Correlation Between Vitamin D and Degenerative Joint Disorders: Review and Meta-Analysis
A B S T R A C T
Objective: Evaluate the evidence of the clinical correlation between vitamin D and symptomatic degenerative joint disease.
Methods: A systematic search and meta-analysis was conducted of randomized controlled studies (RCTs) published in between January 1st, 2010, and March 30th, 2020 on five different databases. The study population consisted of adult patients with symptomatic knee osteoarthritis; the intervention was vitamin D; the comparison was placebo, and the outcomes included the Western Ontario and McMaster Universities Arthritis Index (WOMAC) (pain, function, stiffness), tibial cartilage volume, synovial tissue volume (STV), subchondral bone marrow lesion (BML) volume, effusion-synovitis, serum vitamin D3 levels, serological inflammatory and metabolic biomarkers levels and adverse events.
Results: Nine RCTs involving 2,168 patients were included in this study. Pooled estimates suggested that vitamin D supplementation was associated with reduction in WOMAC pain (Std. Mean=1.08(0.90, 1.25); I2=99%; p=0.00001), function (Mean=1.1(0.92, 1.27); I2=99%; p=0.00001), stiffness (Std. mean= 0.72(0.54, 0.90); I2=98%; p=0.00001) and synovial effusion in the suprapatellar pouch numbers. There was no significant difference in tibial cartilage volume incidence (Std. Mean=0.26(0.44, 0.80); I2=99%; p=0.00001), STV, BML volume, inflammatory biomarkers and adverse events between the vitamin D and the placebo groups.
Conclusion: Vitamin D supplementation was effective in improving WOMAC pain and function in patients with knee OA and also improved serological vitamin D levels. However, it had no beneficial effect on structural cartilage change or inflammatory biomarker reduction. Therefore, there is currently a lack of evidence on vitamin D regimen dosage and length to obtain benefits in preventing knee disease progression.
Keywords
Vitamin D, osteoarthritis, degenerative joint disorders, meta-analysis
Introduction
Osteoarthritis (OA) is the most prevalent and disabling degenerative bone disorder, as reported by the Osteoarthritis Research Society International (OARSI) [1]. OA generates inflammation and changes in joint structures, the most affected being the articular cartilage, synovial membrane and subchondral bones [2, 3]. OA affects between 20 to 30% of the population, with women over 60 years of age being the most at risk [4, 5]. Although the estimated prevalence of OA varies between studies, there is consensus that it is likely to increase due to rising obesity rates and the aging of the population [2, 4, 5].
There are currently no medical treatments established to prevent the disease progression, only to control the symptomatology. Because the signs and symptoms associated with OA incur high costs on patients’ quality of life along with high financial burdens, recent research has focused on demonstrating feasible alternatives and benefits to be able to deal with this disorder [6]. Vitamin D is part of a promising therapeutic option, due to its participation in multiple biological functions. The most important and most studied role of vitamin D is related to bone metabolism, including joint and muscle structures, which can prevent and modify OA progression [7, 8].
The relationship between vitamin D supplementation and OA showed that vitamin D supplementation had effects in reducing pain and improving function versus the placebo group [9, 10]. Even when the results have been less than expected, this could be explained since vitamin D may require more time to exert its effects on OA progression, making longer studies necessary.
Wang et al. demonstrated a beneficial effect of vitamin D supplementation on effusion-synovitis, particularly in the suprapatellar region in knee OA patients with vitamin D deficiency. The synovitis volume increased 1.9 ml in the placebo group, and the size effect was small but statistically significant; vitamin D could thus be the key element in delaying knee OA progression. However, two randomized controlled trials (RCTs) reported contradictory results [11, 12]. Jin et al. did not find significant difference in their results on tibial cartilage measured by MRI or in the WOMAC knee pain score for two years. The study by Perry concludes that vitamin D supplementation does not appear to have an effect on synovitis volume or subchondral bone marrow lesion (BML) volume in patients with symptomatic knee OA. It is thus not possible to confirm the effect of vitamin D in patients with symptomatic OA [11, 12].
As the data is not decisive regarding the success of vitamin D supplementation on the symptoms and progression of degenerative joint disorders, the aim of this study is to undertake a systematic review and meta-analysis of RCTs to determine the efficacy of vitamin D supplementation in knee OA symptoms and progression, compared with placebo.
Methods
I Search Strategy and Study Selection
A systematic literature search was performed in 5 databases: PubMed, The Cochrane Library, Epistemonikos, EBSCO and Scopus, for articles published between January 1st, 2010, and March 30th, 2020. The search terms used were: (“vitamin D”[MeSH Terms] OR “vitamin d”[All Fields] OR “vitamin d deficiency”[MeSH Terms] OR “vitamin d deficiency”[All Fields], “vitamin d supplements”[MeSH Terms] OR “vitamin d supplements”[All Fields]) AND (“degenerative joint disorders”[Mesh Terms] OR “degenerative joint disorders”[All Fields], “osteoarthrosis”[MeSH Terms] OR “osteoarthrosis”[All Fields] OR “osteoarthritis”[Mesh Terms] OR “osteoarthritis”[All Fields]), creating different search strategies with language restriction (English and Spanish). The following inclusion criteria were used for study selection: i) study design: RCTs; ii) population: adult patients diagnosed with degenerative bone disorders; iii) intervention: vitamin D supplementation; iv) comparison: placebo; v) outcome measure: changes from baseline in vitamin D3 levels, changes in inflammatory and metabolic biomarkers, changes in Western Ontario and McMaster Universities Arthritis Index (WOMAC) pain, function and stiffness, changes in tibial cartilage volume (mm3), synovial tissue volume (STV), subchondral bone marrow lesions (BML) and adverse events. Studies were excluded if they were animal studies, or if they had human subjects who were obese, with autoimmune diseases or joint surgical indication.
