Plasma Treatment Effects on Oral Candida albicans Biofilms
A B S T R A C T
The objective of this study is to evaluate the plasma treatment effects on oral fungal biofilms. Candida albicans biofilms were developed on the 48-well plate to serve as a model of oral fungal biofilm. The treatment of 0.2% chlorhexidine digluconate (CHX) was used as a positive control compared with plasma treatments. The efficacy of treatments was determined by 3-(4,5-dimethylazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay and confocal laser scanning microscope (CLSM). The survival percentage of Candida albicans decreased from 52% to 27% as the plasma power increased from 6mA to 8mA and plasma exposure time extended from 2 min to 10 min. Moreover, it was found that there is a synergistic effect of the combination of plasma and CHX treatments. Scanning electron microscopy (SEM) examination indicated severe cell damages resulting from plasma treatment. In conclusion, the low-temperature plasma treatment is effective in deactivating Candida albicans biofilms and thus provides a promising alternative to disinfect oral fungal biofilms.
Keywords
Low-temperature plasma, oral fungal biofilm, biofilm disinfection, Candida albicans
Introduction
Candida albicans (C. albicans), an asexual diploid fungus, is one of the organisms that prefer human mouths as their primary habitat. It is an oral commensal bacterium and can be found in 40% of normal subjects and 75% of patients who wear a denture [1]. C. albicans has the ability to morph from a free-floating form to a biofilm form that grows as a community. Biofilm may allow for overpopulation and nutrient load control. The formation of biofilm requires C. albicans to grow hyphae which are multinucleated cell filaments [2]. The hyphae could help C. albicans survive under unfavourable conditions such as increased temperature, increased pH levels, nutrient starvation and increased cell density [3]. Numerous studies have reported that there is an association between C. albicans and denture-related stomatitis which is termed as Candida-associated denture stomatitis [4, 5]. The characteristic presenting features of denture stomatitis are chronic erythema and inflamed mucosa, particularly under the upper denture. The patients may complain a burning sensation, discomfort, or bad taste [6, 7].
A number of antifungal agents are available on the market. The most popular antifungals used against Candida infection are polyenes (such as amphotericin B and nystatin) and azoles (such as fluconazole and itraconazole) [5, 8, 9]. However, the increased use of antifungal agents in recent years has resulted in the development of resistance to these drugs [10-13]. Besides these two types of drugs, chlorhexidine, contained in mouth rinses, is also an appropriate option to conventional antifungals in the management of oral candidiasis [14]. A common side effect associated with chlorhexidine oral rinses is an increase in staining of teeth and other oral surfaces. Gas plasma, described as the fourth fundamental state of matter, consists of many active plasma species/particles including positively charged ions and negatively charged electrons, electronically excited neutral atoms and molecules, radical, and ultraviolet (UV) photons [15]. These active particles are responsible for the antimicrobial efficacy of plasma [16]. Low-temperature plasma generated at atmospheric pressure becomes a potential tool for oral microbial decontamination [16-19]. Previous studies have demonstrated the low temperature plasma was capable of deactivating planktonic cavity-causing bacteria [20-24]. Several other studies have confirmed the antimicrobial capability for Streptococcus mutans biofilms, which are the most significant cariogenic microorganism [5, 25-28]. These works encourage the application of low-temperature atmospheric plasma on disinfection of oral candidiasis, which is a yeast/fungi infection of the genus Candida that develops on the mucous membranes of the mouth. In this paper, plasma treatment effects on oral Candida albicans biofilms were studied in vitro.
Materials and Methods
I The Low-Temperature Atmospheric Plasma Brush
A low-temperature atmospheric plasma brush was employed in this study. The plasma brush is composed of a gas source, a power supply, and a discharge chamber, as shown in (Figure 1A). The gas, argon and oxygen, pass through the discharge chamber at a controlled flow rate. An electrical field was applied to the two electrodes located inside the chamber to ignite a DC glow discharge by a DC power supply (Spellman SL60, New York, USA). The power input in generating and sustaining DC atmospheric pressure plasma is at the level of several watts or tens of watts. The low power input of this plasma system provides a unique advantage in the low power consumption. The physical properties of the system have been described in the other works [22, 29]. A thermometer (VWR 61066-104, Chicago, IL) was used to measure the plasma temperature. The ignited plasma touched the tip of the thermometer, as seen in (Figure 1B). The temperature was recorded every 20 seconds and the starting temperature was the beginning of plasma ignition. The plasma was operated at 6mA and 8mA with 3000 standard cubic centimeters per minute (sccm) of argon and 30 sccm of oxygen.
