A Survival Analysis of Chinese Patients with Gastrointestinal Mucosal Melanomas: A Single-Centre Retrospective Study
A B S T R A C T
Aim: Mucosal melanomas are highly malignant tumors that are of great interest for their aggressive behaviour and unfavourable prognosis, which could be the result of many reasons. In this paper, we describe a retrospective study performed to investigate the characteristics and prognoses of gastrointestinal mucosal melanomas in a Chinese population to help future clinicians recognize the prognosis of this disease.
Methods: We retrospectively studied 49 patients diagnosed with gastrointestinal mucosal melanomas. Multivariate analysis of prognosis and overall survival (OS) was performed using the Cox proportional hazards model.
Results: The multivariate analysis showed that the condition of the primary or metastatic tumor and tumor location were independent factors that affected the prognosis of patients with gastrointestinal mucosal melanomas. Patients with metastatic tumors had a better prognosis than patients with primary tumors, and tumors that occurred in the lower gastrointestinal tract had a better prognosis than those that occurred in the upper gastrointestinal tract.
Conclusion: Tumor location and the condition of the primary or metastatic tumor may be independent factors that affect the prognosis of patients with gastrointestinal mucosal melanomas.
Keywords
Gastrointestinal mucosal melanoma, melanoma, survival analysis
Introduction
Melanomas are highly malignant tumors that account for 1%~3% of all malignant cancers, and the majority of them are of cutaneous origin. There are approximately 132000 newly diagnosed cases and approximately 48000 patients who die annually from this disease worldwide. Only 0.8%~3.7% of melanomas are mucosal melanomas, which are not well understood, particularly with respect to those that occur in the mucosa of the gastrointestinal tract [1, 2]. Despite their rarity, mucosal melanomas are of great interest due to their aggressive behaviour and less favourable prognosis compared to cutaneous melanomas [2]. Moreover, melanomas of the gastrointestinal tract are mostly metastatic, presenting along with the primary tumor or years later [3]. The most frequent locations of metastasis are the small intestine, colon, and stomach. Primary mucosal melanoma arises in any site of the gastrointestinal tract, but it is most common in the anorectal (31.4% in the anal canal and 22.2% in the rectum) and oropharyngeal (32.8%) regions, while the oesophagus (5.9%), stomach (2.7%), small intestine (2.3%), gallbladder (1.4%) and large intestine (0.9%) are rare sites of origin [4].
A major challenge with mucosal melanomas is that well-established protocols for staging and treatments are lacking [5]. A staging system should be valid as a prognostic tool to target treatment in terms of overall survival (OS), but this tool has not yet been identified. In the seventh edition of the American Joint Committee on Cancer (AJCC) staging manual, the tumor-node-metastasis (TNM) staging system for mucosal melanomas of the head and neck was established, but the establishment of an appropriate staging system of mucosal melanomas in the gastrointestinal tract is still needed. Most of the staging systems currently used by clinicians are those that are used for more common malignancies of the gastrointestinal tract rather than those used specifically for mucosal melanomas [1, 6-8]. Thus, it is difficult for clinicians to quickly evaluate the prognosis of these patients through all the information they collect. Knowledge of which signs affect the prognosis of mucosal melanomas in the gastrointestinal tract may also promote the establishment of a commonly applied staging system.
In recent decades, a large amount of research has been dedicated to factors that possibly affect the OS of patients with mucosal melanomas, including the tumor position, depth of invasion and presence of metastasis [9]. Nevertheless, literature on specific elements such as pathological signs and therapeutic methods that affect the prognosis of mucosal melanomas in the gastrointestinal tract is limited. In this paper, we describe a retrospective study performed to investigate the characteristics and prognosis of gastrointestinal mucosal melanomas in a Chinese population to help future clinicians recognize the prognosis of this disease.
Materials and Methods
I Patients
From January 2010 to October 2020, 49 consecutive patients diagnosed with gastrointestinal mucosal melanomas in The Sixth Affiliated Hospital of Sun Yat-sen University (Guangdong Gastrointestinal Hospital) were retrospectively recruited for this study. The inclusion criteria were as follows: i) pathological confirmation of gastrointestinal mucosal melanomas via biopsy; ii) Chinese race; and iii) performance status (PS) grade ≤ 2.
