Extremely Rapid Response to Pembrolizumab in a SMARCA4 Mutant PD-L1 Highly Expressive Advanced Lung Adenocarcinoma: A Case Report
A B S T R A C T
SMARCA4 mutant non-small cell lung cancer (SMARCA4m-NSCLC) has a poor prognosis owing to rapid growth. Effective treatments for SMARCA4m-NSCLC have not yet been established. Recently, many preclinical studies support the hypothesis that SMARCA4m-NSCLC may be vulnerable to immune checkpoint inhibitors. Here, we report a patient with programmed death-ligand1 (PD-L1) highly expressive SMARCA4m-NSCLC who showed an extremely rapid and long-term response to pembrolizumab. He was referred to our hospital for a mass of the right lung. Positron emission tomography-computed tomography showed right lung tumor, hilar, mediastinal and bone metastases. Pathological and immunohistochemical results showed it was a lung adenocarcinoma and revealed the tumor proportion score of PD-L1 was 80%. SMARCA4 and K-RAS genes were co-mutations. BRG1 protein expression was negative. Subsequently, pembrolizumab treatment as the first line of therapy was commenced for the patient. With only one dose, pembrolizumab significantly inhibited tumor growth and a partial response was obtained. To date, pembrolizumab treatment has been continued for about 29 months. Severe immune-related adverse events were not observed. Our case showed that an extremely rapid and long-term response can be achieved with pembrolizumab for PD-L1 highly expressive SMARCA4m-NSCLC. Immune checkpoint inhibitors treatment may be a promising strategy for PD-L1 positive SMARCA4m-NSCLC.
Keywords
Non-small cell lung cancer, SMARCA4 mutant, PD-L1, immune checkpoint inhibitors
Introduction
Over the past 20 years, NSCLC treatment has changed dramatically with the development of molecular profiling detection and targeted therapeutic agents [1]. EGFR somatic mutation and ALK, ROS1 rearrangements have been validated as “driver” genes in NSCLC and corresponding target drugs have been established [2-4]. SMARCA4 is the most frequent mutated member of the SWI/SNF complex, with mutations happening in 10% of NSCLC patients [5]. The prognosis for SMARCA4m-NSCLC is relatively poor, with median overall survival (OS) around seven months