Figure 1: A) Schematic of plasma brush system and B) a photograph of temperature measurement of the plasma flame.
II Biofilm Preparation
One milliliter of frozen-stored C. albicans (ATCC 18804) was defrosted for 15 minutes and cultured overnight in 25mL Sabouraud (SAB) liquid medium (Fisher Science, Pittsburgh, PA, USA). The sub-cultured C. albicans suspension was diluted to the density of 1×107 cells/mL. Then 600μL of diluted C. albicans suspension was added into each well of the 48-well plate and incubated in an incubator for 24 hours at 37℃.
III Plasma Treatment
As the biofilms were well developed, the SAB medium was removed from the wells by gentle aspiration. The biofilms were washed twice with phosphate-buffered saline (PBS) to remove non-adherent bacterial cells. Before any disinfection treatment, biofilms were dried in the hood for 5 minutes. All prepared biofilms were divided into 8 groups to receive different treatments, as seen in (Table 1). One group of prepared biofilms was treated with 150μL of 0.2% chlorhexidine digluconate (CHX) solution (Sigma Aldrich, St. Louis, MO) for ten minutes. Another six groups of biofilms were treated with selected plasma conditions. The six different plasma conditions applied in this study were 6mA power supply treated for 2, 5 or 10 minutes, and 8mA power supply treated for 2, 5 or 10 minutes. The gas flow was 3000 sccm of argon with 30 sccm of oxygen. In addition, one group without any disinfection treatment was regarded as a control group. Each group contained five samples. In order to investigate the synergistic effect of plasma and CHX treatment on the fungal biofilms, the prepared biofilms were first treated by plasma as described above and followed by CHX treatments [Group 9 and Group 10 shown in (Table 1)]. Five samples were tested for each group.
IV MTT Assay
Cell proliferation kit I (Roche Diagnostics, Fisher Scientific, Pittsburgh, PA, USA) was used for the 3-(4, 5-dimethylazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. This kit includes MTT label reagent and solubilization (check the name) solution. After disinfection treatments, 500μL PBS was added to each well and blown with a pipette for 10 times to detach the bacterial cells from the well substrate. The 500μL PBS suspension containing bacterial cells was transferred into a 1.5mL centrifuge tube and centrifuged at 6000 rpm for eight minutes. After centrifugation, PBS was removed, and cell bullet was kept in the tube. 200μL fresh PBS was added into the tube to suspend cells on a vortexer for 30 seconds. The cell suspension was mixed with 20μL of the MTT label reagent in the tube and incubated for four hours at 37℃.
Afterwards, 200μL of the solubilization solution in the kit was added to the tube. The tubes were put into an incubator overnight. Then, 100μL of the solution was transferred to a 96-well plate for optical density assay at 570 nm. The bacterial survival percentage was calculated against the mean of biofilms on control samples. The experiment was performed three times to obtain means and standard errors of means.
V Confocal Laser Scanning Microscope (CLSM) Analysis
In order to conduct CLSM measurement, C. albicans biofilms were developed on polystyrene strips and treated by following the procedure in (Table 1). The strips were cut from Petri dishes (Fisher Scientific, Pittsburgh, PA 15275, USA). After treatments, the adherent biofilms were stained using a Live/Dead BacLight bacterial viability kit (Invitrogen, Carlsbad, CA), which includes propidium iodide (PI) and SYTO9. In the experiment, SYTO9 and PI were mixed together according to the manufacturer’s instructions. 0.5mL of the mixture was dropped on the biofilm surface and then incubated in the dark for 15 minutes in a hood. After removing the staining medium, the biofilms were immersed in a 10% formalin solution for 30 minutes in order to fix the structure of biofilm. Then, the samples were stored in chambered cover glasses with PBS until CLSM analysis. The biofilms were visualized with a confocal laser scanning microscope (Zeiss LSM 510, Carl Zeiss Micro-Imaging GmbH, Jena, Germany). 3-Dimensional structural reconstruction of CLSM data was performed using Imaris 4.0 (Bitplane AG, Zurich, Switzerland).
