Transplantation of Induced Neural Stem Cells Derived from Human Fibroblasts Promotes Functional Recovery of Spinal Cord Injury in Rats
A B S T R A C T
Background: Traumatic injury to the spinal cord causes cell death, demyelination, axonal degeneration, and cavitation, resulting in functional motor and sensory loss. Stem cell therapy as a promising approach for spinal cord injury (SCI) has some challenges, such as immunological responses to grafted cells. Transplantation of autologous trans-differentiated cells can be a useful strategy to overcome this problem.
Materials and Methods: In this research, we transplanted human-induced neural stem cells (hiNSCs), which were trans-differentiated from adult human fibroblasts into the injured spinal cord of adult rats on day seven post-injury. Before transplantation, hiNSCs were explored for expressing NSCs general protein and genes and also, their normal karyotype was examined. After hiNSCs transplantation, behavioural tests (BBB score and grid walk tests) were performed weekly and finally histological assessment was done for exploring the cell fates and migration.
Results: Our results showed the cell viability, differentiation, and migration of transplanted hiNSCs was significantly improved in the injured site of the spinal cord up to seven weeks after the SCI. Also, the behavioural analysis revealed the enhanced locomotor functions of the animals that underwent transplantation after seven weeks. Our data provide strong evidence in support of the feasibility of hiNSCs for cell-based therapy in SCI rats.
Keywords
Spinal cord injuries, trans-differentiation, differentiation, neural stem cells, zinc finger protein 521, rats, genes
Introduction
Spinal cord injury (SCI) is one of the most disabling injuries. In 2016, there were 930,000 (780,000-1,160,000) new cases of SCI in the world and the number of prevalent cases of SCI was 27 million (25-30 million) [1]. Globally, the incidence of TSCI ranges from 3.6 to 195.4 patients per million [2]. The incidence of SCI in developing countries is 25.5 per million per year (95% confidence interval: 21.7-29.4/million/year) [3]. There is no cure for SCI and working on injury prevention, pathophysiology and treatment all are important options for consideration.
Immediately after SCI, a cascade of cellular and tissue events occurs that haemorrhage initiation and its expansion at the injury site causes migration of inflammatory cells in the next stage. Release of inflammatory mediators such as cytokines from these cells attracts more inflammatory cells which make positive feedback [4]. At the acute phase which may last from several days to weeks, the damaged neuronal and glial cells secrete several factors, contributing to the production of harmful chemical agents. This stage is called the acute phase, which may last from several days to weeks [4]. At the end of the acute phase, due to the function of immune cells, the rate of inflammation would be declined, and a glial scar is formed at the injured site [5]. Reactive astrocytes are the most abundant cells found in glial scar formation and inhibit axonal growth in lesions [5]. The structure of the glial scar in which are formed at the initiation of the sub-acute phase and become complete in the chronic phase prevents the repair of neuronal damage and acts as a barrier [5, 6].
The treatment of SCI is limited as a control of secondary complications, and it is mainly focused on physical rehabilitation and pharmacological therapy [7]. In recent years, two approaches have been proposed to retrieve the functional loss in patients with SCI including, to promote endogenous repair by applying external reagents such as drugs or growth factors and replacing the lost cells by transplantation of the appropriate cell from exogenous sources [8, 9]. Several preclinical studies have applied cell therapies to repair the injured spinal cord. However, there are many challenges to start clinical research because of inconsistencies in the efficacy of therapy, limits on the number of patients in acute/sub-acute SCI, and the high costs of clinical trials [10, 11]. The transplantation of Schwann cells, neural stem cells or progenitor cells, olfactory ensheathing cells, oligodendrocyte precursor cells, and mesenchymal stem cells has been investigated as potential therapies for the treatment of SCI in animal models [12]. Although there is little information about which one of these types of cells can promote the repair process and functional improvement, some therapeutic strategies have been applied, including neuroprotection, immunomodulation, axonal regeneration, neuronal relay formation, and myelin regeneration. However, since the nature of the injury is multifaceted, a combination of approaches would be necessary for the treatment of SCI [13, 14].
This study is focused on the therapeutic effect of neural precursor cells (NSCs) on the neuronal repair process, following SCI. Upon transplantation of NSCs, they can differentiate into either glial or neuronal lineages [15]. However, the process of transplantation is always integrated with immunosuppressive agents, possessing severe side effects on patients, and putting them at risk of developing infections and trauma. In the case of using autologous cells, the rate of immune rejection could be markedly diminished [16]. Reprogramming technology provides the opportunity for the transformation of somatic cells into NSCs by direct conversion. In the present study, the hiNSC cell line, which had been produced by using a single Zinc-Finger transcription factor was transplanted to evaluate the efficacy of this type of cell line in the treatment of SCI [17].
Materials and Methods
I Animal Procedures
All performed procedures on animals comply with the institutional animal care and use committee (IACUC) at the Royan Institute, Iran. A total of 40 adults Wistar Rats (250-280 g) were used in this study. All animals were randomly divided into four groups; control lesion (n=12), fibroblast-transplanted group (n=9), hiNSCs transplanted (n=11), and sham group in which the animals received phosphate-buffered saline (PBS) only (n=10).
