Characteristics of Isolated Small Bowel Crohn’s Disease: A Comprehensive Comparison with Small Bowel and Colon Involved Crohn’s Disease
A B S T R A C T
Background: Some studies showed that small bowel involved Crohn’s Disease (CD) was more serious, which required early identification and aggressive treatment. However, studies investigating small bowel involved CD, especially isolated small bowel CD with large sample size, are limited.
Methods: From November 2013 to August 2018, consecutive patients who were confirmed to have small bowel involved CD by double-balloon enteroscopy (DBE) were retrospectively reviewed. They were divided into two groups based on whether colon involved: isolated small bowel involved (case group) and small bowel + colon involved (control group).
Results: 204 patients (154 males and 50 females) with small bowel involved CD (98 in case group and 106 in control group) were chosen. Compared with the control group, the case group had a higher proportion of bowel resection history and stricturing behaviour, while had a lower proportion of elevated platelet and erythrocyte sedimentation rate level. The two groups had no difference in bowel resection risk since the DBE procedure (P=0.277), but the case group had a higher probability since the onset of the symptoms (P=0.003). Diagnosis at > 40-year-old, thickest bowel wall>7mm, and stricture found in DBE were risk factors for bowel resection, while patients with irregular ulcer, received infliximab, corticosteroids, or enteral nutrition after DBE may have a lower risk.
Conclusion: The isolated small-bowel CD was concealed and would be easily overlooked, though they may be more serious. The prognosis might not be as bad as it used to be considered if the disease could be diagnosed, evaluated comprehensively, and treated appropriately early.
Keywords
Crohn's disease, bowel resection, small bowel, risk, double-balloon enteroscopy
Introduction
Crohn’s Disease (CD) is a chronic, transmural inflammation that can involve any part of the gastrointestinal tract [1]. It was reported that, 10-30% of CD patients had small bowel involvement, and up to 20% suffered from isolated small bowel diseases [2, 3]. The diagnosis of isolated small bowel CD is difficult due to its specific anatomic location. Some studies showed that CD with small bowel involvement was more serious than colonic CD, which required more aggressive treatment [4-6]. Early identification of small bowel involved CD; especially isolated small bowel CD, is of vital importance. The clinical characteristics of isolated small bowel CD have not been sufficiently investigated. Du et al. analysed the clinical features and prognosis of isolated small bowel involved CD and small bowel + colon involved CD, and they found that isolated small bowel CD were more serious and had worse outcomes [7]. However, in their study, they enrolled the patients by the results of capsule endoscopy (CE), ileocolonoscopy, and enhanced computed tomography (CT), without the verification of double-balloon enteroscopy (DBE), which permits direct visualization of the small bowel and biopsy, as well as provide more accurate and convincible diagnosis in small bowel diseases [8, 9]. Moreover, the sample size in their study is limited. So, we selected the patients with small bowel involved CD, who were confirmed by DBE, and compared the clinical and endoscopic differences between isolated small bowel involved CD and small bowel + colon involved CD, so as to provide more information on isolated small bowel CD, hoping to contribute to early identification and draw attention to it.
Methods
I Patients
From November 2013 to August 2018, consecutive patients who were suspected to have small bowel involved CD and underwent DBE in our hospital were retrospectively reviewed. Based on the management consensus of inflammatory bowel disease for the Asia-Pacific region, those who were diagnosed as CD were finally chosen for analysis [10]. The patients were divided into two groups based on whether colon involved: isolated small bowel involved (case group) and small bowel + colon involved (control group).
II Data Collection
A manual chart review was performed to acquire the following data: age, gender, height, weight, symptoms, medical histories, laboratory tests [blood routine, erythrocyte sedimentation rate (ESR), serum albumin (ALB), fecal calprotectin (FC)], imaging results [computed tomography enterography (CTE), magnetic resonance enterography (MRE), and anal magnetic resonance image (MRI)], endoscopic findings (gastroduodenoscopy, ileocolonoscopy, enteroscopy, and capsule endoscopy), pathology results, final diagnosis, and treatments. To obtain the follow-up results of the patients, we firstly searched for the information, including lab results, image studies, surgical history, pathology reports and etc. in our electronic medical records (EMR); then we telephoned the patients who did not have any information since the initial hospitalization when they underwent DBE, mainly to obtain the information regarding bowel resection. The study protocol was approved by the Institutional Review Board of our hospital.
III Outcome Parameters
We compared the differences between the case and control group, mainly from the aspects of symptoms, surgery history, Montreal classification, laboratory results, enteroscopy findings, and risk for bowel resection in follow-up. The time when bowel resection occurred was regarded as the end point of follow-up.
IV Statistical Analysis
IBM SPSS Statistic Version 22 was used to perform the statistical analyses. Continuous variables with normal distribution were presented with a mean (standard deviation, SD), or otherwise used median (interquartile range). Categorical variables were presented with a number (percentage). Continuous variables, with a normal distribution, were tested by the Student’s t-test or otherwise by using nonparametric Mann-Whitney U test. Categorical variables were analysed by using the χ2 test. GraphPad Prism Version 8 was used to draw the Kaplan-Meier curves in order to evaluate the proportion of bowel resection during the follow-up. Cox regression model was used to identify possible risk factors for bowel resection as well as to calculate the hazard ratio (HR) and 95% confidence interval (CI). Univariate analysis was firstly done, and variables with a P-value <0.1 or interested were included in the multivariate analysis (Forward: LR model was chosen). Statistical significance was defined as P< 0.05 (two-tailed).
Results
I Patients
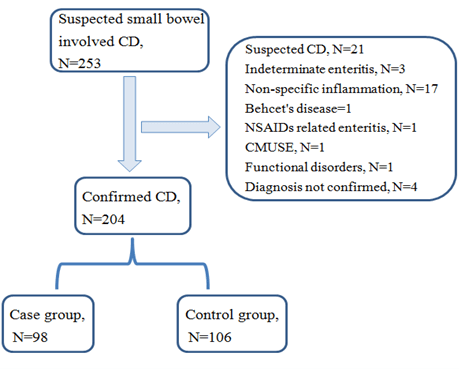
From November 2013 to August 2018, 253 patients suspected to have small bowel involved CD underwent DBE, and 204 patients (154 males and 50 females) confirmed with CD (98 in case group and 106 in control group) were enrolled in this study (Figure 1 shows the selection flow chart).