II Data Collection
We collected the following data from our patients: sex, age, condition of the primary or metastatic tumor, tumor location, number of lesions, initial symptoms, distant metastasis, lymph node metastasis, expression of Melan A in tumor cells, and treatments they received. Tumors with no prior history of cutaneous, mucosal, or uveal melanoma were categorized as primary tumors, while tumors that did not meet these criteria were categorized as metastatic tumors. Tumor location represents the location of the lesion in the gastrointestinal tract when the patient was first diagnosed with gastrointestinal mucosal melanomas. The upper gastrointestinal tract includes the oral cavity, pharynx, oesophagus, stomach, and duodenum. The lower gastrointestinal tract includes the jejunum, ileum, caecum, vermiform appendix, colon, rectum, and anal canal.
The number of lesions, which was proven via the imaging data when the patients were first diagnosed with gastrointestinal mucosal melanomas, was classified as single or multiple (more than one lesion). Moreover, in our study of Melan A (immunohistochemical analysis), sections were incubated overnight at 4°C in humidified chambers with a primary antibody against Melan A (ZM-0398; ZSGB-BIO, Beijing, China) and then washed in phosphate-buffered saline (PBS) three times for 5 minutes. Next, sections were treated with a secondary antibody and then incubated with 3,3’-diaminobenzidine (DAB) chromogen. Finally, sections were washed in PBS and lightly counterstained with haematoxylin to reveal nuclei. A negative control was generated by replacing the specific primary antibody with nonimmune serum immunoglobulins.
Lymph node metastasis was defined via imaging data. Distant metastasis was defined as a gastrointestinal lesion that had metastasized through the blood vessels or lymph vessels to other organs and was determined from the patients’ imaging data. Having received treatments meant that the patients had received targeted treatments, including surgery and/or adjuvant therapy, rather than having received symptomatic treatments only or having not received any treatments. Furthermore, in this retrospective study, data such as patient characteristics that could not be certified were classified as ‘unspecified’.
III Follow-Up and Outcomes
The standard follow-up protocol in our study included a whole-body CT and physical examination every 3 months for 2 years, then every 6 months until 5 years, and then annually after the patients were diagnosed with gastrointestinal mucosal melanomas. The cut-off follow-up date of this study was 26 November 2020. However, 8 of the 49 patients rejected follow-up (censored). The OS was calculated from the time of pathologic diagnosis to the date of death from any cause or the date of the last follow-up.
IV Statistical Analyses
All analyses were performed with the Statistical Package for Social Sciences V24.0 (SPSS Inc., IL, USA). All data was collected as categorical variables and was described in the form of a number (percentage). Multivariate analyses of prognosis and OS were performed using the Cox proportional hazards model [10, 11]. All the tests were two-sided, and a p-value less than 0.05 was considered statistically significant.
Results
I Patient Characteristics
A summary of the clinical characteristics of the 49 patients with gastrointestinal mucosal melanomas we studied is shown in (Table 1). Generally, 19 (38.8%) patients were males, and 30 (61.2%) were females; the median age of our patients when they were first diagnosed with gastrointestinal mucosal melanomas was 58 (range from 29 to 81) years; 43 (87.8%) were primary tumors, while 6 (12.2%) were metastatic tumors, and all of the metastatic gastrointestinal mucosal melanomas of our patients metastasized from the skin; and 28 (57.1%) patients had a single tumor, while 7 (14.3%) had multiple lesions (14 were unspecified due to a lack of imaging data). Regarding the location of the lesions, most (49%) were located in the rectum, and the others were located in the anal canal (35%), small intestine (10%), and stomach (6%). In terms of treatments, 19 (38.8%) patients received only surgery, while 17 (34.7%) received both surgery and chemotherapy as adjuvant therapy. Moreover, one patient (2.0%) received only thermotherapy, and 12 (24.5%) did not receive any treatment for various reasons.
