Gross Total Endoscopic Resection of a Left Lateral Ventricle Central Neurocytoma in an 18-Year-Old Filipino Male: A Case Report
A B S T R A C T
Central neurocytomas are rare intraventricular tumors of neuronal origin that most commonly affect young adults. These tumors have a benign course and excellent prognosis after surgery. We present the first documented case of an 18-year-old healthy Filipino male presenting with new-onset generalized seizures and a heterogeneously enhancing left lateral ventricle tumor on imaging. The tumor was completely excised through endoscopic surgery, after which the patient made a complete recovery. Proper selection of cases, coupled with neurosurgeon expertise, solidifies endoscopic surgery as a safe and reasonable approach to achieve gross total resection of an intraventricular tumor.
Keywords
Central neurocytoma, neuroendoscopy, intraventricular tumor, endoscopic excision
Introduction
Central neurocytomas are uncommon tumors, accounting for less than 1% of all primary brain tumors [1-3]. The rarity of this tumor has impeded standardization of treatment. Prognosis is excellent after gross surgical resection, hence most studies recommend surgery as the primary modality [2-11]. Choosing the endoscopic approach over open surgery is dependent on tumor location and size [10, 12-14]. Most central neurocytomas are intraventricular, making total resection challenging. Neuroendoscopy may be employed to gain access to these deep-seated tumors. Tumors that are ideal for total endoscopic resection are singular lesions with soft consistency, small diameter (<2cm), mild-to-moderate vascularity, low histological grade and a completely intraventricular location accessible through a straight trajectory [10, 12]. Enlarged ventricles confer the advantage of a larger working space. In small or normal-sized ventricles, endoscopic surgery should be attempted only if the surgeon has substantial experience and proficiency [13]. For these cases, neuronavigation increases the accuracy of selecting an entry site [15]. The total endoscopic resection of an intraventricular central neurocytoma has not been reported in the Philippines and only a few cases have been documented in published studies [12-15]. A systematic literature review was performed using PubMed and Herdin (from 1980-2020). Search terms included central neurocytoma, neuroendoscopy, endoscopy, endoscopic surgery, excision and intraventricular tumors.
Case Presentation
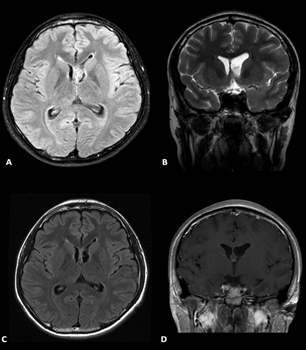
This is a case of an 18 year-old Filipino right-handed male with no history of epilepsy or childhood illnesses presenting with new-onset generalized tonic-clonic seizures triggered by strenuous activity, straining and lack of sleep. There were no headaches, vomiting episodes, behavioural or memory disturbances. On physical examination, he had intact memory, unremarkable fundoscopy and no focal neurologic deficits. An electroencephalogram showed frontal epileptiform discharges. Magnetic resonance imaging revealed a lobulated T1-hypointense, T2-hypointense, faintly enhancing tumor located on the medial wall of frontal horn of the left lateral ventricle, obstructing the foramen of Monro. The tumor measured 1.5×0.9×0.9 cm (AP×T×CC) (Figure 1).
Figure 1: A & B) Axial FLAIR and T2 coronal images show the tumor. C & D) Postoperative FLAIR and contrast images show gross total resection.
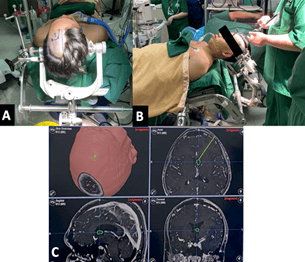
Our patient underwent gross total endoscopic resection of the left lateral ventricle tumor. The goals of the surgery were to relieve cerebrospinal fluid pathway obstruction, obtain a histopathologic diagnosis and accomplish maximal safe resection. The patient was positioned supine with head elevated at 30 degrees and neck at neutral position. The hair on the left side of the head was clipped. The head was then secured with a three-point fixation system. The left Kocher’s point located 3 cm lateral to the midline and 2 cm anterior to the coronal suture along the left midpupillary line was identified. Planned incision was plotted with a surgical marker. Registration with image-guidance system was conducted to plan the ideal trajectory to the ventricle (Figure 2).
Figure 2: Patient position and neuronavigation. A) Left Kocher’s point and planned incision outlined by a surgical marker. B & C) Registration process and trajectory planning with the image-guided system.