Table 1: Treatment
conditions for each group.
|
Group |
Treatment |
Time |
|
1 |
None |
None |
|
2 |
0.2% CHX solution |
10 min |
|
3 |
6 mA Plasma |
2 min |
|
4 |
5 min |
|
|
5 |
10 min |
|
|
6 |
8 mA Plasma |
2min |
|
7 |
5 min |
|
|
8 |
10 min |
|
|
9 |
6mA plasma 2 min+ 0.2% CHX 10 min |
|
|
10 |
8mA plasma 5 min+ 0.2% CHX 10 min |
|
VI Scanning Electronic Microscope (SEM) Examination
The treated and untreated biofilms were prepared as described in (Table 1). Samples were fixed with 2% glutaraldehyde- 2% paraformaldehyde in 0.1 M cacodylate buffer (pH=7.4) for 2 hours at 4℃ and subsequently fixed with 0.1% osmium tetroxide for 1 hour. After being washed in ultrapure distilled water, the biofilms were dehydrated in graded ethanol series (20%, 50%, 70%, 90%, 100%, 100% and 100%) for 15 min each. After critical point drying and platinum sputter coating, samples were examined by using a scanning electron microscope (FEI Quanta 600FEG).
VII Statistical Analyses
For experimental data evaluation, the Sigma statistical analysis programme was used. The comparisons of different treatment groups were performed by using the one-way analysis of variance (ANOVA), and differences with a P value less than 0.05 (p < 0.05) among the groups were considered to be statistically significant.
Results
I Plasma Temperature
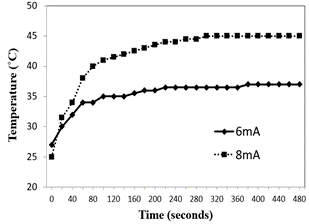
The plasma temperature was measured with a thermal meter, as shown in (Figure 2). At 6mA, the plasma temperature rises sharply from 27℃ to 34℃ during the first 60 seconds. Afterwards, the temperature gently increases to 35℃ in the next 300 seconds. Eventually, the temperature stays at 35℃ till the plasma brush is turned off. In contrast, the temperature at 8mA presents a similar trend but different peak temperature. At 8mA, the plasma temperature rises sharply from 24℃ to 40℃ during the first 80 seconds. Afterwards, the temperature gently increases to 45℃ in the next 200 seconds. Eventually, the temperature stays at 45℃ till the plasma brush is turned off. The peak temperature at 8mA is 10℃ higher than that at 6mA.
Figure 2: Time dependence of plasma flame temperature of the low-temperature atmospheric plasma brush operated under 6mA and 8mA, respectively.
II Disinfection Effect of Plasma Treatment
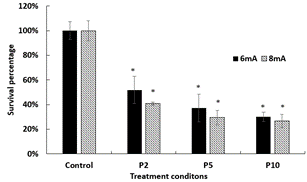
Biofilms were plasma treated for 2, 5 and 10 minutes under 6mA or 8mA power supply, respectively. MTT assay was performed to measure the disinfection efficiency of plasma treatment on the C. albicans biofilm. This is a widely used assay to measure bacterial viability. A significant reduction of fungal survival rate was observed on all plasma treatment groups as compared to the untreated control group shown in (Figure 3). Furthermore, the disinfection efficiency increased with plasma treatment time. At 6mA, the fungal survival rates were 52% for 2 min plasma treatment, 37% for 5 min plasma treatment, and 30% for 10 min plasma treatment, respectively. Likewise, at 8mA, the fungal survival rates were 32% for 2 min plasma treatment, 30% for 5 min plasma treatment and 27% for 10 min plasma treatment, respectively. In addition, a higher plasma power could provide better disinfection efficiency, especially for short plasma treatment time. The survival rate under 8mA was 10% less than that under 6mA for 2 min plasma treatment, while there were only 7% and 3% differences between 8mA and 6mA as the plasma treatment time extends from 5 min to 10 min.