II Establishment and Culture of hiNSC
The generation and culture of neural stem cells derived from human fibroblasts accomplished by the method developed by Shahbazi and his colleagues [17]. Briefly, human fibroblasts were transduced with lentiviral vectors expressing Zfp521 together with a green fluorescent protein (GFP) tracer (Life Technologies). After viral infection, the cells were cultured in the neural cell culture medium, containing DMEM/Ham’s F12 (DMEM/F12; Invitrogen) supplemented with 10% knockout serum replacement (KOSR), 1% non-essential amino acids (NEAA), 1% L-glutamine, ITS (1 mg/ml insulin, 0.55 mg/ml transferrin, 0.67 mg/ml selenium), 1% N2-supplement, 0.05% B27, penicillin/streptomycin (all purchased from Invitrogen), and 1.6 g/l glucose (Sigma-Aldrich). Doxycycline (2 mg/ml) was added to the culture medium to induce Zfp521 expression for 30 days. The epidermal growth factor and basic fibroblast growth factor (EGF, 20 ng/ml and bFGF, 20 ng/ml; Royan Institute) were added to the neural cell culture medium from days 18 to 24. Neurospheres were collected and transferred into a dish and then trypsinized. The isolated cells were subsequently seeded onto tissue culture dishes coated with 0.001% poly-L-ornithine and 1 mg/ml Laminin (Sigma-Aldrich) at 37ºC for 1 hr. and cultured in the neural cell culture medium supplemented with EGF and bFGF in the absence of doxycycline [17].
III SCI and Transplantation Procedures
Animals were anaesthetized with the intraperitoneal injection of a mixture of Ketamine (100 mg/kg) and Xylazine (10 mg/kg). The surgical area was first shaved and disinfected with 70% ethanol and betadine. A midline incision was made at the T9-T11 area, and then the skin and superficial muscles were retracted. A laminectomy was performed at the T10-T11 area. A moderate to severe contusion injury was made by dropping a 10g weight rod from a 25-mm height onto the dorsal surface of the spinal cord via the NYU-impactor device. All rats, post-operation received 5ml of 0.9% saline and 2.5 mg/kg Enrofloxacin subcutaneously. Manual bladder expression was daily administered twice until voiding reflex returned. Cyclosporin A (10 mg/kg) and Enrofloxacin (2.5 mg/kg) were subcutaneously administered. Seven days after SCI modeling prepared cells transplanted into animals. A total number of 2-3×105 hiNSCs and fibroblasts per 5 microliters were injected into the epicenter, upper, and lower region of the injured site.
IV Behavioural Assessments
The Basso, Beattie, and Bresnahan (BBB) open-field locomotor rating scale was performed by two blind observers [18]. The BBB scores of all experimental groups were zero for three days post-SCI. The BBB test was carried out weekly for seven weeks after SCI to evaluate the hind limb recovery [18]. Deficits in hindlimb motor function were measured by the Grid walk test. For this aim, the Grid walk device was used that consisted of a plate (one-meter length) which possesses irregular pores (1-5 cm diameter). When the animals walk on the plate, the number of foot fault errors are recorded. Three days before SCI modeling all rats were trained to walk on the plate to familiarize them with the apparatus. The Grid walk test was carried out every week for seven weeks [19].
V Histological Analysis
Animals were sacrificed and transcardially perfused with 4% paraformaldehyde (pH 7.4). The injured spinal cord was dissected and post-fixed in 4% paraformaldehyde for 2 hours. Then, the tissues were cryopreserved in 30% sucrose dissolved in PBS. One-centimeter length of the spinal cord at the injury site was separated and embedded in optimal cutting temperature (OCT) solution. Spinal cord tissue was sectioned at the thickness of 14 µm using a Cryostat apparatus and then placed on tissue-adhering slides and subsequently stored at -70ºC until use. Before immunofluorescence staining, the slides were air-dried for 10 minutes and washed with PBS-Twin for 5 minutes. The tissue sections were permeabilized by incubation in Triton X-100 for 20 minutes and blocked with the secondary host serum for 1 hour. The sections were incubated at 4ºC overnight with the primary antibodies. After washing in PBS-Tween for three times, the nuclear staining was performed by DAPI. The tissue sections were imaged by an inverted microscope (Olympus, BX51). Primary antibodies were: TUJ1 (Santa Cruz, Sc‐58888, USA), NG2 (Santa Cruz, Sc‐53389, USA), PDGFR-Alpha (Santa Cruz, Sc‐21789, USA), GFAP (Biorbyt, ORB 69320, Sweden), Anti-human nuclear antibody (Abcam, ab191181, USA), NESTIN (Santa Cruz, Sc‐58813, USA), SOX1 (Santa Cruz, Sc‐17318, USA), PAX6 (Santa Cruz, Sc‐81649, USA). Nuclei were counterstained with DAPI (Sigma‐Aldrich, D8417).
VI Statistical Analysis
The obtained data were analysed by the SPSS software (version 16). The difference between the experimental groups was compared using the one-way analysis of variance (ANOVA), followed by Tukey's post hoc test. The values were expressed as the mean and standard error of the mean (mean ± SEM). The level of significance was set at p-value <0.05.