Figure 1: Flow chart of patients selection.
CD: Crohn’s Disease; NSAIDs: Non-Steroidal Anti-Inflammatory Drugs; CMUSE: Cryptogenic Multifocal Ulcerous Stenosing Enteritis.
II Baseline Characteristics
The median age of all enrolled patients was 26.5 (13.8) years old, and there was no statistical difference in sex ratio or body mass index (BMI) between the case and control group. The most common chief complaints were abdominal pain, diarrhea, and weight loss, other symptoms including gastrointestinal bleeding, fever, perianal symptoms etc. A higher proportion of patients in the case group had abdominal pain (72.45% vs 58.49%, P=0.037), while a higher proportion of patients in the control group had a diarrhea (30.19% VS 16.33%, P=0.02) and perianal diseases (34.91% vs 14.29%, P=0.001). Eighteen (8.82%) patients had appendectomy, and 31 (15.2%) patients had bowel resection before this DBE procedure, of which, 28 patients had small bowel resection, and 3 patients had colon resection. More patients in the case group had a history of bowel resection (26.53% vs 4.72%, P<0.001). The reasons for bowel resection were: perforation (10 patients), stricture (12 patients), intestinal fistula or peritoneal abscess (3 patients), massive hemorrhage (3 patients), abdominal exploration during appendectomy (1 patient), to clarify the diagnosis (2 patients). There were slight differences in Montreal classifications between the two groups: the control group had more patients with B1+P (29.25% vs 13.27%, P=0.006), while the case group had more patients with B2 (34.69% vs 16.04%, P=0.002). The baseline characteristics of the patients are shown in (Table 1).
Table 1: Baseline characteristics of the patients with small
bowel involved Crohn’s disease.
|
|
All (n=204) |
Case group (n=98) |
Control group (n=106) |
P value |
|
Age (years) |
26.5 (13.8) |
28.5 (13) |
25 (14) |
0.032 |
|
Sex: male (n, %) |
154 (75.49) |
72 (73.47) |
82 (77.36) |
0.519 |
|
BMI (kg/m2)* |
19.34 (4) |
19.68 (4.1) |
19.14 (4.08) |
0.27 |
|
Chief complaints (n, %) |
|
|
|
|
|
Abdominal pain |
133 (65.2) |
71 (72.45) |
62 (58.49) |
0.037 |
|
Diarrhea |
48 (23.53) |
16 (16.33) |
32 (30.19) |
0.02 |
|
GI bleeding |
31 (15.2) |
14 (14.29) |
17 (16.04) |
0.728 |
|
Weight loss |
46 (22.55) |
19 (19.39) |
27 (25.47) |
0.299 |
|
Fever |
18 (8.82) |
8 (8.16) |
10 (9.43) |
0.749 |
|
Perianal symptoms |
51 (25) |
14 (14.29) |
37 (34.91) |
0.001 |
|
Others |
6 (2.94) |
5 (5.1) |
1 (0.94) |
0.180 |
|
Appendectomy (n, %) |
18 (8.82) |
10 (10.2) |
8 (7.55) |
0.504 |
|
Bowel resection (n, %) |
31 (15.2) |
26 (26.53) |
5 (4.72) |
<0.001 |
|
Montreal classifications |
|
|
|
|
|
A1 |
30 (14.71) |
10 (10.2) |
20 (18.87) |
0.081 |
|
A2 |
145 (71.08) |
71 (72.45) |
74 (69.81) |
0.678 |
|
A3 |
29 (14.22) |
17 (17.35) |
12 (11.32) |
0.218 |
|
B1 |
34 (16.67) |
16 (16.33) |
18 (16.98) |
0.9 |
|
B2 |
51 (25) |
34 (34.69) |
17 (16.04) |
0.002 |
|
B3 |
21 (10.29) |
14 (14.29) |
7 (6.6) |
0.071 |
|
B1+P |
44 (21.57) |
13 (13.27) |
31 (29.25) |
0.006 |
|
B2+P |
40 (19.61) |
15 (15.31) |
25 (23.58) |
0.137 |
|
B3+P |
14 (6.86) |
6 (6.12) |
8 (7.55) |
0.688 |
|
L1 |
66 (32.35) |
66 (67.35) |
0 (0) |
- |
|
L3 |
69 (33.82) |
0 (0) |
69 (65.09) |
- |
|
L4 |
1 (0.49) |
1 (1.02) |
0 (0) |
- |
|
L1+4 |
31 (15.2) |
31 (31.63) |
0 (0) |
- |
|
L3+4 |
37 (18.14) |
0 (0) |
37 (34.91) |
- |
|
Follow-up period |
37.8 (39.9) |
33.1 (30.6) |
39.3 (47.4) |
0.210 |
BMI: Body Mass Index; GI: Gastrointestinal.
*Data missed in 24 patients.
III Laboratory and Endoscopy Characteristics
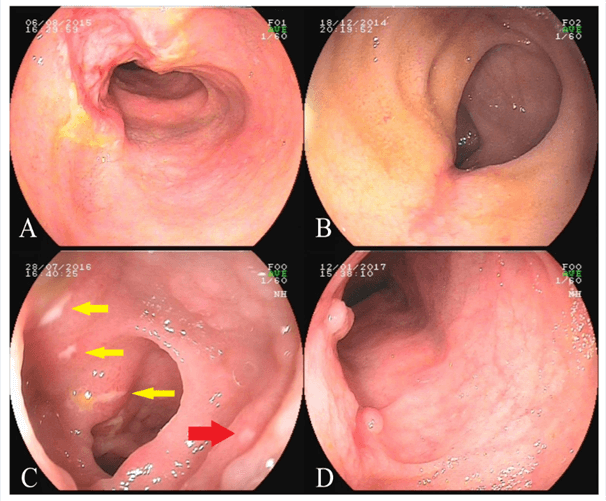
The haemoglobin levels and percentage of patients with anemia had no difference between the two groups, while more patients in the control group had elevated platelet (44.34% vs 27.55%, P=0.011) and erythrocyte sedimentation rate [ESR, (51.89% VS 36.73%, P=0.03)]. As for elevated white blood cells, albumin, and fecal calprotectin levels, there were no differences. In the process of DBE, pseudopolyp or nodular change (41.51% vs 17.35%, P<0.001) and aphthous ulcer (11.32% VS 1.02%, P=0.003) were more common in the control group, while stricture was more common in the case group (33.67% VS 20.75%, P=0.038). Figure 2 shows the endoscopy images for the small bowel involved CD, and the laboratory and endoscopy characteristics of the patients are shown in (Table 2).