because no effective anti-SMARCA4 inhibitors are currently used in clinical practice. Recently, a study reported that immune checkpoint inhibitors (ICIs) as a treatment method in SMARCA4m-NSCLC might improve OS [6]. These results indicated that ICIs might be effective and could obtain clinical benefits for patients with SMARCA4m-NSCLC. However, the mechanisms of ICIs anti-tumor activities have not been fully elucidated. In our case, we reported an extremely rapid and long-term response to pembrolizumab in a SMARCA4 mutant PD-L1 highly expressive advanced lung adenocarcinoma.
Case Report
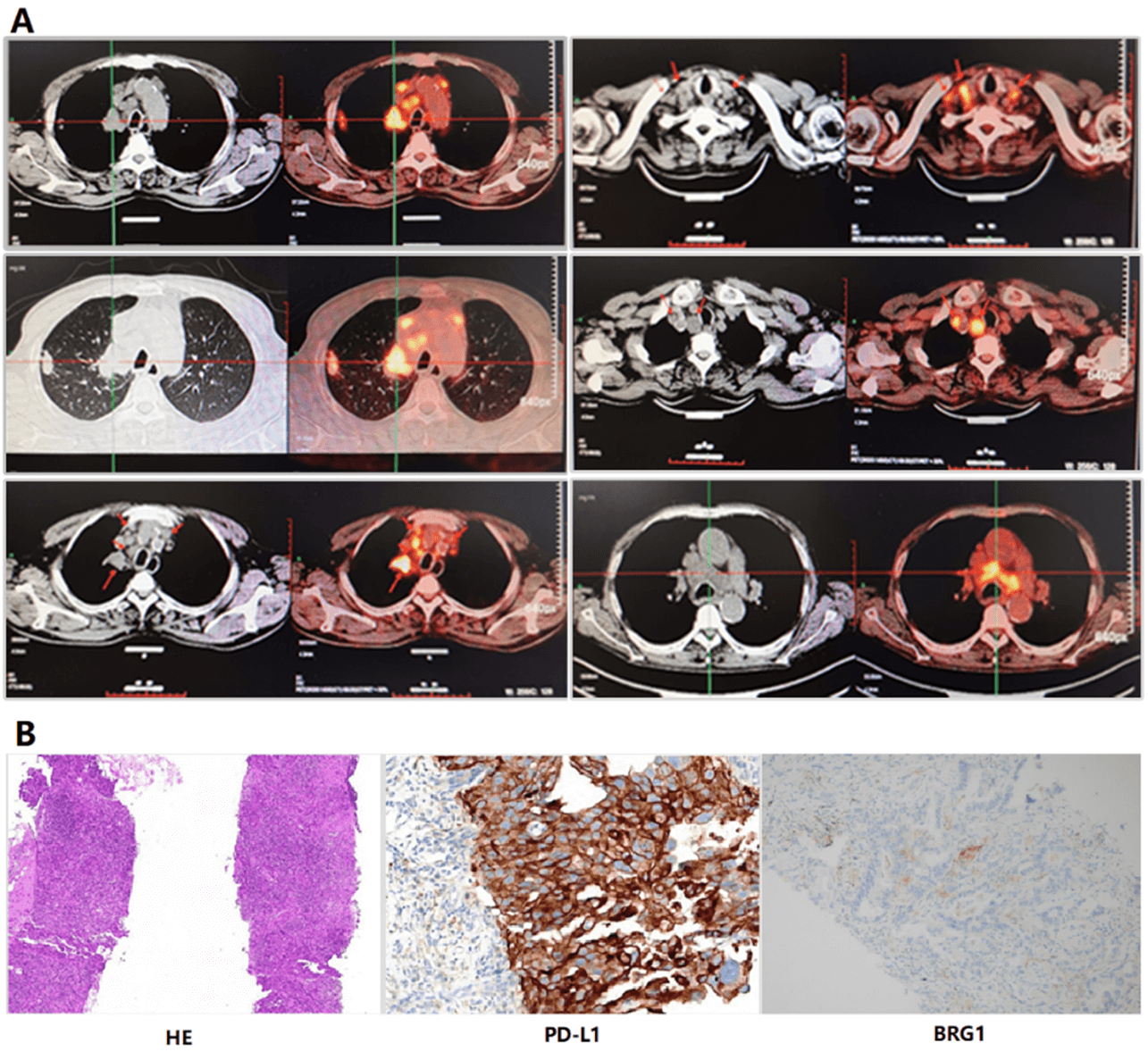
A 67-year-old man was referred to our hospital for a mass of the right upper lung. Positron emission tomography-computed tomography (PET-CT) showed right upper lung tumor, hilar, mediastinal, and bilateral supraclavicular lymph node metastasis and bone metastases (Figure 1A). Transbronchial biopsy and right supraclavicular lymph node biopsy were performed, and he was diagnosed with lung adenocarcinoma in April 2019. Five days after the biopsy, his voice became hoarse. We found that the mediastinal lymph node had invaded the right recurrent laryngeal nerve resulting in the abnormality (Figure 1A). Pathological and immunohistochemical results revealed that the tumor proportion score of PD-L1 was 80% (Figure 1B). Next-generation sequencing was also performed. The results showed that no somatic mutation or rearrangements were found for EGFR, ALK, ROS1 and C-MET genes. SMARCA4 and K-RAS co-mutations were found. BRG1 protein expression was negative (Figure 1B). Taken together, the patient was diagnosed with clinical stage IV, PD-L1 highly expressive SMARCA4m-NSCLC.
Figure 1: The image of positron emission tomography-computed tomography, pathology and immunohistochemistry. A) The image of positron emission tomography-computed tomography. Arrows indicate tumors. B) Hematoxylin and eosin (H&E) staining (100×magnification); Immunohistochemistry stained with PD-L1 antibody (SP263) in a fresh tissue of right supraclavicular lymph node before treatment, exhibiting strong membrane staining in 80% of tumor cells (400×magnification). Immunohistochemistry stained with BRG1 primary antibody.