Results
I Characterization, Differentiation, and Migration of hiNSCs
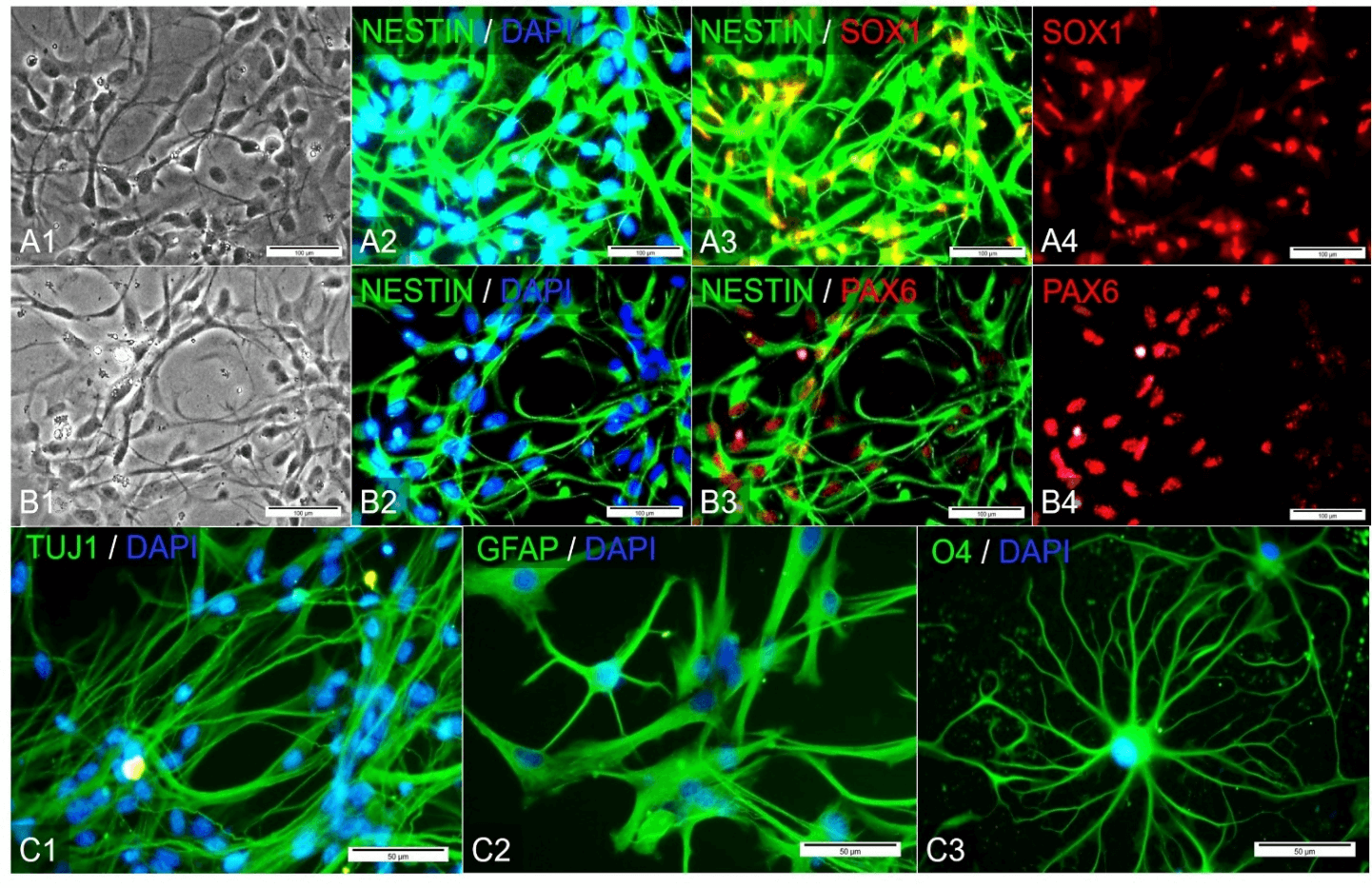
Before transplantation, hiNSCs were characterized according to specific protein expression. The expression of NESTIN, SOX1 (Figure 1A), and PAX6 (Figure 1B) proteins, which are considered specific markers of NSCs, were evaluated. After 14 days of the cell culture period, hiNSCs were spontaneously differentiated into neural-like cells with removing growth factors and express special proteins such as TUJ1 and GFAP (Figure 1C). One week after SCI, hiNSCs were transplanted into the site of injured site. Six weeks after SCI, the histological assessments showed the GFP-positive hiNSCs were rarely observed and then faded. These cells were detected using a human nuclear antigen protein antibody (Figure 2A).
Figure 1: Generation and the cell culture of hiNSCs, A) & B) hiNSCs had typical morphology of neural stem cells before the transplantation process. The expression of protein markers: NESTIN, SOX1, and PAX6. They also were able to express NESTIN, SOX1, and PAX6 markers, C) hiNSCs had the ability to differentiate into TUJ1-, GFAP-, O4-positive cells spontaneously after 14 days.