Table 2: Laboratory, radiology, and endoscopy characteristics
of the patients with small bowel involved Crohn’s disease.
|
|
All (n=204) |
Case group (n=98) |
Control group (n=106) |
P value |
|
Laboratory findings |
|
|
|
|
|
Hb (g/L) |
121 (24) |
120 (25) |
122 (24.25) |
0.425 |
|
Anemia (n, %) |
80 (39.22) |
36 (36.73) |
44 (41.51) |
0.451 |
|
PLT (*10^9/L) |
271 (102) |
256.5 (95) |
280.5 (111) |
0.174 |
|
PLT>300*10^9/L |
74 (36.27) |
27 (27.55) |
47 (44.34) |
0.011 |
|
WBC (*10^9/L) |
6.28 (3.7) |
5.64 (3.11) |
7.17 (3.82) |
<0.001 |
|
WBC>10*10^9/L |
30 (14.71) |
12 (12.24) |
18 (16.98) |
0.34 |
|
N% |
0.64 (0.12) |
0.63 (0.15) |
0.65 (0.14) |
0.271 |
|
ESR (mm/h) # |
18 (26) |
15 (19.5) |
20.5 (29.5) |
0.012 |
|
ESR>20mm/h (n, %)# |
91 (45.73) |
36 (37.11) |
55 (53.92) |
0.017 |
|
Albumin (g/L) |
40.17 (5.56) |
40.50 (5.35) |
40.07 (6.05) |
0.594 |
|
Hypoalbuminemia (n, %) |
32 (15.69) |
14 (14.29) |
18 (16.98) |
0.597 |
|
Fecal calprotectin (μg/g)
* |
902.97 (1073.84) |
886.34 (1584.21) |
945.11 (878.41) |
0.925 |
|
CTE/MRE findings※ |
|
|
|
|
|
Thickest bowel wall (mm) |
8 (4) |
8 (5) |
8 (4) |
0.089 |
|
Thickest bowel
wall>7mm(n, %) |
106 (57.61) |
47 (51.65) |
59 (63.44) |
0.106 |
|
Endoscopy findings (n,
%) |
|
|
|
|
|
Pseudopolyp or nodular change |
61 (29.9) |
17 (17.35) |
44 (41.51) |
<0.001 |
|
Erosion |
16 (7.84) |
7 (7.14) |
9 (8.49) |
0.721 |
|
Ulcer |
163 (79.9) |
77 (78.57) |
86 (81.13) |
0.648 |
|
Longitudinal ulcer |
94 (46.08) |
43 (43.88) |
51 (48.11) |
0.544 |
|
Irregular ulcer |
104 (50.98) |
45 (45.92) |
59 (55.66) |
0.164 |
|
Aphthous ulcer |
13 (6.37) |
1 (1.02) |
12 (11.32) |
0.003 |
|
Stricture |
55 (26.96) |
33 (33.67) |
22 (20.75) |
0.038 |
|
Hyperemia and edema |
45 (22.06) |
23 (23.47) |
22 (20.75) |
0.64 |
Hb: Haemoglobin; PLT:
Platelet; WBC: White Blood Cell; N: Neutrophile granulocyte; ESR: Erythrocyte
Sedimentation Rate; CTE: Computed Tomography Enteroscopy; MRE: Magnetic
Resonance Enterography.
#Data missed in 5 patients; *data missed in
167 patients; ※data missed in 20 patients.
Figure 2: Endoscopy images for the small bowel involved Crohn’s disease: A) wide longitudinal ulcer; B) longitudinal ulcer in healing phase; C) irregular ulcer (yellow arrows) and aphthous ulcer (red arrow); D) Pseudopolyps and scar.
IV Use of DBE in Small Bowel Involved CD
The DBE was of vital importance in the diagnosis, evaluation and endoscopic treatment in small bowel involved CD. In 149 patients (73.04%), DBE helped to make the final diagnosis of CD (among which hemostasis with endotherapy using metal clips was also performed in one patient). Treatment regimens were changed in 35 patients (17.16%) has after evaluated by DBE (among which balloon dilation and capsule endoscopy was also retrieved in one patient), while no change of treatment regimen was made in 20 patients (9.8%) post-DBE.
V Medical Therapy of the Patients
Table 3 shows the medical therapy of the patients with small bowel involved CD after the DBE procedure; the two groups had no obvious statistical difference, except for the use of infliximab. It is worth noting that there were some other factors contributing to the choice of medical therapy choice, such as the economic status of the patients, so we must interpret the results cautiously.
Table 3: Medical therapy of the patients with small bowel
involved Crohn’s disease.
|
Medical therapy* |
All (n=204) |
Case group (n=98) |
Control group (n=106) |
P value |
|
Infliximab/+ AZA/ 6-MP/
EN |
54 (27) |
18 (18.75) |
36 (34.62) |
0.012 |
|
Corticosteroids/+ AZA/ 6-MP/ Thal/ 5-ASA/ EN |
42 (21) |
19 (19.79) |
23 (22.12) |
0.687 |
|
AZA or 6-MP/+ Thal/
5-ASA/ EN |
33 (16.5) |
20 (20.83) |
13 (12.5) |
0.113 |
|
Thal /+ 5-ASA/ EN |
14 (7) |
10 (10.42) |
4 (3.85) |
0.069 |
|
EN/+ 5-ASA |
26 (13) |
16 (16.67) |
10 (9.62) |
0.138 |
|
5-ASA /+ MTX |
29 (14.5) |
12 (12.5) |
17 (16.35) |
0.44 |
|
Adalimumab |
1 (0.5) |
1 (1.04) |
0 (0) |
0.968 |
|
Anti-tuberculosis
therapy# |
1 (0.5) |
0 (0) |
1 (0.96) |
1 |
* Four
patients refused to receive treatment; # the patient received
surgery, and the pathology showed a combination with tuberculosis infection.