Figure 3: Plasma treatment time dependence of C. albicans biofilm survival percentage. Control: untreated. P2: 2-minute plasma treatment. P5: 5-minute plasma treatment. P10: 10-minute plasma treatment. The asterisk shows significant differences between different conditions and the control group (p < 0.05).
III Synergistic Effects of Plasma Treatment with CHX against C. albicans Biofilms
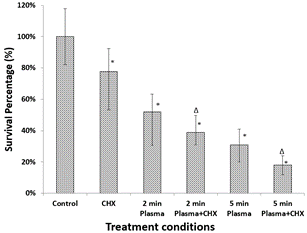
Figure 4 shows the fungal survival rate of the biofilms with CHX treatment only and plasma treatment subsequently followed by CHX treatment. It can be seen that CHX treatment has a certain capability of inactivating C. albicans in the biofilms. With 10 min CHX treatment time, the survival rate was 77% compared with the untreated control group. In contrast, the plasma treatments subsequently followed by the CHX treatment became much more effective in fungal deactivation. CHX treatment on the 2 min plasma-treated biofilms induced as low as 39% fungal survival rate. Likewise, CHX treatment on the 5 min plasma treated biofilms had even lower 18% fungal survival rate. The combination of plasma treatment and CHX treatment also offered better deactivation effects than single plasma treatment. The single 2 min plasma treatment presented 52% survival rate and the single 5 min plasma treatment presented 31% survival rate, as seen in (Figure 4).
Figure 4: Biofilm survival percentage with different plasma and CHX treatments. The asterisk shows significant differences between the different treatment conditions and the control group (p < 0.05). The triangle shows significant differences between the single plasma treatments and the combination of plasma and CHX treatments.
The confocal laser scanning microscope (CLSM) with Live/Dead BacLight method was applied to examine the fungal viability in the biofilms. According to the mechanism of Live/Dead BacLight dyes, the viable fungi would be stained with SYTO9 and emit a green fluorescence, whereas dead fungi would be stained with PI and emit a red fluorescence [30]. As seen in (Figure 5), multilayer biofilms were formed on polystyrene strips. Most fungi on the untreated control samples (Figure 5A) were green, while a majority of fungi on other samples emitted red fluorescence. In other words, all of the treatments used in this experiment were capable of deactivating fungi in biofilms. Moreover, several green dots could be observed in the groups of CHX treatment (Figure 5B), 6mA plasma treatment for 2 min (Figure 5C) and 8mA plasma treatment for 5 min (Figure 5E). However, there are no green dots presented in the samples with combinations of plasma treatment and CHX treatment, as seen in (Figures 5D & 5F). In other words, combined treatments using plasma and CHX were more efficient in deactivating C. albicans biofilms than both the single CHX treatment and single plasma treatment.
Figure 5: CLSM images of A) Untreated Control; B) CHX treatment; C) 5 min treatment by 6mA plasmas; D) 2 min treatment by 6mA plasmas + CHX treatment; E) 5 min treatment by 8mA plasmas; F) 5 min treatment by 8mA plasmas + CHX treatment. Green dots indicate the living fungal cells and red dots indicate the dead fungal cells.
As shown in (Figure 6), SEM was performed to analyse the fungal morphology changes before and after different treatments. In (Figure 6A), both yeast form and hyphae form could be observed, and the morphology of cells was intact in the untreated control samples. In contrast, only yeast form of C. albicans was observed after CHX treatment (Figure 6B). The SEM micrographs in (Figures 6C & 6E) show that single plasma treatments caused damages to fungal cell walls. As the plasma treatments followed by CHX treatment, the damage became increasingly more severe with massive cell wall ruptures as shown in (Figures 6D & 6F). However, most of the damaged cells were located in the superficial layer of the biofilms. There were still some intact fungal cells in the underneath cell layers in the biofilms.
Figure 6: SEM images of A) Untreated Control; B) CHX treatment; C) 2 min treatment by 6mA plasmas; D) 2 min treatment by 6mA plasmas + CHX treatment; E) 5 min treatment by 8mA plasmas; F) 5 min.