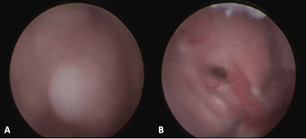
After the optimal point of entry was established, the operative field was sterilized with 95% ethyl alcohol. A relaxed L-shaped incision was created on the left frontal area. The scalp flap was retracted anterolaterally. A frontal precoronal Burr hole followed by a cruciate durotomy large enough to accommodate the caliber of a rigid neuroendoscope (Aesculap® MINOP 0-degree ventricular endoscope) were created. The cortical entry point was located on the left middle frontal gyrus. The pia mater was coagulated and incised. Through an anterior transcortical approach, the ventriculostomy catheter was inserted 5 cm from the outer table of the calvarium, leading to the egress of clear cerebrospinal fluid. The catheter was rotated clockwise as it was being withdrawn to increase the caliber of the corticectomy. The rigid neuroendoscope was traversed through this corridor. Upon entry to the ventricle, the left thalamostriate vein, septal vein, choroid plexus leading to the foramen of Monro and the septum pellucidum were identified to establish orientation. The grayish-white, dome-shaped, smooth-walled, broad-based tumor arising from the septum pellucidum was seen obstructing the foramen of Monro (Figure 3). The neuroendoscope was advanced to the base of the tumor to delineate the tumor-ependyma interface.
Figure 3: Intraoperative images. A) The left lateral ventricle tumor. B) Post-excision of tumor with patent foramen of Monro, intact choroid plexus and vasculature.
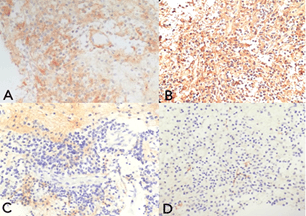
The tumor had a soft, feathery consistency and was excised in piecemeal fashion using biopsy forceps. Gross total excision of the tumor was performed. The tumor bed was coagulated after complete excision of the mass. No intraventricular hemorrhage was encountered. Cerebrospinal fluid egressed to the foramen of Monro. Continuous irrigation with warm lactated Ringer's solution for 15 minutes ensured hemostasis. Final inspection confirmed that the vasculature and surrounding structures were intact. A French 8 feeding tube was inserted 5 cm into the ventricle, anchored to the scalp using silk sutures and connected to an external drainage bag. Closure of the scalp was done. The total operative time was 3 hours. The external ventricular drain was removed on the 2nd post-operative day and the patient was discharged on the 5th post-operative day. Histopathologic studies and immunostaining confirmed the diagnosis of central neurocytoma, WHO Grade II (Figure 4). Repeat imaging done two weeks after surgery showed non-demonstration of the intraventricular mass and confirmed gross total resection (Figure 1). The patient was followed-up every 3 months and has been asymptomatic with no seizure recurrences. Postoperative radiation or chemotherapy was not pursued.
Figure 4: Microscopic images under low-power magnification show round monomorphic cells embedded in a fibrillary matrix. A) The tumor stained diffusely positive for synaptophysin, confirming glial and neuronal origin. B) Positive stain for GFAP confirms that the central neurocytoma originates from bipotential progenitor cells in the subependymal plate of the lateral ventricle. C) Negative for epithelial membrane antigen, ruling out ependymoma. D) Negative for neurofilament, ruling out neuroblastoma.
Discussion
Surgery is curative for central neurocytomas and prognosis is excellent after gross total resection, with a 99% survival rate [16, 17]. Since most central neurocytomas are located within the lateral ventricles and are attached to the septum pellucidum, a transcallosal or transcortical approach is commonly selected [2-5, 18]. Compared to classic open surgery, endoscopic surgery is minimally invasive, provides superior visualization of intraventricular anatomy and the interface between normal and tumoral tissue, avoids brain retraction and parenchymal injury, reduces blood loss and leads to shorter operative times, eventually leading to rapid recovery and shorter hospital stay [12-15, 19]. An additional advantage is the capability to treat altered cerebrospinal fluid circulation [13, 19].
Our patient was an ideal candidate for endoscopic surgery since his tumor was solitary, purely intraventricular, measured 1.5 cm in widest diameter and was accessible through a straight trajectory. Endoscopic surgery is not routinely recommended for patients who do not present with hydrocephalus because of difficulty in cannulation [12]. The ventricles of our patient were asymmetrical but not significantly dilated, hence neuronavigation was employed to establish a more accurate entry point to the ventricle. Through the planned trajectory (Figure 2), we encountered no difficulty with ventricular cannulation and obtained direct access to the frontal horn and the tumor.
Aside from direct visualization of the intraventricular anatomy, the rigid neuroendoscope also enabled the visualization of a plane between the tumor and the normal ependyma, facilitating safe maximal piecemeal resection with preservation of surrounding neurovascular structures. Known limitations of endoscopic surgery are restricted working space, inability to perform bimanual dissection, and possibility of profuse hemorrhage necessitating conversion to open surgery [12, 19]. However, with neurosurgeon expertise and advanced neuroendoscopic instruments, these limitations can be overcome and gross total endoscopic excision is attainable with low risk of morbidity. In this case, the tumor was soft, feathery, and easily grasped by biopsy forceps. Hemorrhage was not encountered due to cautious resection and continuous irrigation. Relief of CSF pathway obstruction was accomplished at the end of surgery. The patient had an unremarkable post-operative course and became seizure-free, demonstrating that neuroendoscopy is a safe and feasible approach to the total excision of an intraventricular central neurocytoma. Appropriate patient selection, neuronavigation and neurosurgeon expertise are essential to improve patient outcome and mitigate the risk of neurologic complications.