Table 1: Characteristics of patients with gastrointestinal
mucosal melanomas.
|
Characteristic |
|
Cases (%) |
|
Total |
|
49 |
|
Sex |
Male |
19 (38.8%) |
|
|
Female |
30 (61.2%) |
|
Age (years) |
≤60 |
25 (51.0%) |
|
|
>60 |
24 (49.0%) |
|
Primary or metastatic |
Primary |
43 (87.8%) |
|
|
Metastatic |
6 (12.2%) |
|
Tumor location |
Upper gastrointestinal tract |
8 (16.3%) |
|
|
Lower gastrointestinal tract |
41 (83.7%) |
|
Number of lesions |
Single |
28 (57.1%) |
|
|
Multiple |
7 (14.3%) |
|
|
Unspecified |
14 (28.6%) |
|
Initial symptoms |
Bleeding (haematochezia or haematemesis) |
21 (42.9%) |
|
|
Non-bleeding |
28 (57.1%) |
|
Distant metastasis |
Yes |
16 (32.7%) |
|
|
No |
18 (36.7%) |
|
|
Unspecified |
15 (30.6%) |
|
Lymph node metastasis |
Yes |
27 (55.1%) |
|
|
No |
8 (16.3%) |
|
|
Unspecified |
14 (28.6%) |
|
Melan A (immunohistochemical analysis) |
(+) |
33 (67.3%) |
|
|
(-) |
13 (26.5%) |
|
|
Unspecified |
3 (6.1%) |
|
Treatments |
No (had not received any treatment or had received only
palliative care) |
12 (24.5%) |
|
|
Only surgery |
19 (38.8%) |
|
|
Only chemotherapy |
1 (2.0%) |
|
|
Both surgery and chemotherapy |
17 (34.7%) |
II Overall Survival
The OS duration of our patients ranged from 2 months to 90 months, and the median OS was 22 months. However, 8 patients were lost to follow-up after diagnosis; thus, they were excluded from the survival analysis.
III Multivariate Survival Analyses of Risk Factors for OS
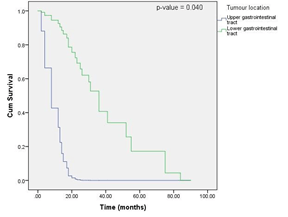
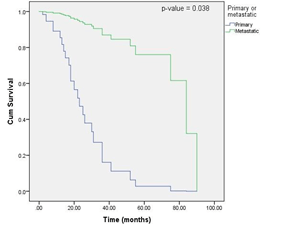
The multivariate analysis showed that the condition of the primary or metastatic tumor (p-value = 0.038) and tumor location (p-value = 0.040) were independent factors that affected the prognosis of patients with gastrointestinal mucosal melanomas. Patients with metastatic gastrointestinal mucosal melanomas had a better prognosis than those with primary tumors [hazard ratio (HR) = 13.062, median OS of primary cases = 30 months, median OS of metastatic cases = 55 months], and tumors that occurred in the lower gastrointestinal tract had a better prognosis than those that occurred in the upper gastrointestinal tract (HR = 14, median OS of cases in upper gastrointestinal tract = 30 months, median OS of cases in lower gastrointestinal tract = 38.850 months). However, according to our multivariate analysis, the factors sex, age, number of lesions, initial symptoms, distant metastasis, Melan A level, and treatment were not significantly associated with OS. The detailed results of the multivariate analysis are shown in (Table 2).
Table 2: Multivariate analyses of prognostic factors for
overall survival in patients with gastrointestinal mucosal melanomas.
|
Variable |
P-value |
Hazard ratio |
95.0% CI for hazard ratio |
||
|
Lower |
Upper |
||||
|
Sex |
Male |
- |
- |
- |
- |
|
|
Female |
0.615 |
0.734 |
0.220 |
2.446 |
|
Age (years) |
≤60 |
- |
- |
- |
- |
|
|
>60 |
0.617 |
0.777 |
0.289 |
2.088 |
|
Primary or metastatic |
Primary |
- |
- |
- |
- |
|
|
Metastatic |
0.038 |
13.062 |
1.160 |
147.127 |
|
Tumor location |
Upper gastrointestinal tract |
- |
- |
- |
- |
|
|
Lower gastrointestinal tract |
0.040 |
14.993 |
1.136 |
197.823 |
|
Number of lesions |
Single |
- |
- |
- |
- |
|
|
Multiple |
0.809 |
0.806 |
0.140 |
4.632 |
|
Initial symptoms |
Bleeding (haematochezia or haematemesis) |
- |
- |
- |
- |
|
|
Non-bleeding |
0.160 |
2.628 |
0.683 |
10.110 |
|
Distant metastasis |
Yes |
- |
- |
- |
- |
|
|
No |
0.660 |
0.729 |
0.178 |
2.988 |
|
Lymph node metastasis |
Yes |
- |
- |
- |
- |
|
|
No |
0.132 |
3.394 |
0.692 |
16.635 |
|
Melan A (immunohistochemical analysis) |
(+) |
- |
- |
- |
- |
|
|
(-) |
0.152 |
5.777 |
0.524 |
63.747 |
|
Treatments |
No (had not received any treatment or had received only
palliative care) |
- |
- |
- |
- |
|
|
Only surgery |
0.061 |
5.770 |
0.922 |
36.103 |
|
|
Only chemotherapy |
0.237 |
6.540 |
0.291 |
147.070 |
|
|
Both surgery and chemotherapy |
0.281 |
2.686 |
0.446 |
16.183 |
Table 3: Mean and median OS of gastrointestinal mucosal
melanoma patients with different characteristics.