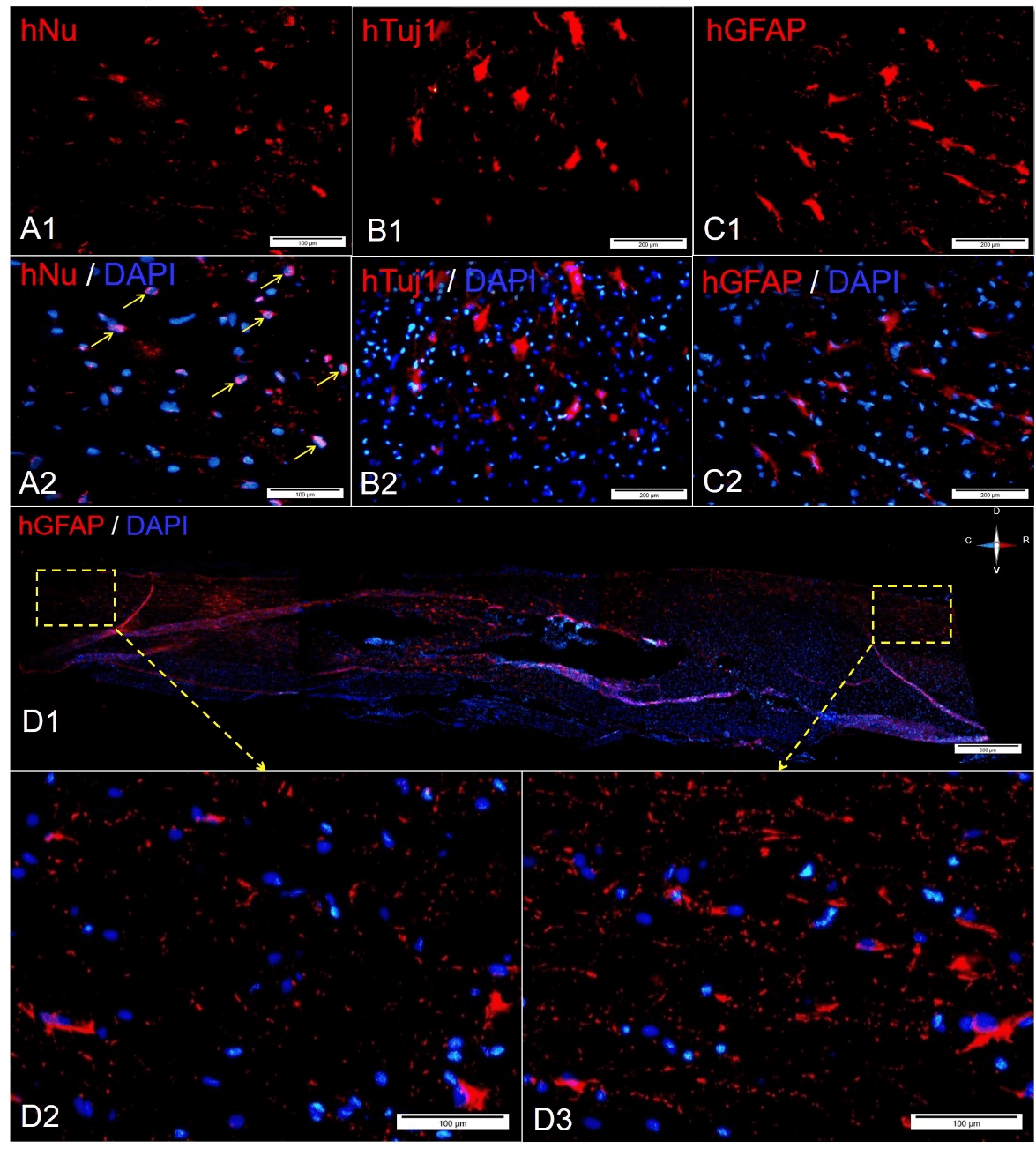
Figure 2: Survival, differentiation, and migration of transplanted hiNSCs, A) Transplanted hiNSCs expressed the human nuclear antigen protein one week after transplantation that shows their viability in the damaged tissue, B) & C) Transplanted hiNSCs expressed hTUJ1- and hGFAP-positive cells seven weeks after SCI, D1) The longitudinal section of the cells in the hiNSCs-transplanted group labeled with hGFAP; D2) & D3) Rostro-caudal migration of hGFAP-positive cells.
Immunohistofluorescence staining demonstrated that the site of injury in the spinal cord has a crucial impact on the fate of engrafted hiNSCs [20]. Our findings showed that human neural and glial cell protein markers (hTuj1 and hGFAP) are expressed six weeks after the process of transplantation (Figures 2B & 2C). Moreover, transplanted hiNSCs were not positive for oligodendrocyte cells protein markers, such as PDGFRa and Olig2. Besides, the migration of transplanted hiNSCs was tracked by hGFAP staining (Figures 2D1, 2D2 & 2D3). It seems that hiNSCs migrated rostro-caudally up to 4 mm from the injured site.