Discussion
C. albicans plays an important role in oral candidiasis, which is the most common fungal infection occurring in the mouth [31]. C. albicans biofilms are also found on the surface of implantable medical devices, including dental implants and dentures, and cause prosthesis stomatitis [1, 5]. One common preventive infection method of oral C. albicans biofilm formation is the use of CHX containing mouth wash [32]. However, biofilm presents CHX resistance, which hinders the efficacy of CHX [33, 34]. Depending on different test systems, the CHX concentration and exposure time also affect the efficacy of CHX in the disinfection of C. albicans biofilms. Our results using CHX treatment are consistent with the results by Koban et al., who reported only a moderate effect on C. albicans biofilms by 0.1% CHX treatment for 10min [35]. Therefore, alternative approaches are needed to enhance the antifungal efficacy.
Low-temperature atmospheric plasma is increasingly claimed to have potent antibacterial activity. It has been shown to be very effective against a wide range of pathogenic bacteria and fungi [17, 18, 35, 36]. Severe damages to the cell wall and rupture of microorganisms can be caused by plasma treatment, leading to cell death [37]. Previous studies have demonstrated that low-temperature atmospheric plasma brush is effective in the inactivation of bacteria in the planktonic form and biofilm form [20, 21, 38]. These findings indicated that the low-temperature atmospheric plasma could be a promising technique in the prevention and treatment of various oral diseases. The working temperature of the plasma is less than 45℃, which indicates no heat damage from plasma heat.
This study demonstrated that low-temperature atmospheric plasma treatment could significantly disinfect C. albicans biofilm. MTT assay results showed that as much as 77% of C. albicans biofilm were inactivated after 10 min of 8mA plasma treatment. Analysis of CLSM indicated that much less viable fungal cells were observed after plasma treatment than that in the untreated control group. As seen in SEM images, plasma treatment resulted in significant morphology changes on fungal cells when compared with the untreated controls. Damages on the cell wall were noticed with the cells on the top layer of the C. albicans biofilms. The results from these assessing methods all showed the capability and efficacy of plasma treatment in deactivating C. albicans biofilms. The plasma treatment effects on C. albicans biofilms were also compared with CHX treatment. The results obtained in this study indicate that plasma treatment is more effective than CHX in deactivating fungal cells in biofilms. As seen in (Figure 4), the fungal reduction by CHX treatment was 33%, whereas the fungal reduction by plasma treatment was as high as 49% (for 2 min treatment by 6mA plasmas) and 70% (for 5 min treatment by 8mA plasmas). Koban et al. also reported similar results with their plasma systems. A log10 reduction factor of 6 was achieved by a plasma jet [35, 39].
It should be noted that the interaction mechanisms of plasma and CHX with bacteria are completely different. The antibacterial action of CHX is due to an increase in cellular membrane permeability followed by coagulation of the cytoplasmic macromolecules [40-42]. In contrast, the antifungal mechanism of plasma treatment is more complex than that of CHX. Plasma treatment affects not only cell walls but also other cell components, including proteins, lipids, and DNA/RNA [43]. Fungal disinfection by plasma treatment is based on reactive plasma species, including ultra-violet (UV) radiation, electronically excited neutral species, free radicals and charged particles in gas plasma [21, 37, 44]. As a result, fungal cell walls can be damaged to cause further cell lyses. Besides cell lyses, when the damage is not very extensive, Na+, Ca2+ and water can enter the cell and cause swelling, which will also cause cell death [45]. This also explains that morphological changes were observed with plasma-treated cells in SEM images, while these cells were found dead by CLSM analysis.
In this study, synergistic effects were observed with plasma treatment and CHX treatment. Fungal cell structure could be severely damaged by plasma treatment. Plasma species are capable of damaging fungal cell walls through ionic bombardment by energetic plasma particles [38]. The fungal cell walls could also be damaged through chemical etching by chemically reactive plasma species [46]. The plasma-induced fungal cell damages provide an opportunity to improve the effectiveness of antiseptics commonly used for various biofilm treatments. Plasma-induced damages on fungal cell walls could make the fungal cells become more accessible by the antimicrobial chemicals to deactivate the fungi. Therefore, a combination of plasma treatment with the commonly being used antimicrobial chemotherapy could further improve the disinfection effectiveness and efficiency for various fungal biofilms, which are usually serious challenges in clinical practice in both dental and medical fields. Furthermore, the combination approach could also shorten the plasma treatment time and therefore will be more practical in clinical applications.