II Data Collection and Analysis
Two researchers independently reviewed the databases’ (GD-CV) data using a standard form and a third reviewer solved discrepancies (GM). For data collection and weighting, a standardized collection and evaluation form was designed. Previously, the researchers were trained and then calibrated with a pilot test of 4 potentially selectable articles to homogenize criteria during the selection and evaluation process (Kappa: 0.73). The researchers were not blinded about the authors.
III Study Selection
We assessed articles by title and abstract. They were subsequently evaluated by full text; those that did not meet requirements with their corresponding justification were discarded. The collected data was organized using the diagram and flow items of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).
IV Bias Risk Assessment and Evidence Quality Grading
Each included article was assessed for bias risk according to the Cochrane collaboration tool Rob-2 using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions [13, 14]. The following biases were reviewed for each study: random sequence generation, allocation concealment, participant and personnel blinding, outcome assessment blinding, incomplete outcome data and sample size. The quality of evidence for outcome measures was evaluated using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach [15].
V Statistical Analysis
A descriptive statistical analysis of the populations studied, and their comparative results were performed and presented in tables. The data was processed to facilitate protocol preparation and systematic review (Cochrane Rev Man 5.3). To measure the treatment effect, the data was dichotomously pooled using risk ratios (RR) to describe effect size and size of continuous data using mean differences (MD).
Results
I Search Results
Studies’ eligibility for inclusion in meta-analysis (PRISMA) (Figure 1).
II Study Characteristics
These RCTs were published between 2013 and 2019. Of these studies, 4 were conducted in Australia, 2 in USA, 2 in UK and 1 in India [6, 7, 9, 10, 12, 16-19]. The daily dosage of vitamin D supplementation varied greatly between the included studies, ranging from 800 IU to 60,000 IU, while treatment duration varied between 3 to 36 months. Age, gender proportion, body mass index, and serum vitamin D3 levels were similar between the trial groups. The baseline characteristics of included studies are presented in (Table 1).
Figure 1: Eligibility of studies for inclusion in meta-analysis (PRISMA).
Table 1: Baseline characteristics of patients included in
the meta-analysis.
|
Study |
Country |
Treatment
regimen |
No.
of patients |
♀ Female/
♂ Male |
Age
(Mean, SD) |
BMI1 (Mean,
SD) |
Vitamin D3 (ng/ml) (Mean, SD) |
|
Sanghi 2013 |
India |
60.000 IU daily vitamin D3, Placebo. |
N=103 VD3=52 PL4=51
|
66♀ and 37♂ VD (30♀/21♂) PL (36♀/16♂) |
VD2: 53.24 (9.64) PL3: 53.00 (7.44) |
VD: 25.86 (2.46) PL: 25.65 (2.58) |
VD: 14.812
(3.012) PL: 15.008 (3.012) |
|
McAlindon
2013 |
USA |
2.000 IU daily vitamin
D3, Placebo. |
N=146 VD=73 PL=73
|
89♀
and 57♂ VD (49♀/24♂) PL (40♀/33♂) |
VD:
61.8 (7.7) PL:
63.0 (9.3) |
VD:
30.5 (5.0) PL:
30.8 (6.4) |
VD:
22.7 (11.4) PL:
21.9 (1.7) |
|
Jin 2016 |
Australia |
50.000 IU daily vitamin D3, Placebo. |
N=413 VD=209 PL=204
|
208♀ and 205♂ VD (106♀/103♂) PL (102♀/102♂) |
VD: 63.5 (6.9) PL: 62.9 (7.2) |
VD: 29.6 (5.4) PL: 29.6 (4.6) |
VD: 17.48 (4.72) PL: 17.52 (5.08) |
|
Arden
2016 |
UK |
800 IU daily vitamin D3, Placebo. |
N=474 VD=237 PL=
237
|
289♀
and 185♂ VD (144♀/93♂) PL (145♀/92♂) |
VD:
64 (8) PL:
64 (8) |
VD:
30 (5) PL:
29 (5) |
VD:
20.7 (8.9) PL:
20.7 (8.1) |
|
Zheng 2017 |
Australia |
50.000 IU daily
vitamin D3, Placebo. |
N=340 CI=46 FL=68 CS=226
|
160♀ and 180♂ CI (24♀ and 22♂) FL (38♀/30♂) CS (98♀/128♂) |
CI4:62.6 (8.0) F5: 62.9 (6.1) CS6: 63.5 (7.2) |
CI: 30.6 (4.6) F: 29.0 (4.5) CS: 29.4 (4.9) |
CI: ≤ 20 ng/ml F: > 20 ng/ml at either point CS: > 20 ng/ml at both months 3 and
24. |
|
Wang
2017 |
Australia |
50.000 IU daily vitamin D3, Placebo. |
N=413 VD=209 PL=204
|
208♀
and 205♂ VD (106♀/103♂) PL (102♀/102♂) |
VD:
63.55 (6.88) PL:
62.55 (7.22) |
VD:
29.57 (5.39) PL:
29.64 (4.62) |
VD:
17.496 (4.72) PL:
17.524 (5.06) |
|
Zheng 2018 |
Australia |
50.000 IU daily
vitamin D3, Placebo. |
N=200 VD=106 PL=94
|
107♀ and 93♂ VD (53♀/53♂) PL (54♀/40♂) |
VD: 63.3 (7.5) PL: 62.8 (7.3) |
VD: 29.4 (7.5) PL: 29.6 (4.0) |
VD: 17 (4.68) PL: 17.4 (5.04) |
|
Barker
2019 |
USA |
4.000 IU daily vitamin D3, Placebo. |
N=29 VD=14 PL=15 |
16♀
and 13♂ VD (8♀/7♂) PL (8♀/6♂) |
VD:
51.1 (1.16) PL:
46.8 (2.7) |
VD:
32.9 (1.7) PL:
31.6 (1.4) |
VD:
27.5 (1.5) PL:
27.5 (1.5) |
|
Perry 2019
|
UK |
800 IU daily vitamin D3, Placebo. |
N=50 VD=24 PL=26 |
37♀ and 13♂ VD (17♀/7♂) PL (20♀/6♂) |
VD: 63.0 (5.8) PL: 63.6 (7.2) |
VD: 28.2 (4.0) PL: 29.2 (5.6) |
VD: 26.6 (8.5) PL: 25.3 (8.7) |
1BMI:
Body Mass Index; 2VD: Vitamin D group; 3PL: Placebo
group; 4CI: Consistently Insufficient; 5F: Fluctuating; 6CS:
Consistently Sufficient.