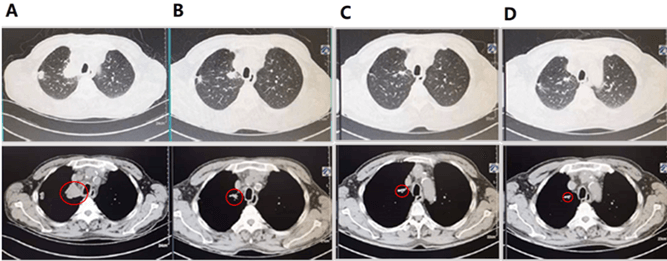
Subsequently, we treated the patient with pembrolizumab as the first line of therapy because of PD-L1 high expression on April 26, 2019. Before treatment, a CT scan was performed (Figure 2A). After one pembrolizumab infusion (200mg/3 weeks), the patient’s voice sound improved, suggesting pembrolizumab may be effective and inhibited tumor growth. After infusion twice, a CT scan was performed. As shown in (Figure 2B), the size of the tumor significantly decreased compared to pre-treatment, resulting in a partial response. After 4 cycles of pembrolizumab treatment, the tumor was getting smaller (Figure 2C).
To date, pembrolizumab treatment for the patient has been continued for about 29 months. He had no obvious clinical symptoms and no severe adverse events. After 41 cycles of pembrolizumab treatment, a CT scan demonstrated that the size of the tumor was tiny compared to pre-treatment, indicating that pembrolizumab was sustained effective (Figure 2D).
Figure 2: The image pre-treatment and after-treatment CT scan. A) Pre-treatment CT scan; B) CT scan after one cycle of pembrolizumab; C) CT scan after two cycles of pembrolizumab; D) CT scan after forty-one cycles of pembrolizumab. Red circles indicate tumors.
Discussion
In our case, we described a PD-L1 highly expressive SMARCA4m-NSCLC patient who obtained an extremely rapid and long-term response for pembrolizumab. To our knowledge, this is the first case of a SMARCA4m-NSCLC patient showing high expression of PD-L1 in the tumor. The survival outcome of SMARCA4m-NSCLC patients was significantly worse. Therefore, this report is important, indicating that PD-1 inhibitors may be effective for SMARCA4m-NSCLC. Recently, several reports showed that ICIs were effective and safe for patients with SMARCA4m-tumors. For example, Henon C et al. reported that ICIs treatment of SMARCA4 deficient thoracic malignant rhabdoid-like tumor obtained long-lasting major response. In this report, TMB is remarkably low, PD-L1 expression was negative in tumor cells and immune cell infiltrates were rare [7]. Jelinic et al. also showed that PD-1 inhibitor was effective in SMARCA4-deficient small-cell carcinoma of the ovary [8]. Therefore, we have a hypothesis that whether SMARCA4m could be a predictor for ICIs response in NSCLC. Notably, there was one previous report that showed that PD-1 inhibitor was effective for a SMARCA4 deficient NSCLC patient with a high TMB and PD-L1 not highly expressed [9]. However, there was another study that reported hyper-progressive disease was obtained after immune checkpoint inhibitor treatment in SMARCA4 deficient SCLC [10].
In all, it remains unproven that whether the identification of SMARCA4m is enough to predict ICIs response. Nowadays, ICIs have been used as standard therapy in a majority of NSCLC patients. However, according to different molecular subtypes, how to select appropriate patients for ICIs treatment will be critical. To date, PD-L1, microsatellite instability and tumor mutation burden (TMB) have been recognized as a predictor of ICIs response [11, 12]. SMARCA4m is reportedly positively associated with PD-L1 expression [13]. In our case, we believe that SMARCA4 and K-RAS co-mutations might have caused PD-L1 increase. Therefore, such a good response is mainly due to the combined effect of PD- L1 high expression and SMARCA4 mutation.
Taken together, this is the first report showing that the patient with PD-L1 highly expressive SMARCA4m-NSCLC has an extremely rapid and long-term response to pembrolizumab. In terms of PD-L1 positive SMARCA4m-NSCLC, ICIs may be an effective therapeutic option. Given the limitations of individual case reports, however, the efficacy of PD-1 inhibitor needs to be further verified in a larger patient cohort with SMARCA4m-NSCLC.
