia |
L3 |
Surgery + RT |
Malignant Schwannoma |
|
Song D, 2014 [3]
|
Male, 44 y |
Back pain and right lower extremity pain |
L5 |
Surgery |
Schwannoma |
|
Zhang F, 2015 [24] |
Male, 71 y
Female, 54 y |
Right lumbocrural pain myelopathy symptoms
Myelopathy symptoms |
L4
D9 |
Surgery
Surgery |
|
|
Wang YQ, 2018 [25] |
Male, 50 y
Female, 49 y
Female, 44 y
Female, 33 y
Female, 55 y
Female, 36 y
Male, 64 y
Male, 68 y |
Asymptomatic
Weakness, numbness, local pain
Numbness
Local pain, weakness
Local pain
Local pain, numbness
Asymptomatic
Local pain, numbness, weakness
|
L5
T9-T11
T2-T4
T7
T12
L5
T8
T5-T6 |
Subtotal resection Surgery (complete resection)
Surgery
Surgery
Surgery
Surgery
Surgery
Surgery |
Schwannoma
Schwannoma
Schwannoma
Schwannoma
Schwannoma
Schwannoma
Schwannoma
Schwannoma |
|
Jia S, 2019 [26] |
Female, 64 y |
Back pain |
D7/D8 + mobile schwannoma D5-D6 |
Surgical resection |
|

References
- Rodriguez FJ, Folpe AL, Giannini C, Perry A (2012) Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol 123: 295-319. [Crossref]
- Chamberlain MC, Tredway TL (2011) Adult primary intradural spinal cord tumors: a review. Curr Neurol Neurosci Rep 11: 320-328. [Crossref]
- Song D, Chen Z, Song D, Li Z (2014) Lumbar intraosseous schwannoma: case report and review of the literature. Turk Neurosurg 24: 982-986. [Crossref]
- Summers S, Jose J, Barrera CM, Pretell Mazzini J, Subhawong T et al. (2018) Intraosseous schwannomas involving the sacrum: Characteristic imaging findings and review of the literature. Neuroradiol J 31: 531-540. [Crossref]
- Parmar HA, Ibrahim M, Castillo M, Mukherji SK et al. (2007) Pictorial essay: diverse imaging features of spinal schwannomas. J Comput Assist Tomogr 31: 329-334. [Crossref]
- Cetinkal A, Atabey C, Kaya S, Colak A, Topuz AK (2009) Intraosseous schwannoma of thoracic 12 vertebra without spinal canal involvement. Eur Spine J 2: 236-239. [Crossref]
- Sridhar K, Ramamurthi R, Vasudevan MC, Ramamurthi B (2001) Giant invasive spinal schwannomas: definition and surgical management. J Neurosurg 94: 210-215. [Crossref]
- Park SC, Chung SK, Choe G, Kim HJ (2009) Spinal intraosseous schwannoma: a case report and review. J Korean Neurosurg Soc 46: 403-408. [Crossref]
- Jiang L, Lv Y, Liu XG, Ma QJ, Wei F et al. (2009) Results of surgical treatment of cervical dumbbell tumors: surgical approach and development of an anatomic classification system. Spine (Phila Pa 1976) 34: 1307-1314. [Crossref]
- Fehlings MG, Nater A, Zamorano JJ, Tetreault LA, Varga PP et al. (2016) Risk Factors for Recurrence of Surgically Treated Conventional Spinal Schwannomas: Analysis of 169 Patients From a Multicenter International Database. Spine (Phila Pa 1976) 41: 390-398. [Crossref]
- Clinical outcomes following resection of giant spinal schwannomas: a case series of 32 patients (2017) J Neurosurg Spine 26: 494-500. [Crossref]
- Ackerman LV, Taylor FH (1951) Neurogenous tumors within the thorax; a clinicopathological evaluation of forty-eight cases. Cancer 4: 669-961. [Crossref]
- Malizos K, Ioannou M, Kontogeorgakos V (2013) Ancient schwannoma involving the median nerve: a case report and review of the literature. Strategies Trauma Limb Reconstr 8: 63-66. [Crossref]
- Lee YS, Kim JO, Park SE (2010) Ancient schwannoma of the thigh mimicking a malignant tumour: a report of two cases, with emphasis on MRI findings. Br J Radiol 83: e154-e157. [Crossref]
- Dickson JH, Waltz TA, Fechner RE (1971) Intraosseous neurilemoma of the third lumbar vertebra. J Bone Joint Surg Am 53: 349-355. [Crossref]
- Nooraie H, Taghipour M, Arasteh MM, Daneshbod K, Erfanie MA (1997) Intraosseous schwannoma of T12 with burst fracture of L1. Arch Orthop Trauma Surg 116: 440-442. [Crossref]
- Chang CJ, Huang JS, Wang YC, Huang SH (1998) Intraosseous schwannoma of the fourth lumbar vertebra: case report. Neurosurgery 43: 1219-1222. [Crossref]
- Ramasamy P, Shackleford I, Al Jafari M (2000) Schwannoma of t12 vertebra: case report and review of literature. Sarcoma 4: 185-190. [Crossref]
- Gupta SP, Agarwal A (2005) Intraosseous neurilemmoma of L2 vertebra--a case report. Indian J Pathol Microbiol 48: 367-369. [Crossref]
- Choudry Q, Younis F, Smith RB (2007) Intraosseous schwannoma of D12 thoracic vertebra: diagnosis and surgical management with 5-year follow-up. Eur Spine J 16: 283-286. [Crossref]
- Kojima M, Seichi A, Yamamuro K, Inoue H, Kimura A et al. (2011) Intraosseous schwannoma originating from the posterior column of the thoracic spine. Eur Spine J 20: S153- S156. [Crossref]
- Youn B, Lee SH, Kim ES, Eoh W (2012) Intraosseous schwannoma with ancient change on the lumbar spine. Br J Neurosurg 26: 561-563. [Crossref]
- Metoui L, Ajili F, Maiza M, Ammar MB, Gharsallah I et al. (2013) Spinal cord compression revealing an intraosseous schwannoma. Case Rep Med 2013: 913218. [Crossref]
- Zhang F, Lu F, Jiang J, Wang H (2015) Two Case Reports and an Updated Review of Spinal Intraosseous Schwannoma. J Korean Neurosurg Soc 57: 478-483. [Crossref]
- Wang YQ, Hu JX, Yang SM, Jiang L, Liu XG et al. (2018) Intraosseous schwannoma of the mobile spine: a report of twenty cases. Eur Spine J 27: 3092-3104. [Crossref]
- Jia S, Zheng W, Ruan J, Chen T, Huang Y et al. (2019) Mobile thoracic schwannoma combined with intraosseous schwannomas: A case report. Medicine (Baltimore) 98: e14153. [Crossref]
- Inaoka T, Takahashi K, Hanaoka H, Aburano R, Tokusashi Y et al. (2001) Paravertebral neurinoma associated with aggressive intravertebral extension. Skeletal Radiology 30: 286-289. [Crossref]