III Risk of Bias Assessment
All the nine trials were classified as being at low bias risk (Figure 2). The GRADE showed high quality of evidence for serum vitamin D3 levels, biomarkers (calcium), WOMAC pain, stiffness, and function, changes in tibial cartilage volume and adverse events (Table 2) [15].
IV Serum Vitamin D3 Levels
Five studies reported the data on vitamin D3 levels [6, 7, 10, 12, 19]. The serum vitamin D3 levels had increased by 7.0136 ng/ml in the vitamin D group and were reduced by -5.76 ng/ml in the placebo group. Pooled estimates suggested the vitamin D supplementation was associated with a significant increase in serum vitamin D3 levels, administered alone or accompanied by multimodal treatment (Std MD=-0.74 [-0.89, -0.60]; I2=92%; p=0.00001). In the Australian studies [16, 17], the mean serum 25(OH) D level increased by 16,24 ng/ml in the vitamin D group and by 2,68ng/ml in the placebo groups over two years. 62% of participants (n=124/200) in the placebo group still reached a sufficient level of serum 25(OH) D. The high proportion of participants achieving sufficient vitamin D levels in the placebo group may thus have masked the beneficial effects of vitamin D supplementation [16, 17]. There was high heterogeneity among the studies.
V Serum Inflammatory and Biochemical Markers Levels
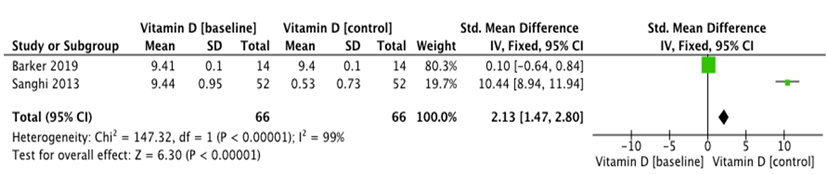
Three studies reported data on biomarker levels [7, 10, 18]. Sanghi et al. demonstrated relevant change of biochemical markers, with serum calcium (MD= 0.67[0.45, 0.89]; p=0.001) and alkaline phosphatase (MD=57.94[41.72, 74.16]; p=0.001) increasing significantly in the vitamin D group versus placebo group [10]. However, the rest of the biomarkers did not show significant differences between both groups at the end of the intervention.
Pooled estimates suggested the serum calcium obtained a significant increase (Std MD=2.13[1.47, 2.80]; I2=99%; p=0.00001) (Figure 3). Meanwhile, the inflammatory biomarkers IL-6 (p= 0.45), IL-8 (p=0.23), IL-10 (p=0.21) and hs-CRP (p=0.60) did not obtain significant decreases in the vitamin D group.
VI Changes in WOMAC Scores
Three studies reported WOMAC score changes [10, 17, 19]. Pain decreased in both the vitamin D and placebo groups. However, pooled results showed that vitamin D supplementation was associated with significantly greater pain reduction compared to the placebo group (Std MD=1.08[0.90, 1.25]; I2=99%; p=0.00001) (Figure 4). There was significant heterogeneity among the studies.
Table 2: Grading of Recommendations Assessment, Development
and Evaluation (GRADE).
|
|
|
|
|
|
|
|
|
Summary of Results |
|
|
||||
|
|
Quality Assessment |
# of patients |
Effect |
Quality |
Importance |
|||||||||
|
# of studies |
Design |
Risk of bias |
Inconsistency |
Indirectness |
Imprecision |
|
Other considerations |
Vitamin D groups |
Placebo groups |
Relative (95% CI) |
Absolute |
|
|
|
|
|
Serological levels of
vitamin D3 |
|||||||||||||
|
4 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
414 |
402 |
- |
-0.75 [-0.89,-0.60] |
⊕⊕⊕⊕ HIGH |
IMPORTANT |
|
|
|
Serological calcium
levels |
|||||||||||||
|
2 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
66 |
66 |
- |
2.13 [1.47, 2.80] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
WOMAC pain |
|||||||||||||
|
3 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
334 |
328 |
- |
1.08 [0.90,1.25] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
WOMAC function |
|||||||||||||
|
3 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
334 |
328 |
- |
1.10 [0.92,1.27] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
WOMAC stiffness |
|||||||||||||
|
2 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
261 |
255 |
- |
0.72 [0.54,0.90] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
Changes in tibial
cartilage volume |
|||||||||||||
|
2 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
282 |
277 |
- |
0.62 [0.44,0.80] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
Adverse events |
|||||||||||||
|
3 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
162/519 (31.2%) |
147/514 (28.6%) |
RR=1.06 |
- |
⊕⊕⊕⊕ HIGH |
IMPORTANT |
|
Figure 2: Risk of bias summary.
Figure 3: Forest plot showing the effect of vitamin D on the serum calcium.
Figure 4: Effect of vitamin D on WOMAC pain.