II Hindlimb Recovery in Behavioural Test
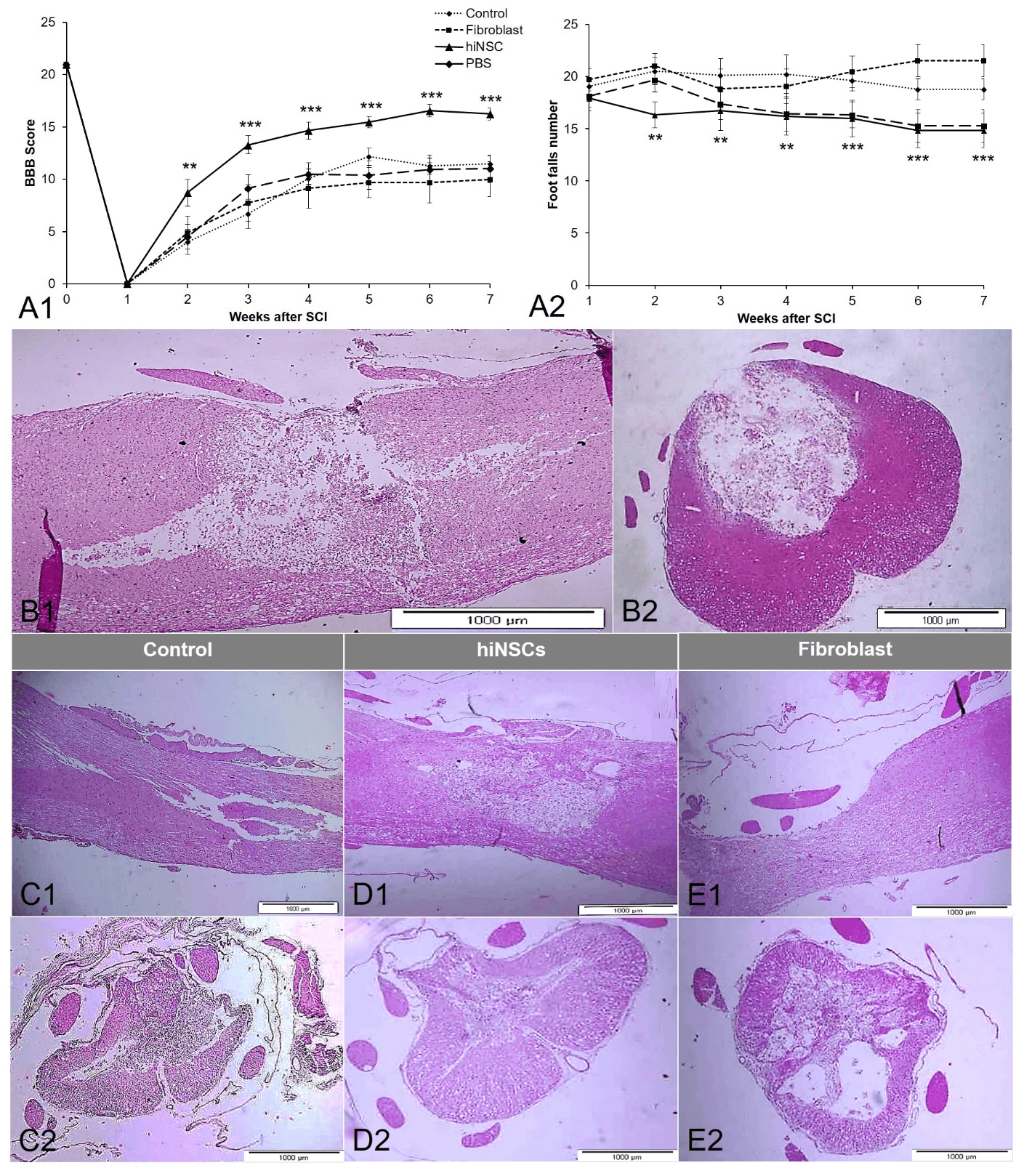
We assessed the recovery of hindlimb movement in all experimental groups (Figures 3A1 & 3A2). According to the BBB test, the fibroblast, and PBS groups did not show any significant difference as compared with the control group (one-way ANOVA with Tukey's post hoc test, NS) and the scores were the same as values observed in the originally-published study [21]. The best walking position in these three groups consisted of frequent weight-supported plantar steps and occasional FL-HL coordination (the mean of BBB scores were 8 to 12); however, the hindlimb movement of the hiNSCs-transplanted group significantly recovered in comparison with other experimental groups during seven weeks (one-way ANOVA with Tukey's post hoc test, Figure 3A1, P < 0.01, P,0.001). Most of these animals had consistent plantar steps while they had FL–HL coordination and occasional toe clearance (the mean of BBB scores was 15 to 17).
The Grid walk test confirmed the results obtained from the BBB open-field test as well. There were significant (Figure 3A2) differences in the frequency of foot fault errors between the hiNSCs-transplanted group, the fibroblast-transplanted and control lesion groups at the second to fourth weeks after the induction of SCI (one-way ANOVA with Tukey's post hoc test, Figure 3A1, P < 0.01, P,0.001)). Nevertheless, there was no significant difference between the hiNSC transplanted and PBS groups (Figure 3A2).
Figure 3: A) hiNPC transplantation improved tissue repair and caused a significant behavioural recovery. One-way ANOVA, followed by Tukey's post hoc test, was performed. The data obtained from the BBB and Grid walk tests were expressed as mean ± SEM. ** shows significant differences with the control group (P<0.01), and *** indicates significant differences with the control group (P<0.001). A1) the hiNSCs-transplanted group had significant motor-neuron recovery in comparison with other groups, initiated from two weeks post-SCI to the end of the study. A2) In the Grid walk test, the number of foot fault errors were substantially lower in the hiNSCs-transplanted group than that of the fibroblast-transplanted and control lesion groups one week after the transplantation process to the end of the study. B) Hematoxylin-eosin staining of the cystic cavity in the spinal cord, A1) Longitudinal section; A2) Transverse section; C), D) & E) Hematoxylin-eosin staining indicated a marked decrease in the cystic cavity of the injured spinal cord after seven weeks of SCI induction. C1) Longitudinal section; C2) Transverse section of the control lesion group. D1) longitudinal section; D2) Transverse section of the hiNSC-transplanted group. E1) longitudinal section; E2) Transverse section of the fibroblast-transplanted group.