AZA, azathioprine; 6-MP,
6- mercaptopurine, EN, enteral nutrition; Thal, thalidomide; 5-ASA, 5-aminosalicylic
acid; MTX, methotrexate.
VI Follow-Up Results
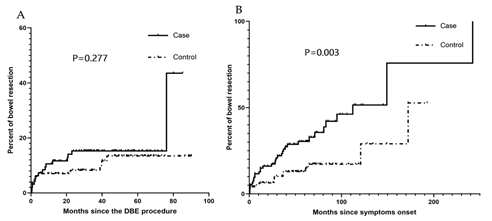
The patients were followed up for a median of 37.8 (39.9) months, with no difference in the two groups. Six patients (2.94%) got lost during follow-up, and 1 patient died of acute myocardial infarction. Twenty-six patients (13.13%, with 15 in the case group and 11 in the control group) underwent bowel resection. Among which, 4 patients (3 in the case group and 1 in the control group) had a history of bowel resection before this DBE procedure and underwent another bowel resection during follow-up. Kaplan-Meier curves showed that (Figure 3), the two groups had no statistical difference in the risk of bowel resection since the DBE procedure (P=0.277), while more patients in the case group suffered from bowel resection since the onset of the symptoms (P=0.003). Multivariate analysis further pointed out that (Table 4), diagnosis at > 40-year-old (HR 10.548, 95%CI 1.098-101.319, P=0.041), thickest bowel wall>7mm (HR 6.299, 95%CI 1.871-21.212, P=0.003), stricture found in DBE (HR 3.145, 95%CI 1.423-6.592, P=0.005) were risk factors for bowel resection in the patients with small bowel involved CD who underwent DBE, while patients with irregular ulcer (HR 0.295, 95%CI 0.123-0.708, P=0.006), received Infliximab (HR 0.138, 95%CI 0.028-0.685, P=0.015), corticosteroids (HR 0.118, 95%CI 0.029-0.487, P=0.003), or enteral nutrition (HR 0.308, 95%CI 0.105-0.902, P=0.032) after DBE may have a lower risk for bowel resection.
Figure 3: Kaplan-Meier curves showing the probability of bowel resection in isolated small-bowel Crohn’s disease patients (case group) and small bowel + colon involved Crohn’s disease patients (control group): A) since the double balloon enteroscopy (DBE) procedure, B) since the symptoms onset.
Table 4: Variables associated with bowel resection of the
patients with small bowel involved Crohn’s disease who underwent DBE.
|
|
Univariate analysis |
Multivariate analysis |
||
|
|
HR |
P |
HR |
P |
|
Groups (Reference: case) |
0.646 (0.295-1.413) |
0.274 |
- |
0.23 |
|
SEX(Reference: male) |
1.357 (0.590-3.124) |
0.472 |
Not included |
- |
|
BMI (Reference:
<17.5) |
|
|
Not included |
- |
|
17.5-25 |
1.358 (0.153-12.085) |
0.784 |
- |
- |
|
≥25 |
1.755 (0.225-13.721) |
0.592 |
- |
- |
|
Age at diagnosis
(Reference: A1) |
|
|
|
|
|
A2 |
3.596 (0.476-27.151) |
0.168 |
3.791 (0.446-32.255) |
0.223 |
|
A3 |
14.324 (1.793-114.452) |
0.012 |
10.548(1.098-101.319) |
0.041 |
|
Behavior at diagnosis
(Reference: B1) |
|
|
|
|
|
B2 |
4.875 (1.409-16.871) |
0.012 |
- |
0.226 |
|
B3 |
7.319 (1.936-27-678) |
0.003 |
- |
0.45 |
|
Perianal disease |
0.622(0.281-1.373) |
0.239 |
- |
0.811 |
|
Thickest bowel
wall>7mm |
3.634 (1.24-10.656) |
0.019 |
6.299 (1.871-21.212) |
0.003 |
|
Pseudopolyp or nodular
change |
0.994 (0.432-2.287) |
0.989 |
Not included |
- |
|
Erosion |
2.33 (0.803-6.764) |
0.12 |
Not included |
- |
|
Stricture |
4.080 (1.873-8.886) |
<0.001 |
3.145 (1.423-6.592) |
0.005 |
|
Hyperemia and edema |
1.102 (0.440-2.759) |
0.836 |
Not included |
- |
|
Longitudinal ulcer |
0.693 (0.314-1.530) |
0.365 |
Not included |
- |
|
Irregular ulcer |
0.344 (0.145-0.818) |
0.016 |
0.111 (0.037-0.335) |
<0.001 |
|
Aphthous ulcer |
1.059 (0.249-4.505) |
0.938 |
Not included |
- |
|
Anemia |
1.797 (0.831-3.889) |
0.137 |
- |
0.572 |
|
Elevated platelet level |
0.57 (0.238-1.361) |
0.205 |
Not included |
- |
|
Elevated WBC level |
1.045 (0.359-3.046) |
0.936 |
Not included |
- |
|
Elevated ESR level |
1.005 (0.464-2.178) |
0.99 |
Not included |
- |
|
Hypoalbuminemia |
1.588 (0.637-3.958) |
0.321 |
Not included |
- |
|
Regimen included in
medical therapy |
|
|
|
|
|
Infliximab |
0.199 (0.047-0.840) |
0.028 |
0.138 (0.028-0.685) |
0.015 |
|
AZA/ 6-MP |
1.164 (0.525-2.583) |
0.708 |
- |
0.209 |
|
Corticosteroids |
0.451 (0.135-1.504) |
0.195 |
0.118 (0.029-0.487) |
0.003 |
|
Enteral nutrition |
2.104 (0.913-4.848) |
0.081 |
0.308 (0.105-0.902) |
0.032 |
DBE: Double-Balloon Enteroscopy; WBC: White Blood Cell; ESR: Erythrocyte Sedimentation Rate; AZA: Azathioprine;
6-MP: 6-Mercaptopurine.
