Treating Acute Myeloid Leukemia in Patients Presenting with COVID-19
A B S T R A C T
Patients with hematological malignancies are at increased risk for severe COVID-19 with a high mortality rate. In AML, more and more patients are treated on an outpatient basis. This gives rise to two dilemmas in the face of COVID-19. The first one is whether to temporarily interrupt the oral antileukemia treatment and the second one is which patients should be intubated. Here, we describe our institutional approach and the underlying rationale in patients on lower intensity treatments for AML presenting with COVID-19.
Keywords
acute myeloid leukemia, COVID-19, IDH1/2-inhibitors, FLT3-inhibitors, hypomethylating agents, treatment discontinuation
Introduction
The management of patients with hematological malignancies has been complicated by the onset of the coronavirus disease 2019 (COVID-19) pandemic. In this article, we will discuss the management of patients with acute myeloid leukemia (AML) who are hospitalized for COVID-19, with a focus on outpatient treatment with oral targeted therapies or hypomethylating agents (HMAs). It is a group that contains both relapsed/refractory young patients and medically unfit older patients. To illustrate our approach, we report on two patients with AML who were hospitalized because of COVID-19 on the same day and followed a synchronous clinical course, which unfortunately resulted in their death. Based on these cases, we will discuss two clinical dilemmas. The first one is whether to interrupt or continue the oral antileukemia treatment, and the second one is whether to intubate these patients.
Case 1
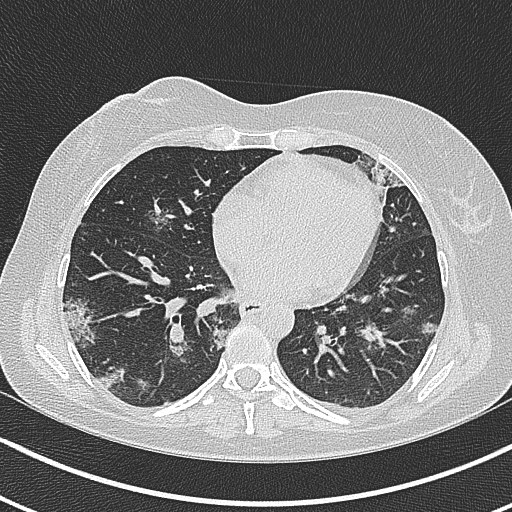
A 46-year-old female was admitted because of COVID-19. She was previously diagnosed with intermediate-risk AML with mutated NPM1 and FLT3-ITD (high). She had a medical history of a hormone receptor-positive non-metastatic breast cancer which was treated in 2007 with surgery, adjuvant radiotherapy and endocrine therapy. She was diagnosed with AML in 2019 and had refractory disease after two cycles of intensive induction chemotherapy. Subsequently, she was started on gilteritinib (an oral FLT3-inhibitor). Three weeks later, she was hospitalized for neutropenic fever and was empirically started on piperacillin-tazobactam. Her white blood cell differential on peripheral blood still showed a markedly elevated blast count (52% of white blood cells or 705 blasts/µL). Chest CT was strongly suggestive of COVID-19 (Figure 1) and SARS-CoV-2 PCR was positive on nasopharyngeal (NP) swab. Treatment with gilteritinib was interrupted.
Figure 1: Chest CT on admission. Bilateral ground-glass opacities and crazy paving appearance with a predominantly peripheral distribution.
During hospitalization, she deteriorated further with a raising oxygen need. High-flow oxygen therapy through a nasal cannula (HFOT) was started and increased to maximal settings, but she continued to decline. After consultation with the patient’s wishes, she was not transferred to the intensive care ward but was started on palliative sedation. She eventually succumbed to the disease on the evening of the sixth day of her hospitalization.
Case 2
On the same day as the first patient, a 77-year-old patient with a history of cardiovascular and chronic kidney disease was admitted to the COVID-19 ward. Because of worsening pancytopenia, on March 10, 2020, a diagnosis of AML with myelodysplasia-related changes with high-risk RUNX1 and ASXL1 mutations was made. Treatment with azacitidine was initiated.
