Ribonucleases and Immunoribonucleases as Potential Modalities of Anticancer Therapy
A B S T R A C T
A novel approach of antitumor treatment, that involves targeting RNA either using specific antisense oligonucleotides or cytostatic/cytotoxic ribonucleases (RNases), is being promoted. Among recently described cytotoxic RNases, amphibian RNases, including ranpirnase (onconase; ONC) and Amphinase (r-Amph), are promising anticancer agents. They manifest strong antitumor effects and act synergistically with several cytostatics. Recently, rapidly developed proteins by engineering of RNases, displayed cytotoxic activity against several types of malignant cells. Most recent data show the role of microRNAs in mediating tumor progression, opening a new field of possible molecular targets for RNases. This review summarizes the current status of those RNases and immunoRNases as promising novel anticancer therapeutics.
Keywords
Rana pipiens, onconase, r-amphinase, ribonuclease, immunoRNases, apoptosis, anticancer properties
Introduction
Antitumor ribonucleases (RNases) are a promising superfamily of small (10-28 kDa) basic proteins with strong cytotoxic and cytostatic potential on cancer cells [1]. This group of secretory enzymes operates at the crossroads of transcription and translation, preferentially degrading tRNA, and as a result, lead to inhibition of protein synthesis. After an early enthusiasm, their anticancer potential in terms of clinical utility suffered a decline until quite recently, when they have attracted attention again, due to the discovery of their remarkable and complex biological activities. Among various RNases described so far, only some of them are considered to be toxic to cancer cells, but they have low cytotoxicity to their normal counterparts. Antitumor potential was observed in RNases derived from bacteria (binase), fungi (streptomycete ribonuclease), mammals (bovine seminal ribonuclease), and last but not least from reptiles [2]. Rana pipiens oocytes contain two homologues of pancreatic RNase A, the widely investigated ranpirnase (onconase; ONC), which has reached the late-stage clinical trials for treatment of malignant mesothelioma, and the more recently described amphinase (Amph) as well as its recombinant form (r-Amph) [3].
It was already discovered that ONC acts strongly synergistic when combined with numerous other antitumor agents and has been granted orphan-drug and fast-track status by the US Food and Drug Administration. Moreover, ONC and Amph have preferential toxicity towards cancer cells [3]. They are highly cationic molecules and have a distinctly higher negative charge compared to the normal cell, which can be a possible explanation of their increased binding efficiency to the cell surface by electrostatic interactions. Mainly due to their cytostatic function, causing minimal side effects, RNases are considered as attractive agents. In addition, RNases are known to inhibit human immunodeficiency virus 1 (HIV-1) replication through the degradation of viral RNA [4].
In this article, we describe structures, functions and mechanisms of antitumor activity of RNases with particular emphasis to the amphibian ONC and Amp. The advancements of recombinant technology have allowed the assembly and conjugation of RNases with monoclonal antibodies, investigated in a variety of human malignancies both in vitro and in animal models. Such combinations called immunoribonucleases (immunoRNases) internalize tumor-targeting and has demonstrated selective antitumor activity against cancer cell [5]. In this review, we describe the development and current status of genetically engineered immunoRNases as some of the most promising novel chemotherapeutic agents given their enzymatic and non-mutagenic character.
Historical Introduction to RNases
RNases have been an extensively studied group of enzymes, since the 1960s. For the last century, they were an important model for the majority of studies concentrating on protein chemistry and folding, stability and enzymatic catalysis. Recent investigation on the RNase superfamily has focused mainly on exploring their biological functions and medical applications. In 1972, Moore, Stein, and Anfinsen were awarded a Nobel prize for their work on RNase. Bovine seminal ribonuclease (BS-RNase), was the first homolog among RNase family, that show to reduce transplanted Crocker tumors in mice in 1973. The toxicity of BS-RNase was not limited only to tumor cells in vivo but was also toxic to embryonic cells, oocytes, and testicular tissue [6-8]. After enormous early enthusiasm, RNases were almost dismissed in terms of their clinical use until quite recently when they attracted attention again due to the discovery of their remarkable and complex biological activities [9, 10]. For instance, they are able to promote angiogenesis, possess immunosuppressive and antitumor activity, and demonstrate aspermatogenic and embryotoxic effects [11, 12].