III Changing in Cavity Size After Cell Therapy Intervention
One week after SCI, animals were transcardially perfused with 4% paraformaldehyde, and their spinal cord tissue was removed and stained with the hematoxylin-eosin staining method. The area of the cystic cavity was evaluated by hematoxylin and eosin staining. A cystic cavity was formed in the epicenter, and the structure of the gray matter was completely destroyed (Figures 3B1 & 3B2). Furthermore, to assess the progress of cystic cavity changes, histological staining was carried out at the seventh week in the control lesion (Figures 3C1 & 3C2), hiNSC-transplanted (Figures 3D1 & 3D2) and fibroblast-transplanted (Figures 3E1 & 3E2) groups. Our results showed that the cystic cavity was improved at the injured site in the hiNSC-transplanted group during the seventh week in comparison with the fibroblast-transplanted and control lesion groups. However, the anatomical structure of the spinal cord was not improved and irregular cell arrangement between collagen scaffolds was observed in the injured site after seven weeks.
Discussion
In the present research, we used hiNSCs which were derived from human fibroblasts by a trans-differentiation method established in our previous study [17]. Studies demonstrated NSCs have a high capacity for the treatment of SCI because of their ability to differentiate into neural-like cells and replace them with the damaged neurons [22]. Accordingly, they release neurotrophic factors that are useful in decreasing inflammation, controlling glial scar formation and stimulation of axonal growth at the injured site [20, 22]. According to the trans-differentiation method, we can produce patient-specific cell lines which are devoid of immunogenicity, suppressing the immune rejection in patients [23, 24]. Having typical morphology of NSCs, expressing specific protein markers of them, and the ability of differentiation into the three main lineages of neural cells are the most important indications that produced cells have exactly the behaviour of natural NSCs. Trans-differentiated hiNSCs induced by the Zfp521 gene, express NESTIN, SOX1, and PAX6, confirming the cell characteristics of NSCs [17]. They also were capable of differentiating spontaneously in vitro into TUJ1- and GFAP- cells implying that these cells are totally potent to make the three lineages of neural cells [17]. These data confirm that Zfp521 can Trans-differentiate fibroblasts into typical NSCs. So, we expected these cells to have the ability of regeneration in SCI rats.
Then, hiNSCs were transplanted into sub-acute rats which underwent SCI to determine whether these cells can reduce SCI-induced disability. One week later, the presence of hiNSCs was corroborated after the transplantation process. Although GFP-labelled cells became faded and few cells were evident, the markers of hNu, hTuj1, and hGFAP which are specific for human cells were used to probe these cells. The transplanted hiNSCs had the capability to differentiate into hTuj1- and hGFAP-positive cells. Although olig2- or PDGFRa-positive human cells were not detected in our study, in our previous study which was the combination of hNSCs and lithium chloride was applied, PDGFRa-positive cells were visible [25]. Therefore, it seems that combinatorial approaches could be more effective on oligodendrocyte differentiation. Some studies examined the survival, migration, and differentiation of NSCs in the spinal cord [25-27]; however, according to other studies conducted on animal models, 1% to 37% of transplanted cells were viable and able to escape from the immune system at the end of the study [28]. Altogether, it can be concluded besides the viability of the hiNSCs, they could differentiate and migrate in the damaged spinal cord which is a substantial point that proves these trans-differentiated cells act exactly like typical ones.
Regarding these obtained data, it is conceivable that suitable combination therapy might promote the differentiation of NSCs with higher cell viability. Besides, several lines of evidence demonstrated that using growth factors, such as bFGF and EGF, as well as PDGF-AA, increases cell survival and proliferation. So, the combinatory use of these factors or any other accessory factors with these cells may enhance the efficiency of cell therapy and protect the cells from the inflamed environment of the damaged tissue [29].
Locomotor recovery was examined through the BBB and Grid walk tests. Our results suggest that locomotor activity was improved in the hiNSC-transplanted group in comparison to the fibroblast-transplanted, sham, and control lesion groups in terms of the BBB scores. This score was plotted in a chart as of the seventh week, denoting that the efficacy of treatment was quite effective [21]. The least BBB score was observed in the fibroblast-transplanted group when compared with other experimental groups (even the control group). Although the difference in the BBB score between the control and fibroblast-transplanted groups was not statistically significant, it may cast doubt on the higher risk of glial scar formation locomotor dysfunction in rats receiving fibroblasts. The results of the Grid walk test showed a less significant difference in terms of the number of foot fault errors suggesting a higher superiority of hiNSCs over the fibroblast and other vehicle treatments. Therefore, these results indicate that hiNSCs could result in locomotor improvement in a SCI model when it comes to the Grid walk and BBB tests. These data confirmed that hiNSCs effectively contribute to the reconstruction of sensory-motor pathways in the spinal cord.