A limitation in this study is the related setup of the plasma treatments. C. albicans biofilms grew on the bottom of a 48-well plate. During the plasma treatment, plasma brush was inserted into the wells to treat the biofilms. The relative position between the plasma flame and the biofilms was fixed with the treatment area limited to the top surfaces of the biofilms. In MTT assay, edge effects were observed with the plasma-treated groups, in which a purple ring formed at the edge of the well bottom. In other words, the fugal cells on edge were still viable because the reactive plasma species did not spread over the entire bottom area. Koban et al. also observed similar effects, which presented clear boundaries of damaged cells between plasma-treated area and non-treated area [35].
Conclusion
The in vitro results obtained in this study conclusively demonstrated that low-temperature atmospheric plasma treatment is very effective in deactivating C. albicans oral biofilms. Plasma treatment exceeded the antifungal effects of the commonly used CHX treatment. Furthermore, the combination of plasma and CHX treatment increased the antifungal efficiency against C. albicans biofilms. The results indicate low-temperature plasma treatment provides a promising alternative to disinfect oral fungal biofilms.
Acknowledgement
This study was supported, in part, by the US National Institute of Health (NIH) under grant numbers of 5R01DE021431 and 5R44DE019041.
Conflicts of Interest
Meng Chen, Hongmin Sun, and Qingsong Yu have financial interest in Nanova, Inc.
Article Info
Article Type
Research ArticlePublication history
Received: Wed 28, Apr 2021Accepted: Fri 14, May 2021
Published: Tue 01, Jun 2021
Copyright
© 2023 Qingsong Yu. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.DOBCR.2021.02.05
Author Info
Qing Hong Xiaoqing Dong Meng Chen Hongmin Sun Liang Hong Yong Wang Qingsong Yu
Corresponding Author
Qingsong YuDepartment of Mechanical and Aerospace Engineering, University of Missouri, Columbia, Missouri, USA
Figures & Tables
Table 1: Treatment
conditions for each group.
|
Group |
Treatment |
Time |
|
1 |
None |
None |
|
2 |
0.2% CHX solution |
10 min |
|
3 |
6 mA Plasma |
2 min |
|
4 |
5 min |
|
|
5 |
10 min |
|
|
6 |
8 mA Plasma |
2min |
|
7 |
5 min |
|
|
8 |
10 min |
|
|
9 |
6mA plasma 2 min+ 0.2% CHX 10 min |
|
|
10 |
8mA plasma 5 min+ 0.2% CHX 10 min |
|



References
1.
Pfaller MA, Diekema DJ (2007) Epidemiology of invasive candidiasis: a
persistent public health problem. Clin Microbiol Rev 20: 133-163. [Crossref]
2.
Sudbery P, Gow N, Berman J (2004) The distinct morphogenic states of
Candida albicans. Trends Microbiol 12: 317-324. [Crossref]
3.
Martins M, Henriques M, Azeredo
J, Rocha SM, Coimbra MA et al. (2007) Morphogenesis control in Candida albicans and
Candida dubliniensis through signaling molecules produced by planktonic and
biofilm cells. Eukaryot Cell 6: 2429-2436. [Crossref]
4.
Webb B, Thomas CJ, Willcox MD, Harty DW, Knox KW (1998)
Candida-associated denture stomatitis. Aetiology and management: a review. Part
3. Treatment of oral candidosis. Aust Dent J 43: 244-249. [Crossref]
5.
Marsh P, Martin M, Lewis M, Williams D (2009) Oral microbiology, 5th
Edition. Elsevier Health Sci.
6.
Wilson J (1998) The aetiology, diagnosis and management of denture
stomatitis. Br Dent J 185: 380-384. [Crossref]
7.
Coco BJ, Bagg J, Cross LJ, Jose A, Cross J et al. (2008) Mixed Candida
albicans and Candida glabrata populations associated with the pathogenesis of
denture stomatitis. Oral Microbiol Immunol 23: 377-383. [Crossref]
8.