In the early 1970s, it was observed by Shogen and Yoon that the extract from embryos of the Northern leopard frog has a strong cytotoxic activity [13]. Twenty years later, it was found that cytotoxicity of the extract is due to the presence of a protein initially named Pannon, then ranpirnase (onconase; ONCONASE®; ONC) from the first letters of Rana pipiens RNase and subsequently Amph [14, 15]. Both enzymes ONC and Amph were discovered and sequenced by Alfacell Corporation (currently TAmiR Biotechnology, Inc, Somerset, NJ, USA), the first one more than two decades ago, while the latter more recently. Nowadays, the advancements of recombinant technology have allowed to assembly and conjugation of RNases with tumor-targeting monoclonal antibodies to enhance specific cancer cell killing. The immunoRNases are recently under investigation in several malignant diseases, both in vitro and in animal models. In this review, we describe the current status of immunoRNases in clinical trials as a novel compound against different types of cancer, which still remains a major challenge for further clinical development [5].
Structure and Biochemical Attributes
ONC and Amph, like most cytotoxic RNases, are basic, single chain proteins. Amino acid sequencing revealed that both ONC and Amph belong to the RNase A superfamily [16]. Amph is the largest among frog homologues and consists of 114 amino acid residues, while ONC, on the other hand, is the smallest of the whole superfamily, having only 104 amino acid residues (20 residues less than RNase A) and a molecular mass of 11.8 kDa. Amph can be found in 4 variants, numbered from 1-4 according to their elution order from a reverse phase HPLC column. All four have highly similar amino acid sequences with 95 residues are invariant (83% conservation) [17]. Amph 1 and 2 differ between each other only by one residue at position 44 (Val and Ile, respectively), while the other variants differ from each other by 12-15 residues (86.8-89.9% identity) at 19 polymorphic positions. The variants are 38.2-40.0% identical with ONC, 40.7-42.5% and 24.8-28.0% with RNase A. Characteristic for ONC and other frog RNases, N-terminal pyroglutamic acid residue, is not observed in Amph variants. They have a highly polar N-terminal extension segment consisting of 6 amino acid residues, which makes them more like mammalian than to amphibian homologues [18].
It was earlier observed that several frog lectins from the eggs of Rana catesbeiana with the amino acid sequence similar to that of RNase A could selectively agglutinate cancer cells [19]. Other homologous lectin was isolated from oocytes of Rana japonica and Rana catesbeiana liver [20, 21]. Both their sequences were highly similar to that of RNase A and had a very low ability to agglutinate cancer cells. ONC and Amph did not agglutinate cancer cells at all. More recently, five more cytotoxic RNases from Rana catesbeiana were purified and cloned [22]. All those RNases clearly belong to the superfamily of RNase A. Examples for cytotoxic and non-cytotoxic members of the RNase A superfamily are shown in (Table 1).
Table 1: Examples for cytotoxic and non-cytotoxic members of the RNase A superfamily.
|
Toxicity |
RNase |
Source |
|
toxic |
BS-RNase |
bovine seminal fluid |
|
toxic |
ONC |
Rana pipiens |
|
toxic |
sialic acid binding lectin (SBL-J / RJL) |
Rana japonica |
|
toxic |
sialic acid binding lectin (SBL-C / RCL) |
Rana catesbeiana |
|
toxic |
Amph 1-4 |
Rana pipens |
|
non-toxic |
RNase A |
bovine pancreas |
|
non-toxic |
RNase 1 |
human pancreas |
|
non-toxic |
eosinophil-derived neurotoxin (EDN) |
human eosinophils |
|
non-toxic |
angiogenin (ANG) |
human serum |
Mechanisms of Cytotoxic Activity
It was reported that ONC is more toxic to neoplastic cells than to normal cells in both in vitro and in vivo studies. The mechanism responsible for this phenomenon is not still clear, and several explanations have been proposed. According to one of the possible hypothesis ONC is more selectively internalized by tumor compared to normal cells due to differences in charge of the plasma membrane [23]. Specifically, in neoplastic cells, the level of sialic acid, rich in gangliosides, is markedly higher, which contributes to electro-negativity of the plasma membrane [24]. Because ONC is strongly cationic, it has a greater electrostatic affinity to the anionic surface and thus is more avidly internalized by tumor cells. ONC, after binding to the cell surface, is internalized by energy-dependent endocytosis, internalized and routed to endosomes. It was observed that agents that disrupt retrograde transport from the trans-Golgi network to the endoplasmic reticulum potentiate the cytotoxic effect of ONC [25, 26]. Another proposed mechanism takes into account different intracellular routes for ONC in neoplastic versus normal cells with different respective rates of RNA degradation, especially in fast-growing tumors [27].