In this study, we investigated the cavity size found in the spinal cord to examine whether the transplanted hiNSCs could participate in repairing cavities. At first, to confirm SCI, 6 days after SCI surgery, animals were transcardially perfused with 4% paraformaldehyde, and their spinal cord tissue was stained with the hematoxylin-eosin staining method. The presence of lesions and loss of approximately 60% of cells in this region represents the severe damage in the spinal cord as evident in the longitudinal and transverse sections [30]. Afterward, spinal cord tissues, which were sectioned and stained with hematoxylin and eosin, seven weeks post-SCI showed a marked improvement in the size of cavities in the hiNSC-transplanted group as compared with other experimental groups. The presence of lesions and loss of approximately 60% of cells in this region represents the severe damage in the spinal cord as evident in the longitudinal and transverse sections [30]. These findings restoring the cell numbers of the cavity in the hiNSCs-transplanted group suggest that the existence of these cells could be beneficial for the repair of cavities and accelerating the recovery process. Other studies showed that transplanted NSCs improve difficulties caused by damage to the spinal cord through the production of neurotrophic factors [20, 22].
Conclusion
Our study showed that transplantation of hiNSCs trans-differentiated by the overexpression of the ZFP521 gene could improve SCI difficulties especially motor abilities, cavity formation and rewiring spinal cord tracts. In the case of the safety of trans-differentiation, as well as the efficient production of hiNSCs, the application of these cells may enrich the armamentarium of researchers in their quest for the treatment of SCI.
Acknowledgements
Special thanks to Prof. Hossein Baharvand, Head of the Stem Cell Biology and Technology Department, for his supportive guidance during the entire research.
Conflicts of Interest
None.
Funding
The present research was funded by the Cell Science Research Center, Royan Institute for Stem Cell Biology and Technology (ACECR).
Author Contributions
A. M.: Participated in drafting the manuscript and preparation of figures; M. K.: Performed spinal cord injury surgery, hiNSCs transplantation, histological assessments, behavioral assessments; E. Sh.: Carried out the cell culture of hiNSCs and their identification; A. A.: Participated in animal care and behavioral assessments; A. Kh.: Assisted in animal care; Z. Gh.: performed editing and approving the manuscript; A. R. V.: performed editing and approving the manuscript; V. R. M.: performed editing and approving the manuscript; S. K.: Contributed to the study design, manuscript writing, and research supervision and supporting research grant.
Article Info
Article Type
Research ArticlePublication history
Received: Sat 22, Aug 2020Accepted: Tue 01, Sep 2020
Published: Thu 10, Sep 2020
Copyright
© 2023 Sahar Kiani. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.NNB.2020.03.12
Author Info
Atiyeh Mohammadshirazi Maedeh Kashkouli Ebrahim Shahbazi Hassan Asghari Abolfazl Kheymeh Zahra Ghodsi Alexander R Vaccaro Vafa Rahimi-Movaghar Sahar Kiani
Corresponding Author
Sahar KianiDepartment of Brain and Cognitive Sciences, Cell Science Research Center, Royan Institute for Stem Cell Biology and Technology, ACECR, Tehran, Iran
Figures & Tables
References
- GBD 2016 Traumatic Brain Injury and Spinal Cord Injury Collaborators (2019) Global, regional, and national burden of traumatic brain injury and spinal cord injury, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol 18: 56-87. [Crossref]
- Seyed Behzad Jazayeri, Sara Beygi, Farhad Shokraneh, Ellen Merete Hagen, Vafa Rahimi Movaghar, (2015) Incidence of traumatic spinal cord injury worldwide: a systematic review. Eur Spine J 24: 905-918. [Crossref]
- Vafa Rahimi Movaghar, Mohammad Kazem Sayyah, Hesam Akbari, Reza Khorramirouz, Mohammad R Rasouli et al. (2013) Epidemiology of traumatic spinal cord injury in developing countries: A systematic review. Neuroepidemiology 41: 65-85. [Crossref]
- Elisa Garcia, Roxana Rodríguez Barrera, Jose Mondragón Caso, Horacio Carvajal and Antonio Ibarra (2018) Pharmacological and Nonpharmacological Therapeutic Strategies Based on the Pathophysiology of Acute and Chronic Spinal Cord Injury. Essentials of Spinal Cord Injury Medicine 23-55.
