Clear Cell Sarcoma of Tendon and Aponeuroses with Excessive Melanin Production – An Unusual Presentation
A B S T R A C T
Clear cell sarcoma of tendons and aponeuroses also called as malignant melanoma of soft parts is a rare tumor of neural crest origin with 70% of them possessing balanced translocation t(12; 22). Approximately 50% of the tumor produces melanin which often shows focal pigmentation or needs special stains to demonstrate melanin. Production of excessive melanin by this tumor is very unusual and very few case reports are available in literature. We report a case of Clear cell sarcoma with excessive melanin production in a 50-year-old female patient involving the left popliteal fossa.
Keywords
Clear cell sarcoma, tendon and aponeuroses, excessive melanin pigmentation
Introduction
Clear cell sarcoma (CCS) is a rare melanin-producing soft tissue sarcoma first described by Enzinger in 1965 [1]. He called it as “clear cell sarcoma of tendons and aponeuroses and is now being also referred to as “malignant melanoma of soft parts,” but it is distinct from cutaneous melanoma despite certain histologic similarities. There have been controversies regarding the histogenesis of CCS. The demonstration of melanin pigment and melanosomes in CCS by light microscopy and electron microscopy, respectively, supports an origin from the neural crest, and 70% possess a consistent balanced translocation t(12; 22) [2]. Presence of abundant melanin production in this tumor is a rare presentation with very few case reports available in literature [3, 4]. We present here such a rare case of CCS with excessive melanin production.
Case Report
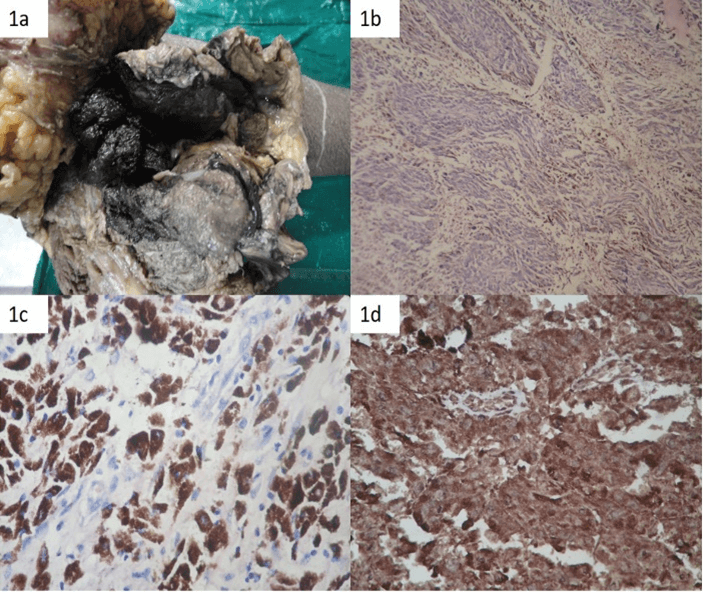
A 50-year-old female patient presented with swelling in the posterior part of the left leg in the popliteal region. On examination was found to have 6×6 cm, firm and mobile swelling. Trucut biopsy done outside from the swelling was reported as fibrosarcoma. Review of the slide showed predominantly necrotic material with few spindled cells and was difficult to opine on a diagnosis. Hence a repeat biopsy was advised which showed compact nests and fascicles of spindle shaped cells separated by fibrous septae with mild to moderate pleomorphism with vesicular nucleus and prominent eosinophilic nucleoli. The cytoplasm showed abundant melanin deposition obscuring the morphology at places. Melanin bleach was done to observe the morphology in detail and immunohistochemical markers S100 and HMB45 were done which showed strong positivity in the tumor cells. Thus, a diagnosis of clear cell sarcoma (malignant melanoma of soft parts) was considered. The patient was then planned for surgical resection and finally proceeded with an above knee amputation surgery. Intraoperatively the tumor was found attached to the inferior part of gastrocnemius tendon. On gross examination the tumor was measuring 13×7×6 cm and was found attached to the gastrocnemius tendon (Figure 1a).
The overlying skin and dermis were free of tumor. The outer surface and cut surface of the tumor appeared totally black, solid with friable areas of necrosis and areas of cystic degeneration. Histopathological examination showed similar morphology as in trucut biopsy (Figures 1b & 1c). There were also large areas of necrosis. The tumor cells showed strong positivity for S100 and HMB45 (Figure 1d) confirming the diagnosis of CCS. The patient was then followed up and after 4 months presented with recurrence at the amputated stump site. Fine needle aspiration cytology was performed which confirmed tumor recurrence.