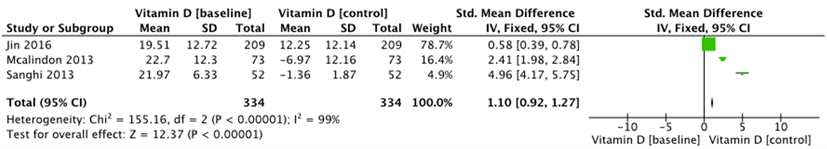
WOMAC function increased in both groups (vitamin D and placebo groups). Pooled results showed that vitamin D supplementation was associated with a significant reduction of functional problems (Std MD=1.10[0.92, 1.27]; I2=99%; p=0.00001) (Figure 5). Significant heterogeneity was found among the studies. Finally, WOMAC stiffness also decreased in both groups and the pooled results showed that vitamin D supplementation had no impact on WOMAC stiffness independently of the dose (Std MD=0.72[0.54, 0.90]; I2=98%; p=0.00001), with significant heterogeneity among the studies.
Figure 5: Effect of vitamin D on WOMAC function.
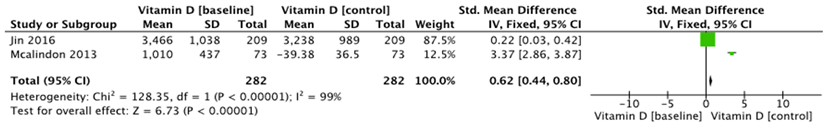
VII Changes in Tibial Cartilage Volume
Two studies reported changes in tibial cartilage volume (17,19). Pooled results showed that there were no significant differences in tibial cartilage volume changes (Std MD=0.62[0.44, 0.80]; I2=99%; P=0.00001) (Figure 6). There was significant heterogeneity among the studies.
Figure 6: Effect of vitamin D on tibial cartilage volumen.
VIII Changes in Synovial Tissue Volume (STV) and Subchondral Bone Marrow Lesions (BML)
Perry et al. found a small decrease in STV in both the vitamin D supplementation group -364.3 mm3 - and in the placebo group -309.6 mm3 (MD=-54.6[-1537.8, 1428.5]). The results showed no significant differences between the groups at 2 years of evaluation - 155.4 mm3 and 61.5 mm3 (MD=93.9[-1605.0, 1792.7]), respectively. No statistically significant differences were found for the BML volume, both at one and two years of control. At one year it decreased by an average of -506.7 mm3 in the placebo group, while in the vitamin D group, the volume increased by 1669.0 mm3 (MD=2175.7[-1263.8, 5615.1]). Furthermore, in the second year there was a small decrease for both groups, -506.9 mm3 for the vitamin D group and -193.4 mm3 for the placebo (MD=-313.5[-4244.7, 3617.7]) [12].
IX Changes in Effusion-Synovitis Volume
One study measured synovial effusion volume, showing statistically significant results in favour of vitamin D for both absolute and relative spillage, specifically in the suprapatellar pouch (Mean= -2,49 ml, 95%IC [-4.74,-0.25]; p=0.03) [9].
X Adverse Events
Three studies reported the data on adverse events [6, 17, 19]. The incidence of adverse events was 31.2% in the vitamin D and 28.6% in the placebo group. The most common being hypercalcemia and hypercalciuria. There were no significant differences in the proportion of patients between the two groups (vitamin D and placebo) (RR=1,063).
Discussion
Several high-quality studies showed a positive correlation between low vitamin D levels and degenerative joint disorders [20-23]. However, some have not provided a consensus on the effect of vitamin D on knee OA progression, presenting negative or weak association [24-26]. These studies vary in design, characteristics of included patients, and evaluation methods; therefore, they may be subject to significant bias.
In this meta-analysis, we found that vitamin D serum levels increased significantly in the vitamin D group. However, arthritic symptoms were only significantly improved in the studies that included patients with low vitamin D levels and with daily doses higher than 2,000 UI. Three articles detail that they used or allowed the use of alternative therapies (25, 115, 116). In addition to the vitamin D intervention, Sanghi et al. performed a multimodal therapy (education, exercise, insoles, application of heat, NSAIDs and/or paracetamol). Barker et al. only accepted the use of acetaminophen and finally, McAlindon et al. allowed concomitant use of NSAIDs and analgesics, recording the information in each control. Results observed in the subset analyses for the WOMAC pain outcome by McAlindon et al. demonstrated the effects were similar (MD=-0.87[-2.12, 0.38]), although the data indicated a benefit in favour of vitamin D supplementation, the difference was not significant (p=0.17) [19].
Another randomized study obtained an increase in WOMAC pain in the placebo group and decrease in vitamin D group, 0.71 and -0.08 (MD=-0.79[-2.31, 0.74]), respectively. Stiffness decreased in both groups (MD=-1.52[-3.24, 0.21]) and WOMAC function increased for both groups (MD= -0.65[-2.09, 0.79]). None of the above parameters reached statistically significant differences [6]. Both studies have limitations. The first study included patients without vitamin D deficiency who may not benefit from vitamin D supplementations and patients whose disease was too severe to respond to vitamin D treatment and the second study lost to follow-up 16% of the placebo group and 21% of the treatment group and the supplementation dose was low (800 IU/day) [6, 19].
Sanghi´s et al. results demonstrated improvement for both pain and functional disability in patients who received vitamin D supplementation (60,000 IU per day for 10 days followed by 60,000 IU once a month for a year)(10). VAS (Visual Analog Scale) pain (100 mm) decreased by -0,26 units in the vitamin D group, while in the placebo group increased 0.13 units (MD=-0.39[-0.71, -0.08]; p=0.020), just as a WOMAC pain (0-100) decreased -0.55 units in the vitamin D group it increased 1.16 units in the placebo group (MD=-1.70[-2.28, 1.12]; p=0.001). Furthermore, the WOMAC function decreased by -1.4 units in the vitamin D group, while the placebo group obtained 0.7 units (MD=-2.05[-2.92, -1.19]; p=0.001). Although the results are better in the group that received vitamin D supplementation (60,000 IU/day), they are insufficient to establish a significant clinical difference [10]. This could be attributable to the nature of pain in knee OA which is a chronic slowly progressive disease. Additionally, a study by Zheng et al. (50,000 IU per month for 24 months) found significant differences in the total WOMAC score (p=0.01) and function (p=0.01) in the three vitamin D groups. However, they did not achieve an important difference in pain (p=0.11) and stiffness (p=0.17) between the groups [7]. They hypothesized that the high proportion of patients included with sufficient vitamin D level in the placebo group might have diluted a beneficial effect of vitamin D supplementation; thus, they performed a post hoc analysis. Those patients who maintained sufficient serum levels over two years were associated with beneficial effects on joint structural and symptomatic changes in knee OA patients.