- Jia Sheng Rao, Can Zhao, Aifeng Zhang, Hongmei Duan, Peng Hao et al. (2018) NT3-chitosan enables de novo regeneration and functional recovery in monkeys after spinal cord injury. Proc Natl Acad Sci U S A 115: E5595-E5604. [Crossref]
- Kyriakos Dalamagkas, Magdalini Tsintou, Alexander M Seifalian (2018) Stem cells for spinal cord injuries bearing translational potential. Neural Regen Res 13: 35-42. [Crossref]
- Shushi Kabu, Yue Gao, Brian K Kwon, Vinod Labhasetwar (2015) Drug Delivery, Cell-based Therapies, and Tissue Engineering Approaches for Spinal Cord Injury. J Control Release 219: 141-154. [Crossref]
- Maria Teresa Dell'Anno, Stephen M Strittmatter (2017) Rewiring the spinal cord: Direct and indirect strategies. Neurosci Lett 652: 25-34. [Crossref]
- Ana Paula Pêgo, Sarka Kubinova, Dasa Cizkova, Ivo Vanicky, Fernando Milhazes Mar et al. (2012) Regenerative medicine for the treatment of spinal cord injury: more than just promises? J Cell Mol Med 16: 2564-2582. [Crossref]
- Margaret Coutts, Hans S Keirstead (2008) Stem cells for the treatment of spinal cord injury. Exp Neurol 209: 368-377. [Crossref]
- Sandrine Thuret, Lawrence D F Moon, Fred H Gage (2006) Therapeutic interventions after spinal cord injury. Nat Rev Neurosci 7: 628-643. [Crossref]
- Wolfram Tetzlaff, Elena B Okon, Soheila Karimi Abdolrezaee, Caitlin E Hill, Joseph S Sparling et al. (2011) A Systematic Review of Cellular Transplantation Therapies for Spinal Cord Injury. J Neurotrauma 28: 1611-1682. [Crossref]
- Anna Badner, Ahad M Siddiqui, Michael G Fehlings (2017) Spinal Cord Injuries: How Could Cell Therapy Help? Expert Opin. Biol. Ther 17: 529-541. [Crossref]
- Anna Lena Hallmann, Marcos J Araúzo Bravo, Christina Zerfass, Volker Senner, Marc Ehrlich et al. (2016) Comparative transcriptome analysis in induced neural stem cells reveals defined neural cell identities in vitro and after transplantation into the adult rodent brain. Stem Cell Res 16: 776-781. [Crossref]
- Yicheng Zhu, Naohiro Uezono, Tetsuro Yasui, Kinichi Nakashima (2018) Neural stem cell therapy aiming at better functional recovery after spinal cord injury. Dev Dyn 247: 75-84. [Crossref]
- Jamal Bamoulid, Oliver Staeck, Fabian Halleck, Dmytri Khadzhynov, Susanne Brakemeier et al. (2015) The need for minimization strategies: current problems of immunosuppression. Transpl Int 28: 891-900. [Crossref]
- Ebrahim Shahbazi, Sharif Moradi, Shiva Nemati, Leila Satarian, Mohsen Basiri et al. (2016) Conversion of Human Fibroblasts to Stably Self-Renewing Neural Stem Cells with a Single Zinc-Finger Transcription Factor. Stem Cell Reports 6: 539-551. [Crossref]
- D M Basso, M S Beattie, J C Bresnahan (1995) A Sensitive and Reliable Locomotor Rating Scale for Open Field Testing in Rats. J Neurotrauma 12: 1-21. [Crossref]
- Dimpy Gupta, Charles H Tator, Molly S Shoichet (2006) Fast-gelling injectable blend of hyaluronan and methylcellulose for intrathecal, localized delivery to the injured spinal cord. Biomaterials 27: 2370-2379. [Crossref]
- P Lu, L L Jones, E Y Snyder, M H Tuszynski (2003) Neural stem cells constitutively secrete neurotrophic factors and promote extensive host axonal growth after spinal cord injury. Exp Neurol 181: 115-129. [Crossref]
- D M Basso, M S Beattie, J C Bresnahan (1996) Graded Histological and Locomotor Outcomes after Spinal Cord Contusion Using the NYU Weight-Drop Device versus Transection. Exp Neurol 139: 244-256. [Crossref]
- Aikeremujiang Muheremu, Jiang Peng, Qiang Ao (2016) Stem cell based therapies for spinal cord injury. Tissue Cell 48: 328-333. [Crossref]
- Penelope J Hallett, Michela Deleidi, Arnar Astradsson, Gaynor A Smith, Oliver Cooper et al. (2015) Successful function of autologous iPSC-derived dopamine neurons following transplantation in a non-human primate model of Parkinson’s disease. Cell Stem Cell 16: 269-274. [Crossref]
- Lauren Host, Mandana Nikpour, Alicia Calderone, Paul Cannell, Janet Roddy (2017) Autologous stem cell transplantation in systemic sclerosis: A systematic review. Clin Exp Rheumatol 106: 198-207. [Crossref]
- Atiyeh Mohammadshirazi, Hoda Sadrosadat, Razieh Jaberi, Masoomeh Zareikheirabadi, Sara Mirsadeghi et al. (2019) Combinational therapy of lithium and human neural stem cells in rat spinal cord contusion model. J Cell Physiol 234: 20742-20754. [Crossref]
- Marina Gazdic, Vladislav Volarevic, C Randall Harrell, Crissy Fellabaum, Nemanja Jovicic et al. (2018) Stem Cells Therapy for Spinal Cord Injury. Int J Mol Sci 19: 1039. [Crossref]
- Hideyuki Okano, Yuto Ogawa, Masaya Nakamura, Shinjiro Kaneko, Akio Iwanami et al. (2003) Transplantation of neural stem cells into the spinal cord after injury. Cell Dev Biol 14: 191-198. [Crossref]
- Mitra J Hooshmand, Christopher J Sontag, Nobuko Uchida, Stan Tamaki, Aileen J Anderson et al. (2009) Analysis of Host-Mediated Repair Mechanisms after Human CNS-Stem Cell Transplantation for Spinal Cord Injury: Correlation of Engraftment with Recovery. PLoS One 4: e5871. [Crossref]
- Lars Olson (2013) Combinatory treatments needed for spinal cord injury. Exp Neurol 248: 309-315. [Crossref]
- T Cheriyan, D J Ryan, J H Weinreb, J Cheriyan, J C Paul et al. (2014) Spinal cord injury models: a review. Spinal Cord 52: 588-595. [Crossref]