|
Characteristic |
Mean OS
(months) |
Median OS
(months) |
|
|
Total |
|
38.395 |
30.000 |
|
Sex |
Male |
38.153 |
36.000 |
|
|
Female |
38.177 |
26.000 |
|
Age (years) |
≤60 |
43.414 |
36.000 |
|
|
>60 |
33.016 |
23.000 |
|
Primary or
metastatic |
Primary |
35.311 |
30.000 |
|
|
Metastatic |
50.600 |
55.000 |
|
Tumor location |
Upper
gastrointestinal tract |
29.208 |
14.000 |
|
|
Lower
gastrointestinal tract |
38.850 |
30.000 |
|
Number of
lesions |
Single |
40.111 |
36.000 |
|
|
Multiple |
22.400 |
20.000 |
|
|
Unspecified |
43.451 |
30.000 |
|
Initial
symptoms |
Bleeding
(haematochezia or haematemesis) |
23.930 |
22.000 |
|
|
Non-bleeding |
50.888 |
55.000 |
|
Distant
metastasis |
Yes |
33.193 |
26.000 |
|
|
No |
39.097 |
25.000 |
|
|
Unspecified |
43.451 |
30.000 |
|
Lymph node metastasis |
Yes |
32.972 |
31.000 |
|
|
No |
49.292 |
26.000 |
|
|
Unspecified |
43.451 |
30.000 |
|
Melan A
(immunohistochemical analysis) |
(+) |
29.796 |
30.000 |
|
|
(-) |
46.222 |
36.000 |
|
|
Unspecified |
39.500 |
20.000 |
|
Treatments |
No (had not
received any treatment or had received only palliative care) |
31.481 |
31.000 |
|
|
Only surgery |
35.604 |
20.000 |
|
|
Only
chemotherapy |
17.000 |
17.000 |
|
|
Both surgery
and chemotherapy |
50.393 |
36.000 |
The mean and median OS of gastrointestinal mucosal melanoma patients with different characteristics are shown in (Table 3). The OS curves according to the condition of the primary and metastatic tumors and different tumor locations were generated using the Cox proportional hazards model and are shown in (Figures 1 & 2).
Figure 1: OS curves according to tumor location (Cox proportional hazards model).
Figure 2: OS curves according to tumor type (primary or metastatic) (Cox proportional hazards model).
Discussion
Researchers have found that in patients with localized cutaneous melanomas, tumor thickness, the mitotic rate (histologically defined as mitoses/mm2), and ulceration are the most dominant prognostic factors [12]. However, another study showed that mucosal melanomas are very different from cutaneous melanomas, so these prognostic factors may not have the same effect in the mucosal melanomas, and the conclusion drawn by the former study may not be appropriate for mucosal melanomas [13]. Therefore, it is of great significance to study mucosal melanomas.