Studies on the association between vitamin D status and chronic pain incidence have been conflicting, and there are many factors influencing variations in clinical trial studies. Recent well-controlled studies have shown that hypovitaminosis D shows a strong correlation with bone pain [27-29]. However, the possible underlying mechanisms by which vitamin D could exert its analgesic effects are poorly understood. The hypothesis that vitamin D influences pain signaling pathways is biologically plausible because the expression of the VDR receptor gene has been demonstrated in relevant tissues such as the skin, which in addition to being the most important organ system protecting the body, is in charge of detecting damage and synthesizing vitamin D [30]. Other important tissues where the VDR receptor is expressed are: dorsal root ganglia (stimulus conduction), spinal cord (transmission/modulation) and the brain where secondary neurons transmit the stimulus to the cortex, decoding the message and allowing pain perception [30].
Pain is a common presenting symptom in patients with OA. However, peripheral and central sensitization alters nociception processing pathways in each individual, so the clinical presentation of pain varies between subjects, even when the damage at the radiographic level is similar [31, 32]. Because pain processing is complex and not fully understood, no conclusive results have been shown to establish a significant correlation [33].
Regarding changes in tibial cartilage volume, two studies found no significant effects on the structural change of knee cartilage and bone marrow lesions [17, 19]. The Osteoarthritis Initiative (OAI) is the largest and most complete epidemiological study of knee OA, with 4,796 participants [34]. The article by Zhang et al. takes a sample of 418 subjects from the mentioned study, which had serum concentrations of vitamin D and parathyroid hormone available, in addition to conventional X-rays to assess the progression of disease. Participants with vitamin D <15 ng/ml had a double risk of knee OA progression compared to those subjects with higher serum vitamin D levels [23]. In the 8-year Framingham cohort study, McAlindon et al. reported that the risk of knee osteoarthritis progression tripled in participants with low serum vitamin D levels [35]. Similarly, two studies demonstrated an association between hypovitaminosis D and decreased cartilage loss assessed by X-rays and MRI (20,22). Therefore, vitamin D supplementation in studies that included subjects with low vitamin D levels suggests effectiveness in both preventing and treating OA (9,17,19). In a study of 175 participants with symptomatic knee OA, Kellgren & Lawrence ≤2 and 25(OH)D <30 ng/ml received 40,000 IU of vitamin D2 per week for 6 months, demonstrating decreased oxidative protein damage and VAS pain (p=0.002), with improvement in these patients’ quality of life and physical performance (p=0.05) [36]. Additionally, a 5-year cohort study (n=769) showed that correcting moderate vitamin D deficiency can attenuate worsening knee and hip pain in older people [37].
However, the necessary clinical dose was not established due to the heterogeneity of the studies regarding intervention dosage and duration. In 2017, a meta-analysis by Gao evaluated the effects of vitamin D supplementation in preventing symptoms and structural progression, and suggested that doses higher than 2,000 IU/day achieved beneficial effects in the clinical symptoms of osteoarthritis (p=0.361)[38].
Regarding safety, there are no significant differences in the incidence of adverse events between both study groups [6, 17, 19]. Hypercalcemia is the most common event, which could be related to high doses of vitamin D [39]. A systematic review investigated the efficacy of a single large bolus dose in treating vitamin D deficiency, concluding that single doses of vitamin D3 ≥300,000 IU are more effective in improving vitamin D status and suppressing PTH levels for up to 3 months. However, it does not rule out that in certain populations lower doses are sufficient. Doses >500,000 should be used with caution to minimize adverse events [40]. All studies agreed that their results could have been more relevant in longer-term measurements of structural and symptomatic changes, including only patients with vitamin D deficiency and representative sample size. This is because OA is a slow-progression chronic disease and the action of vitamin D could have a slow acting effect [9, 10].
In the present review, the methodology was performed according to the Cochrane Manual of Systematic Review. The included articles used the ACR, American College Rheumatology, criteria or conventional/gold standard radiographic methods (MRI, MRI with gadolinium contrast). However, it should be considered that the search was restricted to articles in English and Spanish, so there could be loss of information available in other languages.
Conclusion
Vitamin D supplementation was effective in increasing vitamin D levels and improving pain, physical function and, to a lesser extent, stiffness in patients with symptomatic knee osteoarthritis. However, it showed no beneficial effect in preventing knee OA progression, and the information regarding structural cartilage changes and associated tissue has a weak association with vitamin D supplementation. Therefore, its clinical OA use is recommended as an adjunct to its treatment, because the current lack of evidence means that it cannot be considered as an effective independent treatment for chronic pain and the effective dose and amount of time cannot be determined.
Despite various limitations of our included studies (small sample size, inclusion of patients with sufficient vitamin D levels, duration of the intervention, inclusion of patients with advanced OA (Kellgren and Lawrence 4), without considering seasonal fluctuations, among others), it is possible to detect improvement in the vitamin D supplementation group versus placebo group.
However, the limitations do not allow for the delivery of clinically specific data on certain parameters studied, and may mask the results and show certain controversies, especially in the levels of metabolic and inflammatory biomarkers. There is therefore currently a lack of strong evidence to support the use of vitamin D supplementation in patients with degenerative joint disorders.
Funding
None.
