Nocardial Vertebral Osteomyelitis and Epidural Abscess of Thoracic Spine in an Immunocompromised Patient
A B S T R A C T
Vertebral osteomyelitis and epidural abscess caused by Nocardia brasiliensis is very rare. This report states the successful treatment of Nocardial vertebral osteomyelitis and epidural abscess of the thoracic spine. A 56-year-old Thai female presented with pulmonary nocardiosis and sudden paraplegia. Her underlying disease was Evans syndrome (immunocompromised host). Her physical examination and investigation showed vertebral osteomyelitis and a spinal epidural abscess, which was compressing her spinal cord (T4-T7). Culture pus and tissue specimens from the epidural spine were reported as Nocardia brasiliensis. The patient underwent standard treatment for vertebral osteomyelitis and epidural abscess by surgical decompression, debridement and prolonged antibiotic therapy with intravenous Bactrim (trimethoprim-sulfamethoxazole (TMP-SMX)) for a total of 2 weeks, and then continued with oral Bactrim for 12 months. The patient’s neurological status had significantly improved at a 12-month follow-up. Nocardia brasiliensis osteomyelitis and epidural abscess are very challenging in their management. However, early investigation for diagnosis, followed by medical and surgical treatment they can have a successful outcome.
Keywords
Nocardia brasiliensis, vertebral osteomyelitis, epidural abscess, thoracic spine
Introduction
The most common organism causing vertebral osteomyelitis is Staphylococcus aureus that results from hematogenous spread [1]. Nocardia species are included in the actinomycetes group. Nocardial’s saprophyte is considered to be a slow-growing organism and was described for the first time, approximately 65 years ago [2, 3]. Nocardia brasiliensis very rarely cause vertebral osteomyelitis with an epidural abscess. However, there have been a few case reports of Nocardia infection in immunocompromised patients [3, 4]. The standard treatment for vertebral osteomyelitis and epidural abscess is surgical decompression, debridement and prolonged antibiotic therapy.
Case Report
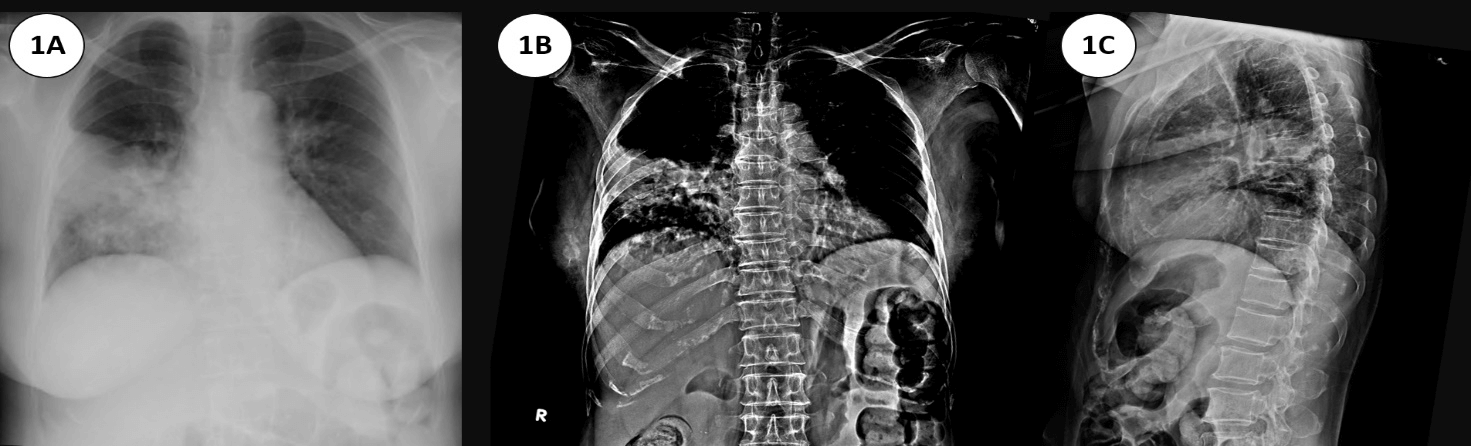
A 56-year-old female was diagnosed with Evans syndrome and was on a high dose of prednisolone for 4 years. At this visit, she presented with a cough and had had dyspnea for 3 weeks. Her chest X-ray showed a heterogeneous wedge-shaped opacity, at the right middle lung field, with a size of about 4.1x7.1 cm (Figure 1A) with adjacent pleural thickening. She was diagnosed with pneumonia, then partially treated with antibiotics but did not recover at the local hospital. She was taken to Songklanagarind Hospital, where her sputum was revaluation with direct gram smear and modified Kinyoun acid-fast stain. The slide showed filamentous branching-beads like an organism coupled with irregular staining that was compatible with pulmonary nocardiosis. The patient was started on intravenous Bactrim (15mg/kg/day of trimethoprim-sulfamethoxazole (TMP-SMX)).