Figure 1: a) Above knee amputation specimen showing the tumor which appears blackish on gross examination; b) Histopathology shows spindle shaped tumor cells with melanin pigmentation (H&E 100X); c) Histopathology section showing tumor cells with extensive cytoplasmic melanin pigmentation (H&E 400X); d) Immunohistochemistry with HMB 45 showing strong positivity (IHC, 400X).
Discussion
CCS mainly affects young adults between the ages of 20 and 40 years with a median age of about 30 years, whereas our patient is of 50 years. Approximately 40% of cases occur on the foot and ankle, 30% on the knee, thigh, and hand. The head and neck region and the trunk are distinctly unusual sites [5]. They arise in deep soft tissue and unless the lesion is extremely large or distal, the overlying skin and dermis is usually not involved. Macroscopically, in a small proportion of cases the melanin may be prominent enough to be visualized as foci of dark brown or black discoloration. But the entire tumor showing prominent blackish discoloration due to melanin deposition as seen in our case is very rare.
On microscopy tumors consist of compact nests or fascicles of predominantly fusiform or spindled cells with a clear cytoplasm bordered and defined by a delicate framework of fibrocollagenous tissue contiguous with adjacent tendons or aponeuroses. The cytological features are reminiscent that of malignant melanoma. The cytoplasm varies from clear to weakly eosinophilic and contains large amounts of intracellular glycogen. Clear cells and eosinophilic cells coexist in different portions of the same neoplasm with focal transitions between the two. A highly characteristic feature is the multinucleated tumor giant cells with 10-15 peripherally placed nuclei. However, this feature was not identified in our case [6].
Melanin is present in over 50% of clear cell sarcomas but is usually not abundant enough to be seen on hematoxylin-eosin stain. It can be detected with appropriate histochemical stains like Fontana or Warthin-Starry or immunohistochemical stains [6]. Presence of excessive melanin as in our case is very unusual in a case of clear cell sarcoma with very few case repots available in literature. Okamoto et al. report a 62-year-old male with a CCS arising in the area of the M. flexor carpi ulnaris. Grossly, the tumor had a black appearance. Light microscopic inspection showed a large amount of melanin [3]. Mukai et al. in his series observed two cases of CCS with excessive melanin production [4]. Immunohistochemically all CCS diffusely express S-100 protein and most also express antigens associated with melanin synthesis like HMB-45, Melan-A, Mel-CAM, MiTF. Other markers that can be expressed are Neuron-specific enolase, Leu-7, and LN3 [7].
Differential diagnosis depends on the presence or absence of melanin. In the absence of melanin, sarcomas with a predominant fascicular growth pattern such as fibrosarcoma, synovial sarcoma, and malignant peripheral nerve sheath tumor are the close differentials. This was experienced even in this case, as this tumor was diagnosed as fibrosarcoma outside. The classical features of clear cell sarcoma including prominent melanoma-like nucleoli, clear cytoplasm, and immunophenotypic profile differentiate it from these spindle cell sarcomas.
On the other hand, distinguishing clear cell sarcoma from other melanin-producing lesions like nodular melanomas, cellular blue cell nevus, paraganglioma-like dermal melanocytic tumor (PDMT), can be more difficult and may require correlation of the histologic, clinical, and molecular data. CCS originates in deep structures, rarely involve the dermis, and have a predominantly and relatively uniform spindle-cell appearance whereas nodular melanomas have the epithelioid appearance. Cellular blue nevus can occur in a similar age and location and have certain common histologic features including spindled cells and giant cells with clear cytoplasm but typically they are dermal-based lesions with a peripheral zone that resembles a neurofibroma by virtue of the interdigitation of slender pigmented dendritic cells with surrounding collagen. The cells lack atypia and have small, pinpoint nucleoli. Recurrent cellular blue nevi, however, can acquire more atypical cytologic features such that a distinction from clear cell sarcoma may not be possible. PDMT, comprises zellballen-like nests of cells of distinctly low nuclear grade with a clear to eosinophilic cytoplasm and the lesion is dermal based and rarely extend to deep structures [6, 8]. However, in all ambiguous situations, molecular genetic analysis is highly recommended since the t(12; 22) is characteristic of CCS has not been identified in other lesions [2].