Conflicts of Interest
None.
Ethical Approval
This study is a part of a research project approved by the Ethics and Scientific Committee of Universidad de Los Andes (CEC 201951).
Consent to Participate
Not applicable.
Author Statement
All authors reviewed and approved the manuscript for publication (statement included). All data are available in the tables and figures of the manuscript.
Code Availability
Not Applicable.
Author Contributions
Gustavo Moncada: Conceived and designed the analysis, performed the analysis, wrote the paper; Gabriela Diaz Mujica: Collected the data, contributed data or analysis tools, performed the analysis, trained and calibration in selection and evaluation process - pilot test, manuscript modifications and approval; Constanza Valdés: Conceived and designed the analysis, collected the data, performed the analysis, trained and calibration in selection and evaluation process - pilot test, manuscript modifications and approval.
Article Info
Article Type
Review ArticlePublication history
Received: Tue 09, Aug 2022Accepted: Wed 31, Aug 2022
Published: Fri 16, Sep 2022
Copyright
© 2023 Gustavo Moncada. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.DOBCR.2022.03.01
Author Info
Gustavo Moncada Gabriela Díaz Mujica Constanza Valdés
Corresponding Author
Gustavo MoncadaDental School, Universidad de Los Andes, Santiago, Chile
Figures & Tables
Table 1: Baseline characteristics of patients included in
the meta-analysis.
|
Study |
Country |
Treatment
regimen |
No.
of patients |
♀ Female/
♂ Male |
Age
(Mean, SD) |
BMI1 (Mean,
SD) |
Vitamin D3 (ng/ml) (Mean, SD) |
|
Sanghi 2013 |
India |
60.000 IU daily vitamin D3, Placebo. |
N=103 VD3=52 PL4=51
|
66♀ and 37♂ VD (30♀/21♂) PL (36♀/16♂) |
VD2: 53.24 (9.64) PL3: 53.00 (7.44) |
VD: 25.86 (2.46) PL: 25.65 (2.58) |
VD: 14.812
(3.012) PL: 15.008 (3.012) |
|
McAlindon
2013 |
USA |
2.000 IU daily vitamin
D3, Placebo. |
N=146 VD=73 PL=73
|
89♀
and 57♂ VD (49♀/24♂) PL (40♀/33♂) |
VD:
61.8 (7.7) PL:
63.0 (9.3) |
VD:
30.5 (5.0) PL:
30.8 (6.4) |
VD:
22.7 (11.4) PL:
21.9 (1.7) |
|
Jin 2016 |
Australia |
50.000 IU daily vitamin D3, Placebo. |
N=413 VD=209 PL=204
|
208♀ and 205♂ VD (106♀/103♂) PL (102♀/102♂) |
VD: 63.5 (6.9) PL: 62.9 (7.2) |
VD: 29.6 (5.4) PL: 29.6 (4.6) |
VD: 17.48 (4.72) PL: 17.52 (5.08) |
|
Arden
2016 |
UK |
800 IU daily vitamin D3, Placebo. |
N=474 VD=237 PL=
237
|
289♀
and 185♂ VD (144♀/93♂) PL (145♀/92♂) |
VD:
64 (8) PL:
64 (8) |
VD:
30 (5) PL:
29 (5) |
VD:
20.7 (8.9) PL:
20.7 (8.1) |
|
Zheng 2017 |
Australia |
50.000 IU daily
vitamin D3, Placebo. |
N=340 CI=46 FL=68 CS=226
|
160♀ and 180♂ CI (24♀ and 22♂) FL (38♀/30♂) CS (98♀/128♂) |
CI4:62.6 (8.0) F5: 62.9 (6.1) CS6: 63.5 (7.2) |
CI: 30.6 (4.6) F: 29.0 (4.5) CS: 29.4 (4.9) |
CI: ≤ 20 ng/ml F: > 20 ng/ml at either point CS: > 20 ng/ml at both months 3 and
24. |
|
Wang
2017 |
Australia |
50.000 IU daily vitamin D3, Placebo. |
N=413 VD=209 PL=204
|
208♀
and 205♂ VD (106♀/103♂) PL (102♀/102♂) |
VD:
63.55 (6.88) PL:
62.55 (7.22) |
VD:
29.57 (5.39) PL:
29.64 (4.62) |
VD:
17.496 (4.72) PL:
17.524 (5.06) |
|
Zheng 2018 |
Australia |
50.000 IU daily
vitamin D3, Placebo. |
N=200 VD=106 PL=94
|
107♀ and 93♂ VD (53♀/53♂) PL (54♀/40♂) |
VD: 63.3 (7.5) PL: 62.8 (7.3) |
VD: 29.4 (7.5) PL: 29.6 (4.0) |
VD: 17 (4.68) PL: 17.4 (5.04) |
|
Barker
2019 |
USA |
4.000 IU daily vitamin D3, Placebo. |
N=29 VD=14 PL=15 |
16♀
and 13♂ VD (8♀/7♂) PL (8♀/6♂) |
VD:
51.1 (1.16) PL:
46.8 (2.7) |
VD:
32.9 (1.7) PL:
31.6 (1.4) |
VD:
27.5 (1.5) PL:
27.5 (1.5) |
|
Perry 2019
|
UK |
800 IU daily vitamin D3, Placebo. |
N=50 VD=24 PL=26 |
37♀ and 13♂ VD (17♀/7♂) PL (20♀/6♂) |
VD: 63.0 (5.8) PL: 63.6 (7.2) |
VD: 28.2 (4.0) PL: 29.2 (5.6) |
VD: 26.6 (8.5) PL: 25.3 (8.7) |
1BMI:
Body Mass Index; 2VD: Vitamin D group; 3PL: Placebo
group; 4CI: Consistently Insufficient; 5F: Fluctuating; 6CS:
Consistently Sufficient.