Acknowledgement
The authors would like to thank the patient for the publication of clinical data and images.
Funding
This work was supported by the Health Bureau of Zhejiang Province (2016KYA023, 2016ZA025, 2018KY466), the Natural Science Foundation of Zhejiang Province (LQ18H160025).
Conflicts of Interest
None.
Article Info
Article Type
Case ReportPublication history
Received: Tue 31, Aug 2021Accepted: Wed 20, Oct 2021
Published: Fri 05, Nov 2021
Copyright
© 2023 Lili Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2021.11.07
Author Info
Xiuming Zhu Hongming Pan Lili Liu
Corresponding Author
Lili LiuDepartment of Medical Oncology, Sir Run Run Shaw Hospital, College of Medicine, Zhejiang University, Hangzhou, Zhejiang, China
Figures & Tables
References
1. Herbst RS, Morgensztern D, Boshoff C (2018)
The biology and management of non-small cell lung
cancer. Nature 553: 446-454.
[Crossref]
2. Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA et
al. (2004) Activating mutations in the epidermal growth factor receptor
underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med
350: 2129-2139. [Crossref]
3. Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y et al.
(2007) Identification of the
transforming EML4-ALK fusion
gene in non-small-cell lung cancer.
Nature 448: 561-566. [Crossref]
4. Kumarakulasinghe NB, van Zanwijk N, Soo RA
(2015) Molecular targeted
therapy in the treatment
of advanced stage non-small cell lung cancer
(NSCLC). Respirology
20: 370-378. [Crossref]
5. Reisman DN, Sciarrotta J, Wang W, Funkhouser WK, Weissman BE (2003)
Loss of BRG1/BRM in human lung cancer cell lines and primary lung cancers: correlation with poor prognosis. Cancer Res 63:
560-566. [Crossref]
6. Schoenfeld AJ, Bandlamudi C, Lavery JA, Montecalvo J, Namakydoust A et al.
(2020) The Genomic Landscape
of SMARCA4 Alterations and Associations with Outcomes in Patients with Lung
Cancer. Clin Cancer
Res 26: 5701-5708. [Crossref]
7. Henon C, Blay JY, Massard C, Mir O, Bahleda R et al. (2019)
Long lasting major response
to pembrolizumab in a thoracic
malignant rhabdoid-like SMARCA4-deficient tumor. Ann Oncol 30: 1401-1403. [Crossref]
8. Jelinic P, Ricca J, Van Oudenhove E, Olvera N, Merghoub T et al. (2018) Immune-Active
Microenvironment in Small Cell Carcinoma
of the Ovary, Hypercalcemic Type: Rationale for Immune
Checkpoint Blockade. J Natl Cancer
Inst 110: 787-790. [Crossref]
9. Arias Ron D, Labandeira CM, Areses Manrique
MC, Sampedro Domarco
P, Abdulkader I et al.
(2019) Dramatic Response
of Leptomeningeal Carcinomatosis to Nivolumab in PD-L1
Highly Expressive Non-small Cell Lung Cancer:
A Case Report. Front Oncol 9: 819. [Crossref]
10. Chiba Y, Kawanami T, Yamasaki K, Uchimura K, Matsuyama A et al. (2020) Hyper-progressive disease after immune checkpoint inhibitor
in SMARCA4-deficient small-cell lung carcinoma. Respirol Case Rep 8: e00667. [Crossref]
11. Shukuya T, Carbone DP (2016)
Predictive Markers for the Efficacy
of Anti-PD-1/PD-L1 Antibodies in Lung Cancer. J Thorac Oncol 11: 976-988. [Crossref]
12. Georgiadis A, Durham JN, Keefer LA, Bartlett BR, Zielonka M et al. (2019) Noninvasive Detection of Microsatellite Instability and High Tumor Mutation Burden in Cancer Patients Treated with PD-1 Blockade. Clin Cancer Res 25: 7024-7034. [Crossref]
13. Naito T, Udagawa H, Umemura S, Sakai T, Zenke Y et al. (2019) Non-small cell lung cancer with loss of expression of the SWI/SNF complex is associated with aggressive clinicopathological features, PD-L1-positive status, and high tumor mutation burden. Lung Cancer 138: 35-42. [Crossref]