Marsh PD (2003) Dental plaque as a microbial biofilm. Caries Res
38: 204-211. [Crossref]
9.
Samaranayake L (2007) Essential Microbiology for Dentistry: 3rd Edition. Elsevier
Health Sci.
10.
Hamada S, Koga T, Ooshima T (1984) Virulence factors of Streptococcus
mutans and dental caries prevention. J Dent Res 63: 407-411. [Crossref]
11.
Hojo K, Nagaoka S, Ohshima T, Maeda N (2009) Bacterial interactions in
dental biofilm development. J Dent Res 88: 982-990. [Crossref]
12.
Sedlacek MJ, Walker C (2007) Antibiotic resistance in an in vitro
subgingival biofilm model. Oral Microbiol Immunol 22: 333-339. [Crossref]
13.
Welin Neilands J, Svensäter G (2007) Acid tolerance of biofilm cells of
Streptococcus mutans. Appl Environ Microbiol 73: 5633-5638. [Crossref]
14.
Marsh PD (1994) Microbial ecology of dental plaque and its significance
in health and disease. Adv Dent Res 8: 263-271. [Crossref]
15.
Chapman B (1980) Glow Discharge Processes: Sputtering and Plasma Etching.
Wiley.
16.
Laroussi M (2005) Low Temperature Plasma‐Based Sterilization: Overview
and State‐of‐the‐Art. Plasma Process Polym 2: 391-400.
17.
Moreau M, Orange N, Feuilloley MGJ (2008) Non-thermal plasma
technologies: new tools for bio-decontamination. Biotechnol Adv 26:
610-617. [Crossref]
18.
Rossi F, Kylián O, Hasiwa M (2006) Decontamination of Surfaces by Low
Pressure Plasma Discharges. Plasma Process Polym 3: 431-442.
19.
Laroussi M (2002) Nonthermal decontamination of biological media by
atmospheric-pressure plasmas: review, analysis, and prospects. IEEE Transact
Plasma Sci 30: 1409-1415.
20.
Yu QS, Li H, Ritts AC, Yang B, Chen M et al. (2012) Non-thermal
Atmospheric Plasma Treatment for Deactivation of Oral Bacteria and Improvement
of Dental Composite Restoration. Plasma Bio-Decontaminat Med Food 215-228.
21.
Yang B, Chen J, Yu Q, Li H, Lin M et al. (2011) Oral bacterial
deactivation using a low-temperature atmospheric argon plasma brush. J Dent
39: 48-56. [Crossref]
22.
Huang C, Yu Q, Hsieh FH, Duan Y (2007) Bacterial deactivation using a low
temperature argon atmospheric plasma brush with oxygen addition. Plasma
Proces Polym 4: 77-87.
23.
Dong X, Ritts AC, Staller C, Yu Q, Chen M et al. (2013) Evaluation of
plasma treatment effects on improving adhesive-dentin bonding by using the same
tooth controls and varying cross-sectional surface areas. Eur J Oral Sci 121:
355-362. [Crossref]
24.
Ritts AC, Li H, Yu Q, Xu C, Yao X et al. (2010) Dentin surface treatment
using a non-thermal argon plasma brush for interfacial bonding improvement in
composite restoration. Eur J Oral Sci 118: 510-516. [Crossref]
25.
Imolam, Judit P, Alpar S, Dan AS (2013) Deactivation of Streptococcus
mutans biofilms on a tooth surface using He dielectric barrier discharge at
atmospheric pressure. Plasma Sci Technol 15: 535.
26.
Huang WK, Weng CC, Liao JD, Wang YC, Chuang SF (2013)
Capillary-tube-based micro-plasma system for disinfecting dental biofilm. Int
J Radiat Biol 89: 364-370. [Crossref]
27.
Lu X, Xiong Z, Zhao F, Xian Y, Xiong Q et al. (2009) A simple atmospheric
pressure room-temperature air plasma needle device for biomedical applications.
Appl Phys Lett 95: 181501.
28.
Sladek REJ, Filoche SK, Sissons CH, Stoffels E (2007) Treatment of
Streptococcus mutans biofilms with a nonthermal atmospheric plasma. Lett
Appl Microbiol 45: 318-323.