Table 2: Grading of Recommendations Assessment, Development
and Evaluation (GRADE).
|
|
|
|
|
|
|
|
|
Summary of Results |
|
|
||||
|
|
Quality Assessment |
# of patients |
Effect |
Quality |
Importance |
|||||||||
|
# of studies |
Design |
Risk of bias |
Inconsistency |
Indirectness |
Imprecision |
|
Other considerations |
Vitamin D groups |
Placebo groups |
Relative (95% CI) |
Absolute |
|
|
|
|
|
Serological levels of
vitamin D3 |
|||||||||||||
|
4 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
414 |
402 |
- |
-0.75 [-0.89,-0.60] |
⊕⊕⊕⊕ HIGH |
IMPORTANT |
|
|
|
Serological calcium
levels |
|||||||||||||
|
2 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
66 |
66 |
- |
2.13 [1.47, 2.80] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
WOMAC pain |
|||||||||||||
|
3 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
334 |
328 |
- |
1.08 [0.90,1.25] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
WOMAC function |
|||||||||||||
|
3 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
334 |
328 |
- |
1.10 [0.92,1.27] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
WOMAC stiffness |
|||||||||||||
|
2 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
261 |
255 |
- |
0.72 [0.54,0.90] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
Changes in tibial
cartilage volume |
|||||||||||||
|
2 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
282 |
277 |
- |
0.62 [0.44,0.80] |
⊕⊕⊕⊕ HIGH |
CRITICAL |
|
|
|
Adverse events |
|||||||||||||
|
3 |
RCTs |
No serious risk of bias |
No serious inconsistency |
No serious indirectness |
No serious imprecision |
|
None |
162/519 (31.2%) |
147/514 (28.6%) |
RR=1.06 |
- |
⊕⊕⊕⊕ HIGH |
IMPORTANT |
|


References
1. Zhang W, Nuki G,
Moskowitz RW, Abramson S, Altman RD et al. (2010) OARSI recommendations
for the management of hip and knee osteoarthritis: part III: Changes in
evidence following systematic cumulative update of research published through
January 2009. Osteoarthritis Cartilage 18: 476-499. [Crossref]
2. Johnson VL, Hunter
DJ (2014) The epidemiology of osteoarthritis. Best Pract Res Clin Rheumatol
28: 5-15. [Crossref]
3. Mabey T, Honsawek S
(2015) Role of Vitamin D in Osteoarthritis: Molecular, Cellular, and Clinical
Perspectives. Int J Endocrinol 2015: 383918. [Crossref]
4. Zhang Y, Jordan JM
(2010) Epidemiology of Osteoarthritis. Clin Geriatr Med 26: 355-369. [Crossref]
5. Neogi T, Zhang Y
(2013) Epidemiology of Osteoarthritis. Rheum Dis Clin North Am 39: 1-19.
[Crossref]
6. Arden NK, Cro S,
Sheard S, Doré CJ, Bara A et al. (2016) The effect of vitamin D
supplementation on knee osteoarthritis, the VIDEO study: a randomised
controlled trial. Osteoarthritis Cartilage 24: 1858-1866. [Crossref]
7. Zheng S, Wang B,
Han W, Zhu Z, Wang X et al. (2018) Vitamin D supplementation and
inflammatory and metabolic biomarkers in patients with knee osteoarthritis:
post hoc analysis of a randomised controlled trial. Br J Nutr 120:
41-48. [Crossref]
8. Bikle DD (2012)
Vitamin D and bone. Curr Osteoporos Rep 10: 151-159. [Crossref]
9. Wang X, Cicuttini
F, Jin X, Wluka AE, Han W et al. (2017) Knee effusion-synovitis volume
measurement and effects of vitamin D supplementation in patients with knee
osteoarthritis. Osteoarthritis Cartilage 25: 1304-1312. [Crossref]
10. Sanghi D, Mishra A,
Sharma AC, Singh A, Natu SM et al. (2013) Does vitamin D improve
osteoarthritis of the knee: a randomized controlled pilot trial. Clin Orthop
Relat Res 471: 3556-3562. [Crossref]
11. Jin X, Wang BH,
Wang X, Antony B, Zhu Z et al. (2017) Associations between endogenous sex
hormones and MRI structural changes in patients with symptomatic knee
osteoarthritis. Osteoarthritis Cartilage 25: 1100-1106. [Crossref]
12. Perry TA, Parkes MJ,
Hodgson R, Felson DT, O’Neill TW et al. (2019) Effect of Vitamin D
supplementation on synovial tissue volume and subchondral bone marrow lesion
volume in symptomatic knee osteoarthritis. BMC Musculoskelet Disord 20:
76. [Crossref]
13. Sterne JAC, Savović
J, Page MJ, Elbers RG, Blencowe NS et al. (2019) RoB 2: a revised tool for
assessing risk of bias in randomised trials. BMJ 2019: l4898. [Crossref]
14. Higgins JPT, Green
S (editors) (2021) Cochrane Handbook for Systematic Reviews of
Interventions Version 5.1.0. The Cochrane Collaboration, 2011.
Available from www.cochrane-handbook.org.
15. Sanabria AJ, Rigau D, Rotaeche R, Selva A, Marzo
Castillejo M et al. (2015) GRADE: Methodology for formulating and grading
recommendations in clinical practice. Aten Primaria 47: 48-55. [Crossref]
16. Zheng S, Jin X,
Cicuttini F, Wang X, Zhu Z et al. (2017) Maintaining Vitamin D Sufficiency
Is Associated with Improved Structural and Symptomatic Outcomes in Knee
Osteoarthritis. Am J Med 130: 1211-1218. [Crossref]
17. Jin X, Jones G,
Cicuttini F, Wluka A, Zhu Z et al. (2016) Effect of Vitamin D
Supplementation on Tibial Cartilage Volume and Knee Pain Among Patients With
Symptomatic Knee Osteoarthritis: A Randomized Clinical Trial. JAMA 315:
1005-1013. [Crossref]
18. Barker T, Rogers
VE, Henriksen VT, Levy M, Schneider ED et al. (2019) Circulating cytokine
concentrations are not altered by supplemental vitamin D in knee
osteoarthritis: A pilot study. J Nutr Intermed Metab 18.