In this study, we retrospectively examined the correlation between the clinical characteristics and prognoses of Chinese patients with gastrointestinal mucosal melanomas to identify factors associated with OS. The condition of the primary or metastatic tumor and tumor location were found to be the independent factors that affected the prognosis of patients with gastrointestinal mucosal melanomas. With respect to the condition of the primary or metastatic tumor, another study showed that the prognosis was worse for primary intestinal melanomas, which tend to grow faster and more aggressively [14, 15]. We drew a similar conclusion in this study. But it still needs further study to find the reason for this. Certainly, when a mucosal melanoma is detected, a total body skin examination is paramount to rule out a primary cutaneous melanoma that has metastasized [8]. Therefore, we suggest that clinicians, especially when facing patients with melanomas, review the potential primary lesions at the location they were first discovered. A thorough examination of the potential primary lesions may also help future researchers to draw a more accurate result.
Moreover, we found that the tumor location in the gastrointestinal tract was an independent factor affecting the prognosis of patients, which is different from the conclusion drawn from a previous study of 444 German mucosal melanoma patients in which multivariate Cox regression could not validate tumor location as an independent risk factor [15]. Additionally, in another comparison of 706 mucosal melanomas from different anatomic sites, the researchers did not identify any significant survival differences when patients were matched for stage and prognostic and molecular factors [9]. Nevertheless, patients with mucosal melanomas all over the body, not just the gastrointestinal tract, were enrolled in both studies. When the scope of the study is confined to the gastrointestinal tract, there may be some differences. That is to say, the two studies are not specifically analysing the prognosis of cases in the upper and lower gastrointestinal tract, rather, they analysed melanomas in all parts of mucosa (though which also contains the gastrointestinal tract), and there is no separately analysed gastrointestinal mucosal melanoma [10, 15]. Thus, the differences between us are reasonable and understandable.
However, since we examined only 49 patients (the former study enrolled 121 cases in the gastrointestinal tract, while the latter study enrolled 226 cases), the small sample size may cause the potential bias [9, 15]. What is more, the patients involved in the former study were all in German population, and the latter study did not illustrate which population their cases came from. As all the patients in this study were collected from a Chinese population, the races and customs may also cause the differences in our conclusions. Further studies on a larger number of patients with gastrointestinal mucosal melanomas are needed to confirm the conclusion of our study. Other important epidemiological factors for many diseases, such as sex, age, number of lesions, initial symptoms, distant metastases, lymph node metastases, and immunohistochemical indexes, as well as the condition of treatments were found not to be independent prognostic factors of our patients.
A previous study demonstrated that sex was a significant prognostic factor, with the male sex being associated with worse OS than the female sex for mucosal melanomas in the gastrointestinal tract, which was similar to other published studies noting sex as an independent prognostic factor associated with both cutaneous and mucosal melanomas. It is notable that hormonal influences have been postulated to play a role in the sex effect [16]. Nevertheless, in this study, we did not find that sex was significantly associated with the OS of our patients. The small number of patients examined, and race differences may account for this difference.
It has been reported that the prognosis of patients with lymph node metastases from vaginal and anal melanomas is distinctly poor [9, 17-19]. Nevertheless, in our series, we concluded that lymph node metastasis was not an independent factor that significantly affected the prognosis of patients with gastrointestinal melanomas. Actually, the technique of lymphatic mapping and sentinel node biopsy is now gradually being used as the standard method for staging patients seemed to have a significant risk of clinically occult nodal metastases. This approach greatly facilitates the identification of nodal metastases relative to a clinical examination or imaging studies, and it has led to a dramatic shift in the prognosis of patients with melanoma metastatic to regional lymph nodes [9]. However, in our study, lymph node metastasis was confirmed through imaging data. Some of the patients who may have had distant lymph node metastasis may not have been correctly included in our study, which may have resulted in a bias of our multivariate analysis.
With regard to the immunohistochemical indexes that can indicate the expression of certain molecules in cells, this study was one of the few to analyse the association between the expression of certain immunohistochemical indexes and the OS of patients with gastrointestinal mucosal melanomas. Our study demonstrated that the expression of Melan A had no significant association with the OS of our patients. However, due to the inherent disadvantages of a retrospective study, we did not collect all of the immunohistochemical indicators of our patients. Further exploration is needed to determine whether other indicators are associated with the prognosis of patients with gastrointestinal mucosal melanomas. However, our study shows that immunohistochemical indicators may be associated with the prognosis of patients (since in our study, those without Melan A expression seemed to have longer OS to some extent) and thus provide clues for future studies. We believe that in the future exploration of gastrointestinal mucosal melanomas, we will obtain some significant results upon analysing immunohistochemical indicators.