Discussion
Our study found that there were some differences between the isolated small-bowel CD patients (case group) and small bowel + colon involved CD patients (control group). Compared with the control group, a higher proportion of patients in the case group had abdominal pain, history of bowel resection, and stricturing behaviour, while a lower proportion had diarrhea, perianal diseases, non-stricturing and non-penetrating behaviour, elevated platelet, and ESR level, pseudopolyp, or nodular change, and aphthous ulcer. As for the probability of bowel resection, the two groups had no statistical difference since the DBE procedure, but the case group had a higher probability of bowel resection since symptoms onset. Furthermore, diagnosis at > 40-year-old, thickest bowel wall>7mm, and stricture found in DBE were risk factors for bowel resection, while patients with irregular ulcer, received infliximab, corticosteroids or enteral nutrition after DBE may have a lower risk.
Du et al. also studied the characteristics and prognosis of isolated small-bowel Crohn’s disease, which was similar to our results [7]. They found that a higher percentage of patients with isolated small-bowel CD had stricture, lower platelet, and erythrocyte sedimentation rate, though their data suggested that isolated small-bowel CD patients had a higher risk for intestinal resection surgery. As mentioned above, our study showed no difference in case and control group regarding the risk for bowel resection, which may result from different studies’ design and methods. Firstly, Du et al. selected patients who had undergone CE, ileocolonoscopy, and enhanced abdominal CT, however the CE findings implicating CD could be rather non-specific, and the European Society of Gastrointestinal Endoscopy guidelines recommended device-assisted enteroscopy with small bowel biopsy in patients suspected to have small bowel involved CD [11-13]. In our center, DBE was usually recommended if CE or enhanced abdominal CT indicated small bowel CD; thus, we chose the patients with confirmation of DBE. Secondly, Du et al. selected newly diagnosed patients, and their median follow-up time was longer (55.2 months), while we also included those who had been diagnosed with CD for a long time, and a certain part of these patients (especially in the isolated small bowel CD group) already had surgery history before this DBE, so this might explain why the risk for surgery for isolated small bowel CD since DBE was lower in our study. Nevertheless, it was unlikely to influence the evaluation of the risk since symptoms onset. Thirdly, they did not identify the risk factors for surgery. We used Cox regression model to analyse the time-to-event data, and to find risk factors for surgery. There were some other differences between our study and Du et al.’s study; for example, Du et al. pointed out that, isolated small bowel CD had a lower Harvey-Bradshaw index but a higher Lewis score, while we couldn’t obtain these data, which was one of our limitations.
The image captured by CE was random, resulting in higher negative predictive value and lower specificity, and it failed in obtaining tissues to have pathology confirmation [13]. While compared with CT, DBE was superior in detecting superficial lesions such as erosions and aphthous ulcers, and that’s why we chose patients confirmed by DBE [14, 15]. On the other hand, we also realized that the risk for bowel resection might be underestimated during follow-up if we chose the patients confirmed by DBE. In order to make up for this defect, we drew the Kaplan-Meier curves to demonstrate the risk for bowel resection since the symptom onset, which indicated that isolated small bowel CD indeed had a higher risk, which was in accordance with previous studies [7, 16].
Multivariate Cox regression analysis indicated that, diagnosis at > 40-year-old, thickest bowel wall>7mm, and stricture found in DBE were risk factors for bowel resection since the DBE procedure. Studies evaluating the risk factors for bowel resection for small bowel involved CD were rare, but there were some studies that discussed the risk factors for CD. Chen et al. demonstrated that, the risk for surgery was lower in patients <16-year-old at diagnosis, and the risk increased in patients with stricturing type [17]. When performing the multivariate analysis, the behaviour of the disease at diagnosis was also a risk factors in some steps. However, we did not include it in the final formula, given that it might have interaction with stricture found in DBE. Anyway, it still supported that, stricturing behaviour was risk factor. Rispo et al. also showed that, Simple Endoscopic Score for CD (SES-CD)≥9, bowel wall thickness≥7 mm, small bowel CD extension at bowel sonography≥33 cm, and stricturing/penetrating behaviour were independent factors for surgery at 1 year [18]. Our results were close to theirs, but we lacked the data in bowel sonography. There were other factors indicating the probability for surgery, such as jejunum involvement and perianal lesions at diagnosis [19]. In the future, studies comprehensively including all the possible risk factors and adjusting for confounders is needed to give an authoritative voice.
Use of Infliximab, corticosteroids, and enteral nutrition were protective factors for surgery in the small bowel involved CD. In consistency with previous studies, anti-tumor necrosis factor (anti-TNF) therapy corresponded with lower risk for surgery among patients with inflammatory-type CD, and a large population-based cohort study also concluded that, earlier use of biologic therapy in CD patients could decrease the risk of intestinal surgery [20, 21]. Safroneeva et al. also emphasized the importance of early treatment with immunomodulators or anti-TNF regimens, as they showed that, initiating these drugs< 2 years of CD diagnosis was associated with reduced risk of developing bowel strictures [22]. Though we did not prove the use of azathioprine (AZA)/6-mercaptopurine (6-MP) could reduce the risk for surgery, the above studies all implied that adopting a top-down or accelerated step-up treatment strategy may be effective at reducing the risk for bowel resection, and we believed this applied to the small bowel involved CD well [21].
Our data showed that, patients with irregular ulcers might have a lower risk for bowel resection. The possible reasons could be as follows. First, the irregular ulcer was mostly small in size and shallow. Second, the clinicians have adjusted the treatment regimens based on the findings of DBE, and maybe this type of ulcers responded well, reducing the risk of surgery. Based on others’ findings, we believed that Lewis score could possibly be an important risk factor in predicting bowel resection, which needed to be further verified [7]. Overall, early diagnosis and comprehensive evaluation was very important in the treatment of small bowel involved CD.
There were some limitations in our study. First, though with the exhausted search for EMR, some information still could not be obtained due to its retrospective nature, such as Harvey-Bradshaw index, Crohn’s disease activity index, Lewis score and so on. Second, the median follow-up time was not long enough, so the number of the patients who received surgery since DBE procedure was relatively small; moreover, some information such as the Lewis score lacked, making the identification of risk factors difficult. Third, we only compared the risk for bowel resection, while the medical therapeutic effect, mucous membrane healing, recurrence rate was not compared, which should be taken into consideration in future studies. Hence, well-designed prospective study with long-term follow-up is needed to confirm our findings.