However, her symptoms suddenly developed to acute paraplegia. Physical examination showed loss of motor power in the lower extremity and loss of sensation below the T7 level. Rectal examination showed loss of sphincter tone, no melena and no mass. The plain film of the thoracic spine was unremarkable in both anteroposterior (AP) (Figure 1B) and lateral views (Figure 1C).
Figure 1: Wedge-shaped opacity at right middle lung field; 1A) suspected lung atelectasis or pneumonia. 1B) The x-ray films: anteroposterior (AP) view and 1C) lateral view of the thoracic spine.
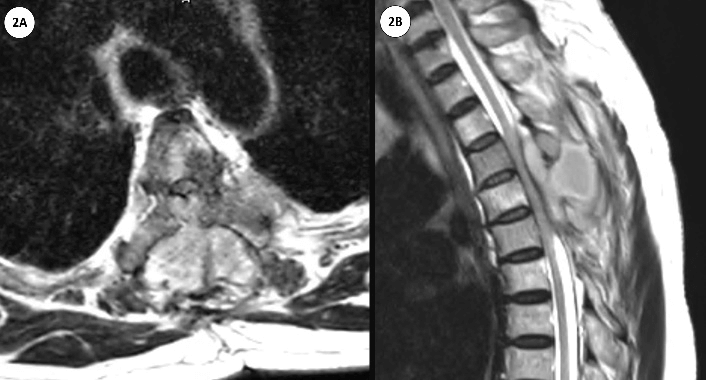
An MRI of the spine showed a few rim enhanced lesions within the deep subcutaneous tissue of both sides of the back, about 1-3 cm wide, which indicated likely evidence of her known, underlying disseminated nocardiasis. Deeper extension, to involve the left paravertebral soft tissue, transverse process and epidural extension compressing upon the spinal cord within a narrow central spinal canal at T5 level were noted. Additionally, Superior extension involving the spinal canal at T4, down to T7 level are also detected. Mild to moderate degree narrowing of the central spinal canal at C4 down to C7 levels was also seen, whilst the conus medullaris at the T12 level appeared unremarkable. Consolidation pneumonia of partly seen RML and RLL, with multiple scatter infiltrations, in the rest of both lungs were also most likely due to disseminated nocardiasis. A small amount of bilateral pleural effusions was seen. The MRI was disseminated in nocardiasis, involving the lungs, subcutaneous tissue with left paraspinal and epidural extension, resulting in spinal cord compression from T4-T7 (Figure 2).
Figure 2: 2A) Axial T2-weighted magnetic resonance imaging (MRI) scan; depicting spinal epidural abscess compressing the cervical cord posteriorly at T4. 2B) Sagittal T2-weighted MRI scan; demonstrating spinal epidural abscess extending down to T7.
The patient was put under general anaesthesia in the prone position, wherein, the authors then performed a posterior approach to the upper thoracic area. Posterior decompression, debridement and laminectomy on T4 to T7 was then performed. The surgeon found 20 ml of pus from the epidural area, so both tissue and pus samples were sent for culture diagnosis. Direct Gram smear and modified Kinyoun acid-fast staining revealed numerous branching filamentous bacillus. The culture pus and tissue specimens intraoperative reported "Nocardia brasiliensis". For this, the patient was continuously treated by intravenous Bactrim, for a total of 2 weeks, then continued oral Bactrim for 12 months. The patient’s neurological status significantly improved at a 12-month follow-up.
Discussion
Nocardia brasiliensis is an aerobic actinomycetes, and it is an acid-fast positive, branching filamentous bacillus. Although commonly found in water, soil, and decaying organic matter, Nocardia spp. are not common in immunocompromised patients [5]. Nocardia infections in immunocompromised patients enables the involvement of the pulmonary parenchyma, the skin and subsequent development of brain abscesses, epidural abscess and vertebral osteomyelitis [3, 6]. Nocardia is a slow-growing organism, which takes 5-21 days to exhibit growth; therefore, it is important not to discard the cultures early if nocardiosis is suspected [3]. Evans syndrome is an autoimmune disease, in which simultaneous or sequential direct Coombs-positive autoimmune hemolytic anemia (AIHA) in conjunction with immune-mediated thrombocytopenia can occur, with no known underlying etiology [7]. Patients also receive high steroid doses to combat Evans syndrome, making for an immunocompromised host, so the infectious agent was subacute process with fungal and bacterial infection.