At day nine of the second cycle of azacytidine, he was hospitalized for recurrent fever and was empirically started on ceftazidime. SARS-CoV-2 PCR on NP swab turned positive. Arterial blood gas on ambient air showed a PaO2 of 50.4 mmHg (ref. value 83-108 mmHg), and supplemental oxygen was started. The following days he rapidly declined with an increasing oxygen need and rising inflammatory markers. On the fifth night of his hospitalization, he developed respiratory distress, and HFOT was started. He was not transferred to the intensive care ward as the patient’s life expectancy was deemed too short for mechanical ventilation. Unfortunately, the patient died on the evening of the sixth day of his hospitalization.
Discussion
These two cases highlight the potentially rapid fatal course of COVID-19 in patients with AML. Even when patients with AML do not fall in the typical risk categories for COVID-19, they are at increased risk of severe disease due to their underlying disease- and treatment-related immunodeficiency [1, 2]. It is currently not known whether it is wise to interrupt oral anticancer treatment during active infection or COVID-19. For example, limited clinical and pathophysiological data suggest that ibrutinib, a Btk-inhibitor, may protect against pulmonary injury in COVID-19 patients due to its mediation of pro-inflammatory signaling [3]. Similar to the situation in our first case, the decision to interrupt treatment is even more difficult in patients who are not (yet) in complete or partial remission. Specifically, in AML, the most frequently used targeted therapies are FLT3- and IDH1/2 inhibitors.
FLT3 can activate downstream signaling pathways responsible for survival, maturation, and proliferation of hematopoietic cells and inhibition of FLT3-signaling has been shown to significantly reduce the number of B-cell progenitors, dendritic cells, and natural killer cells [4]. In the ADMIRAL trial, patients on gilteritinib had a higher risk of grade 3 or higher febrile neutropenia and fatal infection compared to the salvage chemotherapy group (45.9% resp. 36.7%, and 11.4% resp. 6.4%) [5].
IDH1 and IDH2 mutations, on the other hand, confer a neomorphic enzymatic activity, resulting in the reduction of α-ketoglutarate to the oncometabolite R-2-hydroxyglutarate, which leads to epigenetic alterations and impaired hematopoietic differentiation [6]. Inhibition of mutated IDH1 or IDH2 induced differentiation in models of IDH-mutated tumors [7]. As such, IDH1 and IDH2 inhibitors should theoretically decrease infection risk by restoring normal hematopoiesis. As a matter of fact, patients who had a response to the IDH1 inhibitor ivosidenib had a lower rate of infections and febrile neutropenia than those who did not [8].
The effect of HMAs on the risk of infection is poorly understood. They transiently worsen the neutropenia, especially during the initial treatment cycles, but the risk of febrile neutropenia, pneumonia and pyrexia (25%, 20% and 10% resp.) seems to be similar in older patients treated with HMAs, low-dose cytarabine or best supportive care [9]. Adding the BCL-2 inhibitor, venetoclax to HMAs seems to improve response rates (CR+CRi 67% resp. 27.8%) but with more frequent grade 3 or 4 febrile neutropenia (in 43% resp. 28% of patients), when we compare the studies of Dombret et al. and DiNardo et al. [9, 10]. We await the phase 3 trial of venetoclax plus azacitidine (NCT02993523) to confirm whether the addition of venetoclax to azacitidine increases the risk of infection.
Based on these data, we interrupt FLT3-inhibitors but continue IDH1/2-inhibitors in patients presenting with COVID-19. In the case of HMAs, with or without venetoclax, continuing or interrupting depends on the number of cycles the patient has had and whether there were significant cytopenias in the previous cycles. In the first patient, we opted to interrupt gilteritinib, whereas patient 2 had completed the second cycle of azaciditine the week before, so delaying treatment was not an issue (he did not receive venetoclax as it was not reimbursed). Besides continuing or interrupting targeted therapy, another major issue is whether to intubate these patients or not. The rate of ICU admission in COVID-19 is much higher in cancer and immunocompromised patients than in the general population [1, 2]. Moreover, the disease course of serious COVID-19 is characterized by long-lasting mechanical ventilation of usually more than two weeks, leading to a lengthy revalidation with a serious impact on the quality of life [11].
In the high-risk group of older, medically unfit patients who receive HMAs or patients with a limited life expectancy, we do not initiate mechanical ventilation but focus instead on the quality of life. After discussing the expected disease course, patient 2 declined intubation. We did, however, initiate HFOT, primarily to allow him to say goodbye to his relatives. The discussion about mechanical ventilation becomes even more complex and emotionally charged in young patients with relapsed or refractory AML. Although the median survival of targeted therapies in the relapsed/refractory setting is only around nine months, they have a chance of survival if they can proceed to an allogeneic stem cell transplantation [6, 9]. However, when we discussed the options and possible outcomes with patient 1, she told us that she was tired of the struggle and declined mechanical ventilation.