In conclusion, the isolated small-bowel CD was concealed and would be easily overlooked by clinicians, which mainly manifested as abdominal pain and possibly without significant changes in laboratory results. However, the probability for surgery was higher, if the symptoms were obvious, as more patients in the case group would suffer from bowel resection since symptoms onset. On the other hand, the prognosis might not be inferior to small bowel + colon involved CD if the disease could be diagnosed, evaluated comprehensively, and treated appropriately in time.
Conflicts of Interest
None.
Article Info
Article Type
Research ArticlePublication history
Received: Mon 19, Apr 2021Accepted: Tue 04, May 2021
Published: Sat 29, May 2021
Copyright
© 2023 Min Zhi. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2021.05.03
Author Info
Yi Lu Xianhua Zhuo Lu Chen Jiachen Sun Yanan Liu Min Zhi
Corresponding Author
Min ZhiGuangdong Provincial Key Laboratory of Colorectal and Pelvic Floor Diseases, The Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou, P.R. China
Figures & Tables
Table 1: Baseline characteristics of the patients with small
bowel involved Crohn’s disease.
|
|
All (n=204) |
Case group (n=98) |
Control group (n=106) |
P value |
|
Age (years) |
26.5 (13.8) |
28.5 (13) |
25 (14) |
0.032 |
|
Sex: male (n, %) |
154 (75.49) |
72 (73.47) |
82 (77.36) |
0.519 |
|
BMI (kg/m2)* |
19.34 (4) |
19.68 (4.1) |
19.14 (4.08) |
0.27 |
|
Chief complaints (n, %) |
|
|
|
|
|
Abdominal pain |
133 (65.2) |
71 (72.45) |
62 (58.49) |
0.037 |
|
Diarrhea |
48 (23.53) |
16 (16.33) |
32 (30.19) |
0.02 |
|
GI bleeding |
31 (15.2) |
14 (14.29) |
17 (16.04) |
0.728 |
|
Weight loss |
46 (22.55) |
19 (19.39) |
27 (25.47) |
0.299 |
|
Fever |
18 (8.82) |
8 (8.16) |
10 (9.43) |
0.749 |
|
Perianal symptoms |
51 (25) |
14 (14.29) |
37 (34.91) |
0.001 |
|
Others |
6 (2.94) |
5 (5.1) |
1 (0.94) |
0.180 |
|
Appendectomy (n, %) |
18 (8.82) |
10 (10.2) |
8 (7.55) |
0.504 |
|
Bowel resection (n, %) |
31 (15.2) |
26 (26.53) |
5 (4.72) |
<0.001 |
|
Montreal classifications |
|
|
|
|
|
A1 |
30 (14.71) |
10 (10.2) |
20 (18.87) |
0.081 |
|
A2 |
145 (71.08) |
71 (72.45) |
74 (69.81) |
0.678 |
|
A3 |
29 (14.22) |
17 (17.35) |
12 (11.32) |
0.218 |
|
B1 |
34 (16.67) |
16 (16.33) |
18 (16.98) |
0.9 |
|
B2 |
51 (25) |
34 (34.69) |
17 (16.04) |
0.002 |
|
B3 |
21 (10.29) |
14 (14.29) |
7 (6.6) |
0.071 |
|
B1+P |
44 (21.57) |
13 (13.27) |
31 (29.25) |
0.006 |
|
B2+P |
40 (19.61) |
15 (15.31) |
25 (23.58) |
0.137 |
|
B3+P |
14 (6.86) |
6 (6.12) |
8 (7.55) |
0.688 |
|
L1 |
66 (32.35) |
66 (67.35) |
0 (0) |
- |
|
L3 |
69 (33.82) |
0 (0) |
69 (65.09) |
- |
|
L4 |
1 (0.49) |
1 (1.02) |
0 (0) |
- |
|
L1+4 |
31 (15.2) |
31 (31.63) |
0 (0) |
- |
|
L3+4 |
37 (18.14) |
0 (0) |
37 (34.91) |
- |
|
Follow-up period |
37.8 (39.9) |
33.1 (30.6) |
39.3 (47.4) |
0.210 |
BMI: Body Mass Index; GI: Gastrointestinal.
*Data missed in 24 patients.
Table 2: Laboratory, radiology, and endoscopy characteristics
of the patients with small bowel involved Crohn’s disease.
|
|
All (n=204) |
Case group (n=98) |
Control group (n=106) |
P value |
|
Laboratory findings |
|
|
|
|
|
Hb (g/L) |
121 (24) |
120 (25) |
122 (24.25) |
0.425 |
|
Anemia (n, %) |
80 (39.22) |
36 (36.73) |
44 (41.51) |
0.451 |
|
PLT (*10^9/L) |
271 (102) |
256.5 (95) |
280.5 (111) |
0.174 |
|
PLT>300*10^9/L |
74 (36.27) |
27 (27.55) |
47 (44.34) |
0.011 |
|
WBC (*10^9/L) |
6.28 (3.7) |
5.64 (3.11) |
7.17 (3.82) |
<0.001 |
|
WBC>10*10^9/L |
30 (14.71) |
12 (12.24) |
18 (16.98) |
0.34 |
|
N% |
0.64 (0.12) |
0.63 (0.15) |
0.65 (0.14) |
0.271 |
|
ESR (mm/h) # |
18 (26) |
15 (19.5) |
20.5 (29.5) |
0.012 |
|
ESR>20mm/h (n, %)# |
91 (45.73) |
36 (37.11) |
55 (53.92) |
0.017 |
|
Albumin (g/L) |
40.17 (5.56) |
40.50 (5.35) |
40.07 (6.05) |
0.594 |
|
Hypoalbuminemia (n, %) |
32 (15.69) |
14 (14.29) |
18 (16.98) |
0.597 |
|
Fecal calprotectin (μg/g)
* |
902.97 (1073.84) |
886.34 (1584.21) |
945.11 (878.41) |
0.925 |
|
CTE/MRE findings※ |
|
|
|
|
|
Thickest bowel wall (mm) |
8 (4) |
8 (5) |
8 (4) |
0.089 |
|
Thickest bowel
wall>7mm(n, %) |
106 (57.61) |
47 (51.65) |
59 (63.44) |
0.106 |
|
Endoscopy findings (n,
%) |
|
|
|
|
|
Pseudopolyp or nodular change |
61 (29.9) |
17 (17.35) |
44 (41.51) |
<0.001 |
|
Erosion |
16 (7.84) |
7 (7.14) |
9 (8.49) |
0.721 |
|
Ulcer |
163 (79.9) |
77 (78.57) |
86 (81.13) |
0.648 |
|
Longitudinal ulcer |
94 (46.08) |
43 (43.88) |
51 (48.11) |
0.544 |
|
Irregular ulcer |
104 (50.98) |
45 (45.92) |
59 (55.66) |
0.164 |
|
Aphthous ulcer |
13 (6.37) |
1 (1.02) |
12 (11.32) |
0.003 |
|
Stricture |
55 (26.96) |
33 (33.67) |
22 (20.75) |
0.038 |
|
Hyperemia and edema |
45 (22.06) |
23 (23.47) |
22 (20.75) |
0.64 |
Hb: Haemoglobin; PLT:
Platelet; WBC: White Blood Cell; N: Neutrophile granulocyte; ESR: Erythrocyte
Sedimentation Rate; CTE: Computed Tomography Enteroscopy; MRE: Magnetic
Resonance Enterography.