The present study can also be criticized for the following limitations. We selected patients who received a pathological diagnosis of gastrointestinal mucosal melanomas. While some of the pathological sections were obtained from an outpatient consultation, we did not have complete data for certain variables potentially related to the prognosis of patients with gastrointestinal mucosal melanomas, such as tumor thickness and ulceration, which seem to be the dominant prognostic factors of cutaneous melanomas according to the staging system [12]. Moreover, the retrospective review of data is associated with inherent selection bias for treatment plans depending on the treatment providers and other disease factors. In addition, due to the loss of follow-up information, we did not obtain the OS data for some of our patients. These limitations may have influenced our results.
Moreover, melanoma is a disease with high ethnic variations [13]. Distinct racial and geographic differences are observed in mucosal melanomas, as they are in cutaneous melanomas [2]. There may be some differences between our study and others that may be attributed to unique environmental factors specific to geographic locations and variation in genetic and ethnic backgrounds. Whether our results can be applied to other regions and ethnic groups remains to be further explored. Although the sample size of our study was small and the results may have been affected by the various factors mentioned above, this study is expected to provide some clues for future exploration that will help clinicians improve the accuracy of judging prognosis.
Conclusion
In summary, our multivariate analysis showed that the condition of the primary or metastatic tumor and tumor location were independent factors that affected the prognosis of patients with gastrointestinal mucosal melanomas. Patients with metastatic gastrointestinal mucosal melanomas had a better prognosis than those with primary tumors, tumors that occurred in the lower gastrointestinal tract had a better prognosis than those that occurred in the upper gastrointestinal tract.
Ethical Approval
The authors state that they have followed the principles outlined in the Declaration of Helsinki for all investigations.
Consent
The requirement for individual consent for this retrospective analysis was waived.
Availability of Data and Materials
All data generated or analysed during this study are included in this published article.
Conflicts of Interest
None.
Article Info
Article Type
Research ArticlePublication history
Received: Thu 10, Jun 2021Accepted: Wed 23, Jun 2021
Published: Thu 15, Jul 2021
Copyright
© 2023 Manxia Lin. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2021.07.04
Author Info
Jiayao Huang Junrong Chen Xiaoying Lou Yi Lu Jiayin Yao Manxia Lin Jiachen Sun
Corresponding Author
Manxia LinDepartment of Ultrasound, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, China
Figures & Tables
Table 1: Characteristics of patients with gastrointestinal
mucosal melanomas.
|
Characteristic |
|
Cases (%) |
|
Total |
|
49 |
|
Sex |
Male |
19 (38.8%) |
|
|
Female |
30 (61.2%) |
|
Age (years) |
≤60 |
25 (51.0%) |
|
|
>60 |
24 (49.0%) |
|
Primary or metastatic |
Primary |
43 (87.8%) |
|
|
Metastatic |
6 (12.2%) |
|
Tumor location |
Upper gastrointestinal tract |
8 (16.3%) |
|
|
Lower gastrointestinal tract |
41 (83.7%) |
|
Number of lesions |
Single |
28 (57.1%) |
|
|
Multiple |
7 (14.3%) |
|
|
Unspecified |
14 (28.6%) |
|
Initial symptoms |
Bleeding (haematochezia or haematemesis) |
21 (42.9%) |
|
|
Non-bleeding |
28 (57.1%) |
|
Distant metastasis |
Yes |
16 (32.7%) |
|
|
No |
18 (36.7%) |
|
|
Unspecified |
15 (30.6%) |
|
Lymph node metastasis |
Yes |
27 (55.1%) |
|
|
No |
8 (16.3%) |
|
|
Unspecified |
14 (28.6%) |
|
Melan A (immunohistochemical analysis) |
(+) |
33 (67.3%) |
|
|
(-) |
13 (26.5%) |
|
|
Unspecified |
3 (6.1%) |
|
Treatments |
No (had not received any treatment or had received only
palliative care) |
12 (24.5%) |
|
|
Only surgery |
19 (38.8%) |
|
|
Only chemotherapy |
1 (2.0%) |
|
|
Both surgery and chemotherapy |
17 (34.7%) |
Table 2: Multivariate analyses of prognostic factors for
overall survival in patients with gastrointestinal mucosal melanomas.