Radical surgery is the mainstay of therapy, and chemotherapy have little efficacy. Recurrences range from 14% to 39%, whereas metastases to lung or lymph node develop in approximately one-half of patients within an interval of 2-8 years. Late metastases after 10-20 years have been reported in patients with repeated local recurrences [9, 10]. Several studies have attempted to identify other prognostic factors and found size, followed by necrosis, to be the most robust prognostic factors with tumors >5 cm having a significantly worse outcome than those that are smaller. The size of the tumor in our case is >5cm and there were large areas of necrosis indicating a poor prognosis [10].
Conclusion
Clear cell sarcoma with excessive melanin production is a rare presentation. The tumor can be differentiated from malignant melanoma when the epidermis is not involved by the tumor. It should also be differentiated from other mimics and genetic analysis for translocation t(12; 22) is advised in ambiguous situations especially in tumors without melanin pigmentation.
Conflicts of Interest
None.
Funding
None.
Article Info
Article Type
Case ReportPublication history
Received: Mon 13, Sep 2021Accepted: Sat 13, Nov 2021
Published: Mon 29, Nov 2021
Copyright
© 2023 Kalaivani Selvi. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2021.11.09
Author Info
Kalaivani Selvi Rajesh Nachiappa Ganesh
Corresponding Author
Kalaivani SelviDepartment of Pathology, Sri Venkateswaraa Medical College Hospital and Research Centre, Puducherry, India
Figures & Tables
References
1. Enzinger FM (1965)
CLEAR-CELL SARCOMA OF TENDONS AND APONEUROSES. AN ANALYSIS OF 21 CASES. Cancer
18: 1163-1174. [Crossref]
2. Mrozek K,
Karakousis CP, Perez Mesa C, Bloomfield CD (1993) Translocation t(12; 22)(q13;
q12.2–12.3) in a clear cell sarcoma of tendons and aponeuroses. Genes
Chromosomes Cancer 6: 249-252. [Crossref]
3. Okamoto T, Tsuji H,
Mizobuchi K (1984) A case of clear cell sarcoma of tendons and aponeuroses with
a large amount of melanin--malignant melanoma of the soft part. Gan No
Rinsho 30: 1417-1421. [Crossref]
4. Mukai M, Torikata C, Iri H, Mikata
A, Kawai T et al. (1984)
Histogenesis of clear cell sarcoma of tendons and aponeuroses. An
electron-microscopic, biochemical, enzyme histochemical, and
immunohistochemical study. Am J Pathol 114: 264-272. [Crossref]
5. Montgomery EA, Meis
JM, Ramos AG, Frisman DM, Martz KL (1993) Clear Cell Sarcoma of Tendons and
Aponeuroses: A Clinicopathologic Study of 58 Cases With Analysis of Prognostic
Factors. Int J Surg Pathol 1: 89-99.
6. Weiss SW, Goldblum
JR (2008) Malignant Tumors of the
Peripheral Nerves. In: Enzinger and Weiss ‘s Soft Tissue Tumors. 5th
ed. St. Louis: Mosby 926-931.
7. Kindblom LG,
Lodding P, Angervall L (1983) Clear cell sarcoma of tendons and aponeuroses. An
immunohistochemical and electron microscopic analysis indicating neural crest
origin. Virchows Arch A Pathol Anat Histopathol 401: 109-128. [Crossref]
8. Deyrup AT, Althof
P, Zhou M, Morgan M, Solomon AR et al. (2004) Paraganglioma-like dermal
melanocytic tumor: a unique entity distinct from cellular blue nevus, clear
cell sarcoma, and cutaneous melanoma. Am J Surg Pathol 28: 1579-1586. [Crossref]
9. Ferrari A, Casanova M, Bisogno G, Mattke A, Meazza C et al. (2002) Clear cell sarcoma of tendons and aponeuroses in pediatric patients: a report from the Italian and German Soft Tissue Sarcoma Cooperative Group. Cancer 15: 3269-3276. [Crossref]
10. Lucas DR, Nascimento AG, Sim FH (1992) Clear cell sarcoma of soft tissues: Mayo Clinic experience with 35 cases. Am J Surg Pathol 16: 1197-1204. [Crossref]