#Data missed in 5 patients; *data missed in
167 patients; ※data missed in 20 patients.
Table 3: Medical therapy of the patients with small bowel
involved Crohn’s disease.
|
Medical therapy* |
All (n=204) |
Case group (n=98) |
Control group (n=106) |
P value |
|
Infliximab/+ AZA/ 6-MP/
EN |
54 (27) |
18 (18.75) |
36 (34.62) |
0.012 |
|
Corticosteroids/+ AZA/ 6-MP/ Thal/ 5-ASA/ EN |
42 (21) |
19 (19.79) |
23 (22.12) |
0.687 |
|
AZA or 6-MP/+ Thal/
5-ASA/ EN |
33 (16.5) |
20 (20.83) |
13 (12.5) |
0.113 |
|
Thal /+ 5-ASA/ EN |
14 (7) |
10 (10.42) |
4 (3.85) |
0.069 |
|
EN/+ 5-ASA |
26 (13) |
16 (16.67) |
10 (9.62) |
0.138 |
|
5-ASA /+ MTX |
29 (14.5) |
12 (12.5) |
17 (16.35) |
0.44 |
|
Adalimumab |
1 (0.5) |
1 (1.04) |
0 (0) |
0.968 |
|
Anti-tuberculosis
therapy# |
1 (0.5) |
0 (0) |
1 (0.96) |
1 |
* Four
patients refused to receive treatment; # the patient received
surgery, and the pathology showed a combination with tuberculosis infection.
AZA, azathioprine; 6-MP,
6- mercaptopurine, EN, enteral nutrition; Thal, thalidomide; 5-ASA, 5-aminosalicylic
acid; MTX, methotrexate.
Table 4: Variables associated with bowel resection of the
patients with small bowel involved Crohn’s disease who underwent DBE.
|
|
Univariate analysis |
Multivariate analysis |
||
|
|
HR |
P |
HR |
P |
|
Groups (Reference: case) |
0.646 (0.295-1.413) |
0.274 |
- |
0.23 |
|
SEX(Reference: male) |
1.357 (0.590-3.124) |
0.472 |
Not included |
- |
|
BMI (Reference:
<17.5) |
|
|
Not included |
- |
|
17.5-25 |
1.358 (0.153-12.085) |
0.784 |
- |
- |
|
≥25 |
1.755 (0.225-13.721) |
0.592 |
- |
- |
|
Age at diagnosis
(Reference: A1) |
|
|
|
|
|
A2 |
3.596 (0.476-27.151) |
0.168 |
3.791 (0.446-32.255) |
0.223 |
|
A3 |
14.324 (1.793-114.452) |
0.012 |
10.548(1.098-101.319) |
0.041 |
|
Behavior at diagnosis
(Reference: B1) |
|
|
|
|
|
B2 |
4.875 (1.409-16.871) |
0.012 |
- |
0.226 |
|
B3 |
7.319 (1.936-27-678) |
0.003 |
- |
0.45 |
|
Perianal disease |
0.622(0.281-1.373) |
0.239 |
- |
0.811 |
|
Thickest bowel
wall>7mm |
3.634 (1.24-10.656) |
0.019 |
6.299 (1.871-21.212) |
0.003 |
|
Pseudopolyp or nodular
change |
0.994 (0.432-2.287) |
0.989 |
Not included |
- |
|
Erosion |
2.33 (0.803-6.764) |
0.12 |
Not included |
- |
|
Stricture |
4.080 (1.873-8.886) |
<0.001 |
3.145 (1.423-6.592) |
0.005 |
|
Hyperemia and edema |
1.102 (0.440-2.759) |
0.836 |
Not included |
- |
|
Longitudinal ulcer |
0.693 (0.314-1.530) |
0.365 |
Not included |
- |
|
Irregular ulcer |
0.344 (0.145-0.818) |
0.016 |
0.111 (0.037-0.335) |
<0.001 |
|
Aphthous ulcer |
1.059 (0.249-4.505) |
0.938 |
Not included |
- |
|
Anemia |
1.797 (0.831-3.889) |
0.137 |
- |
0.572 |
|
Elevated platelet level |
0.57 (0.238-1.361) |
0.205 |
Not included |
- |
|
Elevated WBC level |
1.045 (0.359-3.046) |
0.936 |
Not included |
- |
|
Elevated ESR level |
1.005 (0.464-2.178) |
0.99 |
Not included |
- |
|
Hypoalbuminemia |
1.588 (0.637-3.958) |
0.321 |
Not included |
- |
|
Regimen included in
medical therapy |
|
|
|
|
|
Infliximab |
0.199 (0.047-0.840) |
0.028 |
0.138 (0.028-0.685) |
0.015 |
|
AZA/ 6-MP |
1.164 (0.525-2.583) |
0.708 |
- |
0.209 |
|
Corticosteroids |
0.451 (0.135-1.504) |
0.195 |
0.118 (0.029-0.487) |
0.003 |
|
Enteral nutrition |
2.104 (0.913-4.848) |
0.081 |
0.308 (0.105-0.902) |
0.032 |
DBE: Double-Balloon Enteroscopy; WBC: White Blood Cell; ESR: Erythrocyte Sedimentation Rate; AZA: Azathioprine;
6-MP: 6-Mercaptopurine.