Conflicts of Interest
None.
Funding
None.
Acknowledgement
We would like to acknowledge the following for their invaluable contributions to this case report: Dr. Ibet Marie Y. Sih, Dr. Carlo G. Barredo and Dr. Erickson F. Torio for their unwavering mentorship and intellectual support, Dr. Emelia Katrina I. Cafugauan for co-managing the patient’s case, Dr. Rajiv Hans S. Menghrajani for editing the intraoperative photos, and Dr. Hazel Mae T. Panganiban for the providing the histopathologic slides.
Article Info
Article Type
Case ReportPublication history
Received: Thu 30, Sep 2021Accepted: Thu 21, Oct 2021
Published: Mon 07, Feb 2022
Copyright
© 2023 Charlene Mary Mercado. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2021.11.08
Author Info
Charlene Mary Mercado Jo Celine Leong Juan Manuel Mariano Carlo Barredo Manuel Mariano
Corresponding Author
Charlene Mary MercadoSt. Luke’s Medical Center, Quezon City, Philippines
Figures & Tables
References
1. Yang I, Ung N,
Chung LK, Nagasawa DT, Kimberly Thill et al. (2015) Clinical manifestations of
central neurocytoma. Neurosurg Clin N Am 26: 5-10. [Crossref]
2. Schmidt MH,
Gottfried ON, Koch CSV, Chang SM, McDermott MW (2004) Central neurocytoma: a
review. J Neurooncol 66: 377-384. [Crossref]
3. Dutta SW, Kaleem
TA, Muller DA, Peterson J, Harrell AC et al. (2018) Central neurocytoma:
Clinical characteristics, patterns of care, and survival. J Clin Neurosci
53: 106-111. [Crossref]
4. Choudhari KA,
Kaliaperumal C, Jain A, Sarkar C, Soo MYS et al. (2009) Central neurocytoma: A multi-disciplinary review. Br J
Neurosurgery 23: 585-595. [Crossref]
5. Chen H, Zhou R, Liu
J, Tang J et al. (2012) Central neurocytoma. J Clin Neurosci 19:
849-853. [Crossref]
6. Donoho D and Zada G
(2015) Imaging of central neurocytomas. Neurosurg Clin N Am 26: 11-19. [Crossref]
7. Hassoun J, Gambarelli D, Grisoli F, Pellet W, Salamon G et al. (1982) Central
neurocytoma. An electron-microscopic study of two cases. Acta Neuropathol
56: 151-156. [Crossref]
8. Agarwal A, Kanekar
S (2016) Intraventricular Tumors. Semin Ultrasound CT MR 37: 150-158. [Crossref]
9. Leenstra JL,
Rodriguez FJ, Frechette CM, Giannini C, Stafford SL et al. (2007) Central
neurocytoma: management recommendations based on a 35-year experience. Int J
Radiat Oncol Biol Phys 67: 1145-1154. [Crossref]
10. Ahmad F, Sandberg
DI (2010) Endoscopic management of intraventricular brain tumors in pediatric
patients: a review of indications, techniques, and outcomes. J Child Neurol
25: 359-367. [Crossref]
11. Karthigeyan M,
Gupta K, Salunke P (2017) Pediatric Central Neurocytoma: A Short Series With
Literature Review. J Child Neurol 32: 53-59. [Crossref]
12. Cappabianca P, Cinalli G, Gangemi M, Brunori A, Cavallo LM et al. (2008) Application
of neuroendoscopy to intraventricular lesions. Neurosurgery 62: 575-597.
[Crossref]
13. Russo N, Brunori A, Delitala A et al. (2015) Endoscopic Approaches to
Intraventricular Lesions. J Neurol Surg A Cent Eur Neurosurg 76:
353-360. [Crossref]
14. Cheng CH, Liu CL,
Chen CC, Lin SZ, Cho DY (2010) Single-port endoscopic removal of
intraventricular central neurocytoma. J Clin Neurosci 17: 1417-1420. [Crossref]
15. Chowdhry SA, Cohen
AR (2013) Intraventricular neuroendoscopy: complication avoidance and
management. World Neurosurg 79: S15.e1-S15.e10. [Crossref]
16. Lee SJ, Bui TT,
Chen CHJC, Lagman C, Chung LK et al. (2016) Central Neurocytoma: A Review of
Clinical Management and Histopathologic Features. Brain Tumor Res Treat
4: 49-57. [Crossref]
17. Rades D, Fehlauer F
(2002) Treatment options for central neurocytoma. Neurology 59:
1268-1270. [Crossref]
18. Chen CL, Shen CC, Wang J, Lu CH, Lee HT (2008). Central neurocytoma: A clinical, radiological and pathological study of nine cases. Clin Neurol Neurosurg 110: 129-136. [Crossref]
19. Kim MH (2017) Transcortical Endoscopic Surgery for Intraventricular Lesions. J Korean Neurosurg Soc 60: 327-334. [Crossref]