29.
Hong Q, Dong X, Chen M, Xu Y, Sun H et al. (2016) Disinfection of
Streptococcus mutans biofilm by a non-thermal atmospheric plasma brush. Japan
J Appl Phys 55: 07LG02.
30.
Berney M, Hammes F, Bosshard F, Weilenmann HU, Egli T (2007) Assessment
and interpretation of bacterial viability by using the LIVE/DEAD BacLight Kit
in combination with flow cytometry. Appl Environ Microbiol 73:
3283-3290. [Crossref]
31.
Neville BW, Bouquot JE, Neville B, Allen CM (2002) Oral &
Maxillofacial Pathology, 2nd Edition.
32.
Shapiro S, Giertsen E, Guggenheim B (2002) An in vitro oral biofilm model
for comparing the efficacy of antimicrobial mouthrinses. Caries Res 36:
93-100. [Crossref]
33.
Lamfon H, Porter SR, McCullough M, Pratten J (2004) Susceptibility of
Candida albicans biofilms grown in a constant depth film fermentor to
chlorhexidine, fluconazole and miconazole: a longitudinal study. J
Antimicrob Chemother 53: 383-385. [Crossref]
34.
LaFleur MD, Kumamoto CA, Lewis K (2006) Candida albicans biofilms produce
antifungal-tolerant persister cells. Antimicrob Agents Chemother 50:
3839-3846. [Crossref]
35.
Koban I, Matthes R, Hübner NO, Welk A, Meisel P et al. (2010) Treatment
of Candida albicans biofilms with low-temperature plasma induced by dielectric
barrier discharge and atmospheric pressure plasma jet. New J Phys 12:
073039.
36.
Gaunt LF, Beggs CB, Georghiou GE (2006) Produced by Gas-Discharge
Nonthermal Plasma at Atmospheric Pressure: A Review. IEEE Transact Plasma
Sci 34: 1257-1269.
37.
Park GY, Park SJ, Choi MY, Koo IG, Byun JH et al. (2012)
Atmospheric-pressure plasma sources for biomedical applications. Plasma
Sources Sci Technol 21: 043001.
38.
Yu Q, Huang C, Hsieh FH, Huff H, Duan Y (2007) Bacterial inactivation
using a low-temperature atmospheric plasma brush sustained with argon gas. J
Biomed Mater Res B Appl Biomater 80: 211-219. [Crossref]
39.
Koban I, Holtfreter B, Hübner NO, Matthes R, Sietmann R et al. (2011)
Antimicrobial efficacy of non-thermal plasma in comparison to chlorhexidine
against dental biofilms on titanium discs in vitro - proof of principle
experiment. J Clin Periodontol 38: 956-965. [Crossref]
40.
Jones CG (1997) Chlorhexidine: is it still the gold standard? Periodontol
2000 15: 55-62. [Crossref]
41.
Jenkins S, Addy M, Wade W (1988) The mechanism of action of
chlorhexidine. A study of plaque growth on enamel inserts in vivo. J Clin
Periodontol 15: 415-424. [Crossref]
42.
Barrett Bee K, Newboult L, Edwards S (1994) The membrane destabilising
action of the antibacterial agent chlorhexidine. FEMS Microbiol Lett
119: 249-253. [Crossref]
43.
Güçeri S, Fridman A, Gibson K, Haas C (2008) Plasma Assisted
Decontamination of Biological and Chemical Agents. Springer.
44.
De Geyter N, Morent R (2012) Nonthermal plasma sterilization of living
and nonliving surfaces. Annu Rev Biomed Eng 14: 255-274. [Crossref]
45.
Filoche SK, Sissons CH, Sladek REJ, Stoffels E (2008) Cold Plasma
Treatment of In Vitro Dental Plaque. Plasma Assis Decontaminat Biol Chem
Agent 161-172.
46. Lerouge S, Wertheimer MR, Marchand R, Tabrizian M, Yahia L (2000) Effect
of gas composition on spore mortality and etching during low-pressure plasma
sterilization. J Biomed Mater Res 51: 128-135. [Crossref]