CD: Crohn’s Disease; NSAIDs: Non-Steroidal Anti-Inflammatory Drugs; CMUSE: Cryptogenic Multifocal Ulcerous Stenosing Enteritis.
References
1.
Cosnes J, Gower
Rousseau C, Seksik P, Cortot A (2011) Epidemiology and natural history of
inflammatory bowel diseases. Gastroenterology 140: 1785-1794. [Crossref]
2.
Farmer RG, Hawk
WA, Turnbull RB Jr (1975) Clinical patterns in Crohn's disease: a statistical
study of 615 cases. Gastroenterology 68: 627-635. [Crossref]
3.
Molinié F, Gower
Rousseau C, Yzet T, Merle V, Grandbastien B et al. (2004) Opposite evolution in
incidence of Crohn's disease and ulcerative colitis in Northern France
(1988-1999). Gut 53: 843-848.
[Crossref]
4.
Higuero T, Merle
C, Thiéfin G, Coussinet S, Jolly D et al. (2004) Jejunoileal Crohn's disease: a
case-control study. Gastroenterol Clin Biol 28: 160-166. [Crossref]
5.
Attard TM, Horton
KM, DeVito K, Darbari A, Oliva-Hemker M et al. (2004) Pediatric jejunoileitis:
a severe Crohn's disease phenotype that requires intensive nutritional
management. Inflamm Bowel Dis 10: 357-360. [Crossref]
6.
Park SK, Yang SK,
Park SH, Park SH, Kim JW et al. (2013) Long-term prognosis of the jejunal
involvement of Crohn's disease. J Clin Gastroenterol 47: 400-408. [Crossref]
7.
Du J, Du H, Chen
H, Shen L, Zhang B et al. (2020) Characteristics and prognosis of isolated
small-bowel Crohn's disease. Int J Colorectal Dis 35: 69-75. [Crossref]
8.
Schulz C,
Mönkemüller K, Salheiser M, Bellutti M, Schütte K et al. (2014) Double-balloon
enteroscopy in the diagnosis of suspected isolated Crohn's disease of the small
bowel. Dig Endosc 26: 236-242.
[Crossref]
9.
Halloran BP,
Jamil LH, Lo SK, Reeson M, Vasiliauskas EA et al. (2020) Double-Balloon
Endoscopy in Crohn Disease: A Tertiary Referral Center Experience. Inflamm
Bowel Dis izaa287. [Crossref]
10. Ouyang Q, Tandon R, Goh KL, Pan GZ, Fock KM et al.
(2006) Management consensus of inflammatory bowel disease for the Asia-Pacific
region. J Gastroenterol Hepatol 21: 1772-1782. [Crossref]
11. Sidhu R, Brunt LK, Morley SR, Sanders DS, McAlindon ME
(2010) Undisclosed use of nonsteroidal anti-inflammatory drugs may underlie
small-bowel injury observed by capsule endoscopy. Clin Gastroenterol Hepatol
8: 992-995. [Crossref]
12. Saibeni S, Rondonotti E,
Iozzelli A, Spina L, Tontini GE et al. (2007)
Imaging of the small bowel in Crohn's disease: a review of old and new
techniques. World J Gastroenterol 13: 3279-3287. [Crossref]
13. Pennazio M, Spada C,
Eliakim R, Keuchel M, May A et al. (2015) Small-bowel capsule endoscopy and
device-assisted enteroscopy for diagnosis and treatment of small-bowel
disorders: European Society of Gastrointestinal Endoscopy (ESGE) Clinical
Guideline. Endoscopy 47: 352-376. [Crossref]
14. Huang Z, Liu X, Yang F, Wang G, Ge N et al. (2020)
Diagnostic efficacy of double-balloon enteroscopy in patients with suspected
isolated small bowel Crohn's disease. BMC Gastroenterol 20: 42. [Crossref]
15. Oshitani N, Yukawa T, Yamagami H, Inagawa M, Kamata N
et al. (2006) Evaluation of deep small bowel involvement by double-balloon
enteroscopy in Crohn's disease. Am J Gastroenterol 101: 1484-1489. [Crossref]
16. Attard TM, Horton KM, DeVito K, Darbari A,
Oliva-Hemker M et al. (2004) Pediatric jejunoileitis: a severe Crohn's disease
phenotype that requires intensive nutritional management. Inflamm Bowel Dis 10:
357-360. [Crossref]
17. Chen M, Yi F, Zhou F, Huang M, Li J et al. (2015) Risk
factors for initial surgery in patients with Crohn's disease in Central China. Surg
Today 45: 1002-1008. [Crossref]
18. Rispo A, Imperatore N,
Testa A, Bucci L, Luglio G et al. (2018) Combined Endoscopic/Sonographic-based Risk
Matrix Model for Predicting One-year Risk of Surgery: A Prospective
Observational Study of a Tertiary Centre Severe/Refractory Crohn's Disease
Cohort. J Crohns Colitis 12: 784-793. [Crossref]
19. Park Y, Cheon JH, Park YL, Ye BD, Kim YS et al. (2017)
Development of a Novel Predictive Model for the Clinical Course of Crohn's
Disease: Results from the CONNECT Study. Inflamm Bowel Dis 23:
1071-1079. [Crossref]
20. Nagata Y, Esaki M,
Moriyama T, Hirano A, Umeno J et al. (2019) Anti-tumor necrosis factor therapy decreases
the risk of initial intestinal surgery after diagnosis of Crohn's disease of
inflammatory type. J Gastroenterol 54: 330-338. [Crossref]
21. Jenkinson PW, Plevris N, Siakavellas S, Lyons M,
Arnott ID et al. (2020) Temporal Trends in Surgical Resection Rates and
Biologic Prescribing in Crohn's Disease: A Population-based Cohort Study. J
Crohns Colitis 14: 1241-1247. [Crossref]
22. Safroneeva E, Vavricka SR, Fournier N, Pittet V, Peyrin Biroulet L et al. (2015) Impact of the early use of immunomodulators or TNF antagonists on bowel damage and surgery in Crohn's disease. Aliment Pharmacol Ther 42: 977-989.