19. McAlindon T,
LaValley M, Schneider E, Nuite M, Lee JY et al. (2013) Effect of vitamin D
supplementation on progression of knee pain and cartilage volume loss in
patients with symptomatic osteoarthritis: a randomized controlled trial. JAMA
309: 155-162. [Crossref]
20. Bergink AP,
Uitterlinden AG, Van Leeuwen JPTM, Buurman CJ, Hofman A et al. (2009)
Vitamin D Status, Bone Mineral Density, and the Development of Radiographic
Osteoarthritis of the Knee: The Rotterdam Study. J Clin Rheumatol 15:
230-237. [Crossref]
21. Heidari B, Heidari
P, Hajian Tilaki K (2011) Association between serum vitamin D deficiency and
knee osteoarthritis. Int Orthop 35: 1627-1631. [Crossref]
22. Ding C, Cicuttini
F, Parameswaran V, Burgess J, Quinn S et al. (2009) Serum levels of vitamin D,
sunlight exposure, and knee cartilage loss in older adults: The Tasmanian older
adult cohort study. Arthritis Rheum 60: 1381-1389. [Crossref]
23.
Zhang
FF, Driban JB, Lo GH, Price LL, Booth S et al. (2014) Vitamin D deficiency
is associated with progression of knee osteoarthritis. J Nutr 144:
2002-2008. [Crossref]
24. Konstari S, Paananen M, Heliövaara M, Knekt P,
Marniemi J et al. (2012) Association of 25-hydroxyvitamin D with the incidence
of knee and hip osteoarthritis: a 22-year follow-up study. Scand J Rheumatol
41: 124-131. [Crossref]
25. Felson DT, Niu J,
Clancy M, Aliabadi P, Sack B et al. (2007) Low levels of vitamin D and
worsening of knee osteoarthritis: Results of two longitudinal studies. Arthritis
Rheum 56: 129-136. [Crossref]
26. Muraki S, Dennison
E, Jameson K, Boucher BJ, Akune T et al. (2011) Association of vitamin D
status with knee pain and radiographic knee osteoarthritis. Osteoarthritis
Cartilage 19: 1301-1306. [Crossref]
27. Brady SRE,
Naderpoor N, de Courten MPJ, Scragg R, Cicuttini F et al. (2019) Vitamin D
supplementation may improve back pain disability in vitamin D deficient and
overweight or obese adults. J Steroid Biochem Mol Biol 185: 212-217. [Crossref]
28.
Ghai
B, Bansal D, Kanukula R, Gudala K, Sachdeva N (2017) Vitamin D Supplementation
in Patients with Chronic Low Back Pain: An Open Label, SingleArm Clinical
Trial. Pain Physician 1: E99-E105. [Crossref]
29. Morandi G, Maines E, Piona C, Monti E, Sandri M
et al. (2015) Significant
association among growing pains, vitamin D supplementation, and bone mineral
status: results from a pilot cohort study. J Bone Miner Metab 33:
201-206. [Crossref]
30. Habib AM, Nagi K,
Thillaiappan NB, Sukumaran V, Akhtar S (2020) Vitamin D and Its Potential
Interplay With Pain Signaling Pathways. Front Immunol 11: 820. [Crossref]
31. Fingleton C, Smart
K, Moloney N, Fullen BM, Doody C (2015) Pain sensitization in people with knee
osteoarthritis: a systematic review and meta-analysis. Osteoarthritis
Cartilage 23: 1043-1056. [Crossref]
32. Roesel TR (2009)
Does the central nervous system play a role in vitamin D deficiency-related
chronic pain? Pain 143: 159-160. [Crossref]
33.
Cakar
M, Ayanoglu S, Cabuk H, Seyran M, Dedeoglu SS et al. (2018) Association between
vitamin D concentrations and knee pain in patients with osteoarthritis. PeerJ 6: e4670. [Crossref]
34. Nevitt M, Felson DT, Lester G (2021) La Iniciativa de
Osteoartritis: protocolo para el estudio de cohorte.
35. McAlindon TE,
Felson DT, Zhang Y, Hannan MT, Aliabadi P et al. (1996) Relation of Dietary
Intake and Serum Levels of Vitamin D to Progression of Osteoarthritis of the
Knee among Participants in the Framingham Study. Ann Intern Med 125:
353. [Crossref]
36. Manoy P,
Yuktanandana P, Tanavalee A, Anomasiri W, Ngarmukos S et al. (2017)
Vitamin D Supplementation Improves Quality of Life and Physical Performance in
Osteoarthritis Patients. Nutrients 9: 799. [Crossref]
37. Laslett LL, Quinn
S, Burgess JR, Parameswaran V, Winzenberg TM et al. (2014) Moderate
vitamin D deficiency is associated with changes in knee and hip pain in older
adults: a 5-year longitudinal study. Ann Rheum Dis 73: 697-703. [Crossref]
38. Gao XR, Chen YS,
Deng W (2017) The effect of vitamin D supplementation on knee osteoarthritis: A
meta-analysis of randomized controlled trials. Int J Surg 46: 14-20. [Crossref]
39. Tebben PJ, Singh RJ, Kumar R (2016) Vitamin D-Mediated Hypercalcemia: Mechanisms, Diagnosis, and Treatment. Endocr Rev 37: 521-547. [Crossref]
40. Kearns M, Alvarez J, Tangpricha V (2014) Large, single-dose, oral vitamin D supplementation in adult populations: a systematic review. Endocr Pract 20: 341-351. [Crossref]