|
Variable |
P-value |
Hazard ratio |
95.0% CI for hazard ratio |
||
|
Lower |
Upper |
||||
|
Sex |
Male |
- |
- |
- |
- |
|
|
Female |
0.615 |
0.734 |
0.220 |
2.446 |
|
Age (years) |
≤60 |
- |
- |
- |
- |
|
|
>60 |
0.617 |
0.777 |
0.289 |
2.088 |
|
Primary or metastatic |
Primary |
- |
- |
- |
- |
|
|
Metastatic |
0.038 |
13.062 |
1.160 |
147.127 |
|
Tumor location |
Upper gastrointestinal tract |
- |
- |
- |
- |
|
|
Lower gastrointestinal tract |
0.040 |
14.993 |
1.136 |
197.823 |
|
Number of lesions |
Single |
- |
- |
- |
- |
|
|
Multiple |
0.809 |
0.806 |
0.140 |
4.632 |
|
Initial symptoms |
Bleeding (haematochezia or haematemesis) |
- |
- |
- |
- |
|
|
Non-bleeding |
0.160 |
2.628 |
0.683 |
10.110 |
|
Distant metastasis |
Yes |
- |
- |
- |
- |
|
|
No |
0.660 |
0.729 |
0.178 |
2.988 |
|
Lymph node metastasis |
Yes |
- |
- |
- |
- |
|
|
No |
0.132 |
3.394 |
0.692 |
16.635 |
|
Melan A (immunohistochemical analysis) |
(+) |
- |
- |
- |
- |
|
|
(-) |
0.152 |
5.777 |
0.524 |
63.747 |
|
Treatments |
No (had not received any treatment or had received only
palliative care) |
- |
- |
- |
- |
|
|
Only surgery |
0.061 |
5.770 |
0.922 |
36.103 |
|
|
Only chemotherapy |
0.237 |
6.540 |
0.291 |
147.070 |
|
|
Both surgery and chemotherapy |
0.281 |
2.686 |
0.446 |
16.183 |
Table 3: Mean and median OS of gastrointestinal mucosal
melanoma patients with different characteristics.
|
Characteristic |
Mean OS
(months) |
Median OS
(months) |
|
|
Total |
|
38.395 |
30.000 |
|
Sex |
Male |
38.153 |
36.000 |
|
|
Female |
38.177 |
26.000 |
|
Age (years) |
≤60 |
43.414 |
36.000 |
|
|
>60 |
33.016 |
23.000 |
|
Primary or
metastatic |
Primary |
35.311 |
30.000 |
|
|
Metastatic |
50.600 |
55.000 |
|
Tumor location |
Upper
gastrointestinal tract |
29.208 |
14.000 |
|
|
Lower
gastrointestinal tract |
38.850 |
30.000 |
|
Number of
lesions |
Single |
40.111 |
36.000 |
|
|
Multiple |
22.400 |
20.000 |
|
|
Unspecified |
43.451 |
30.000 |
|
Initial
symptoms |
Bleeding
(haematochezia or haematemesis) |
23.930 |
22.000 |
|
|
Non-bleeding |
50.888 |
55.000 |
|
Distant
metastasis |
Yes |
33.193 |
26.000 |
|
|
No |
39.097 |
25.000 |
|
|
Unspecified |
43.451 |
30.000 |
|
Lymph node metastasis |
Yes |
32.972 |
31.000 |
|
|
No |
49.292 |
26.000 |
|
|
Unspecified |
43.451 |
30.000 |
|
Melan A
(immunohistochemical analysis) |
(+) |
29.796 |
30.000 |
|
|
(-) |
46.222 |
36.000 |
|
|
Unspecified |
39.500 |
20.000 |
|
Treatments |
No (had not
received any treatment or had received only palliative care) |
31.481 |
31.000 |
|
|
Only surgery |
35.604 |
20.000 |
|
|
Only
chemotherapy |
17.000 |
17.000 |
|
|
Both surgery
and chemotherapy |
50.393 |
36.000 |
References
1.
Yde SS, Sjoegren
P, Heje M, Stolle LB (2018) Mucosal Melanoma: a Literature Review. Curr
Oncol Rep 20: 28. [Crossref]
2.