In conclusion, in patients with AML presenting with COVID-19, we interrupt FLT3-inhibitors but continue IDH1/2-inhibitors. (Dis)continuation of HMAs and venetoclax is evaluated on an individual basis. Upon hospitalization, we discuss with our patients if they want to be intubated should the need arise.
Author Contributions
R.C. wrote the manuscript. D.S., S.D., E.P. and I.M. read and corrected where needed. All authors took part in the discussion leading up to the manuscript. All authors agree with the final versions of the manuscript.
Conflicts of Interest
None.
Article Info
Article Type
Case ReportPublication history
Received: Sat 13, Jun 2020Accepted: Thu 02, Jul 2020
Published: Sat 18, Jul 2020
Copyright
© 2023 Ine Moors. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.COR.2020.07.05
Author Info
Rutger Callens Dieter Stevens Sebastiaan Dhont Elise Peys Ine Moors
Corresponding Author
Ine MoorsDepartment of Hematology, Ghent University Hospital, Ghent, Belgium
Figures & Tables
References
- Wenhua Liang, Weijie Guan, Ruchong Chen, Wei Wang, Jianfu Li et al. (2020) Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol 21: 335-337. [Crossref]
- Wenjuan He, Lei Chen, Li Chen, Guolin Yuan, Yun Fang et al. (2020) COVID-19 in persons with haematological cancers. Leukemia 34: 1637-1645. [Crossref]
- Steven P Treon, Jorge J Castillo, Alan P Skarbnik, Jacob D Soumerai, Irene M Ghobrial et al. (2020) The BTK-inhibitor ibrutinib may protect against pulmonary injury in COVID-19 infected patients. Blood 135: 1912-1915. [Crossref]
- D Gary Gilliland, James D Griffin (2002) The roles of FLT3 in hematopoiesis and leukemia. Blood 100: 1532-1542. [Crossref]
- Alexander E Perl, Giovanni Martinelli, Jorge E Cortes, Andreas Neubauer, Ellin Berman et al. (2019) Gilteritinib or Chemotherapy for Relapsed or Refractory FLT3-Mutated AML. N Engl J Med 381: 1728-1740. [Crossref]
- Patrick S Ward, Jay Patel, David R Wise, Omar Abdel Wahab, Bryson D Bennett et al. (2010) The common feature of leukemia-associated IDH1 and IDH2 mutations is a neomorphic enzyme activity converting alpha-ketoglutarate to 2-hydroxyglutarate. Cancer Cell 17: 225-234. [Crossref]
- Hansen E, Quivoron C, Straley K, Lemieux RM, Popovici Muller J et al. (2014) AG-120, an Oral, Selective, First-in-Class, Potent Inhibitor of Mutant IDH1, Reduces Intracellular 2HG and Induces Cellular Differentiation in TF-1 R132H Cells and Primary Human IDH1 Mutant AML Patient Samples Treated Ex Vivo. Blood 124: 3734-3734.
- Courtney D DiNardo, Eytan M Stein, Stéphane de Botton, Gail J Roboz, Jessica K Altman et al. (2018) Durable Remissions with Ivosidenib in IDH1-Mutated Relapsed or Refractory AML. N Engl J Med 378: 2386-2398. [Crossref]
- Hervé Dombret, John F Seymour, Aleksandra Butrym, Agnieszka Wierzbowska, Dominik Selleslag et al. (2015) International phase 3 study of azacitidine vs conventional care regimens in older patients with newly diagnosed AML with >30% blasts. Blood 126: 291-299. [Crossref]
- Courtney D DiNardo, Keith Pratz, Vinod Pullarkat, Brian A Jonas, Martha Arellano et al. (2019) Venetoclax combined with decitabine or azacitidine in treatment-naive, elderly patients with acute myeloid leukemia. Blood 133: 7-17. [Crossref]
- Giacomo Grasselli, Alberto Zangrillo, Alberto Zanella, Massimo Antonelli, Luca Cabrini et al. (2020) Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA 323: 1574-1581. [Crossref]