The immunocompromised host may have had a mixed infection, so the empiric antibiotic was useful. The other differential diagnosis is malignancy; therefore, chest and spine imaging should be performed. The lab should also investigate a septic workup for bacterial and fungus. The time of starting an empiric antibiotic to cover nosocomial infections and to deescalate is very important. Biopsy of soft tissue for pathological diagnosis and an MRI of the spine to evaluate cord lesions is the standard protocol, with the standard treatment for vertebral osteomyelitis and epidural abscess being surgical posterior decompression and debridement for elucidating the source of infection. The treatment of choice for the infection is most commonly Bactrim (trimethoprim-sulfamethoxazole (TMP-SMX)) [8]. The recommendation of TMP-SMX, with higher daily doses, can also be considered: 15 mg/kg TMP and 75 mg/kg SMX. After 3-6 weeks, the dosage can be reduced and switched to the oral form [9]. In previous reviews, prolonged antibiotic administration for immunocompromised patients is preferred for up to 12 months [1-9]. Nocardia brasiliensis osteomyelitis and epidural abscess are very challenging in their management and require early investigation for diagnosis, followed by medical and surgical treatment for a successful outcome.
Funding
None.
Ethical Approval
The patient provided written informed consent to share his case details, and any accompanying images published. Prince of Songkla University Institutional Review Board (IRB number REC 63-176-11-4), Faculty of Medicine, Songklanagarind Hospital and Prince of Songkla University provided its approval to publish the case details. The patient’s personal information remains confidential. There was no cost, payment made or harm to the patient as a result of this study.
Consent
Written informed consent was obtained from the patient for publication of this case report, and the accompanying images.
Guarantor
The author has read and approved the manuscript and accepts full responsibility for the work.
Conflicts of Interest
None.
Acknowledgment
The authors wish to thank Andrew Jonathan Tait, from the International Affairs Department of Prince of Songkla University, for his assistance in proofreading the English of this report.
Article Info
Article Type
Case ReportPublication history
Received: Wed 17, Jun 2020Accepted: Thu 02, Jul 2020
Published: Wed 08, Jul 2020
Copyright
© 2023 Wongthawat Liawrungrueang. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.AJSCR.2020.03.02
Author Info
Piyawat Bintachitt Rattanaporn Chamnan Weera Chaiyamongkol Wongthawat Liawrungrueang
Corresponding Author
Wongthawat LiawrungrueangDepartment of Orthopedics, Faculty of Medicine, Prince of Songkla University, Songkhla, Thailand
Figures & Tables
References
- Zimmerli W (2010) Clinical Practice. Vertebral Osteomyelitis. N Engl J Med 362: 1022-1029. [Crossref]
- Minero MV, Marín M, Cercenado E, Rabadán PM, Bouza E et al. (2009) Nocardiosis at the turn of the century. Medicine (Baltimore) 88: 250-261. [Crossref]
- Johnson P, Ammar H (2013) Nocardia brasiliensis vertebral osteomyelitis and epidural abscess. BMJ Case Rep 2013: bcr2012008400. [Crossref]
- Boulios EA, Konstantopoulou PP, Bailas GD, Klagkou AK, Tseliou GC et al. (2017) Nocardia osteomyelitis in an immunosuppressed patient. J Frailty Sarcopenia Falls 2: 88-91. [Crossref]
- Muñoz J, Mirelis B, Aragón LM, Gutiérrez N, Sánchez F et al. (2007) Clinical and microbiological features of nocardiosis 1997-2003. J Med Microbiol 56: 545-550. [Crossref]
- Trujillo DE, Ortiz S, Pérez O, Cortés CA, Carrillo JA (2020) Multiple brain abscesses due to Nocardia spp. in an immunocompetent patient. Biomedica 40: 27-33. [Crossref]
- Shaikh H, Mewawalla P (2020) Evans Syndrome. Treasure Island (FL): StatPearls Publishing. StatPearls [Internet].
- Tan YE, Chen SC, Halliday CL (2020) Antimicrobial susceptibility profiles and species distribution of medically relevant Nocardia species: Results from a large tertiary laboratory in Australia. J Glob Antimicrob Resist 20: 110-117. [Crossref]
- Agterof MJ, van der Bruggen T, Tersmette M, ter Borg EJ, van den Bosch JMM et al. (2007) Nocardiosis: a case series and a mini review of clinical and microbiological features. Neth J Med 65: 199-202. [Crossref]