Postow MA, Hamid
O, Carvajal RD (2012) Mucosal melanoma: pathogenesis, clinical behavior, and
management. Curr Oncol Rep 14: 441-448. [Crossref]
3.
Loganathan G,
Singh P (2014) Gastric malignant melanoma. Indian J Gastroenterol 33:
199. [Crossref]
4.
Kouvaras S,
Rokkas T, Goga H, Gakiopoulou H, Arapantoni P et al. (2019) Multifocal Gastrointestinal
Melanoma. J Gastrointestin Liver Dis 28: 237-240. [Crossref]
5.
D’Angelo SP,
Larkin J, Sosman JA, Lebbé C, Brady B et al. (2017) Efficacy and Safety of
Nivolumab Alone or in Combination With Ipilimumab in Patients With Mucosal
Melanoma: A Pooled Analysis. J Clin Oncol 35: 226-235. [Crossref]
6.
Tacastacas JD,
Bray J, Cohen YK, Arbesman J, Kim J et al. (2014) Update on primary mucosal melanoma.
J Am Acad Dermatol 71: 366-375.
[Crossref]
7.
Mihajlovic M,
Vlajkovic S, Jovanovic P, Stefanovic V et al. (2012) Primary mucosal melanomas:
a comprehensive review. Int J Clin Exp Pathol 5: 739-753. [Crossref]
8.
Patrick RJ,
Fenske NA, Messina JL (2007) Primary mucosal melanoma. J Am Acad Dermatol
56: 828-834. [Crossref]
9.
Cui C, Lian B, Zhou L, Song X, Zhang X et al. (2018) Multifactorial Analysis of Prognostic Factors
and Survival Rates Among 706 Mucosal Melanoma Patients. Ann Surg Oncol
25: 2184-2192. [Crossref]
10.
Bradburn MJ,
Clark TG, Love SB, Altman DG (2003) Survival analysis part II: multivariate
data analysis--an introduction to concepts and methods. Br J Cancer 89:
431-436. [Crossref]
11.
Bradburn MJ, Clark
TG, Love SB, Altman DG (2003) Survival analysis Part III: multivariate data
analysis -- choosing a model and assessing its adequacy and fit. Br J Cancer
89: 605-611. [Crossref]
12.
Balch CM,
Gershenwald JE, Soong SJ, Thompson JF, Atkins MB et al. (2009) Final version of
2009 AJCC melanoma staging and classification. J Clin Oncol 27:
6199-6206. [Crossref]
13.
Zhang Y, Fu X, Qi
Y, Gao Q (2019) A study of the clinical characteristics and prognosis of
advanced mucosal and cutaneous melanoma in a Chinese population. Immunotherapy
11: 91-99. [Crossref]
14.
Sinagra E, Sciumè
C (2020) Ileal Melanoma, A Rare Cause of Small Bowel Obstruction: Report of a
Case, and Short Literature Review. Curr Radiopharm 13: 56-62. [Crossref]
15.
Heppt MV, Roesch
A, Weide B, Gutzmer R, Meier F et al. (2017) Prognostic factors and treatment
outcomes in 444 patients with mucosal melanoma. Eur J Cancer 81: 36-44. [Crossref]
16.
Al Haseni A,
Vrable A, Qureshi MM, Mathews S, Pollock S et al. (2019) Survival outcomes of
mucosal melanoma in the USA. Future Oncol 15: 3977-3986. [Crossref]
17.
Balch CM,
Gershenwald JE, Soong SJ, Thompson JF, Ding S et al. (2010) Multivariate
analysis of prognostic factors among 2,313 patients with stage III melanoma:
comparison of nodal micrometastases versus macrometastases. J Clin Oncol
28: 2452-2459. [Crossref]
18. Frumovitz M, Etchepareborda M, Sun CC, Soliman PT, Eifel PJ et al. (2010) Primary malignant melanoma of the vagina. Obstet Gynecol 116: 1358-1365. [Crossref]
19. Iddings DM, Fleisig AJ, Chen SL, Faries MB, Morton DL (2010) Practice patterns and outcomes for anorectal melanoma in the USA, reviewing three decades of treatment: is more extensive surgical resection beneficial in all patients? Ann Surg Oncol 17: 40-44. [Crossref]