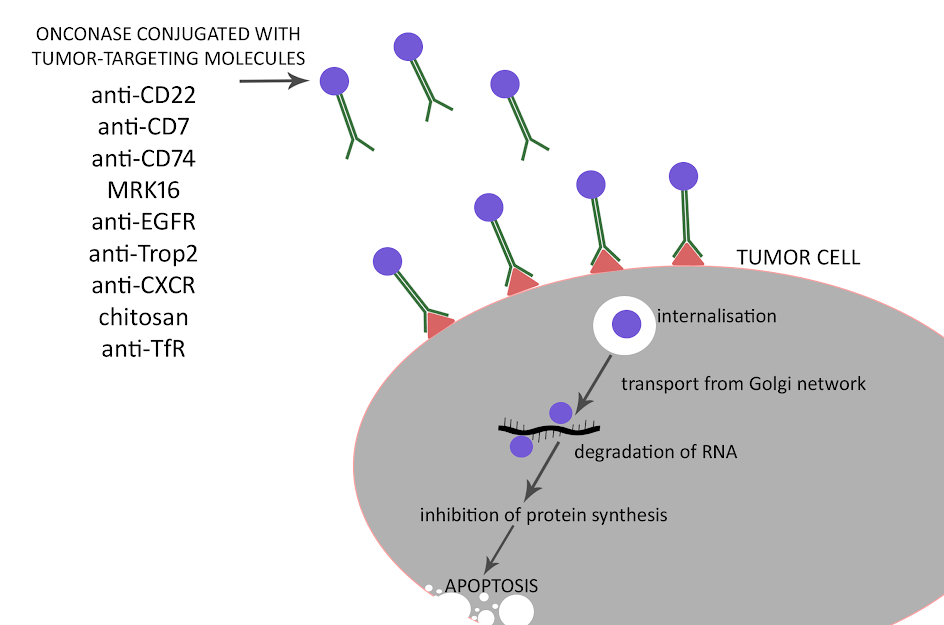
The cytostatic and cytotoxic properties of ONC and r-Amph depend on their enzymatic activity targeting the crossroads between transcription and translation and catalyzing RNA degradation. It was initially considered that the key targets of these RNases are rRNA and/or tRNA, and their degradation led to inhibition of protein synthesis [28]. According to recent findings, however, the suspected target of RNases can also be the non-coding RNA (microRNAs), which is responsible for the regulation of gene expression through RNA interference (RNAi). Consistent with the latter they can as well modulate the process of cell differentiation in the early embryos by strictly targeting RNAi. The mechanism of action is shown on (Figure 1).
Figure 1: Mechanism of action of cytotoxic RNase. Cytotoxicity of RNase is connected with internalization particularly to cancer cells and after intracellular transport degradation of different types of RNA (mRNA, rRNA, tRNA) into smaller components. It leads to inhibition of protein synthesis and as a result programmed cell death (apoptosis).
While ONC penetrates into the cytosol, it degrades cellular RNA. ONC is an unusual homologue of RNase that seems to evade the cytosolic ribonuclease inhibitor protein (RI) completely [29]. RI is a protein that binds to certain members of the RNase A superfamily and inactive them. The complex made of human RI and RNase is one of the tightest known in biology so far [30]. It is believed that some RNases are tightly inhibited by RI, yet become cytotoxic when successfully routed into the cytosol. Moreover, it was observed that non-cytotoxic so far RNases become cytotoxic by incorporating residues that enable RI evasion [31]. On the other hand, RI insensitivity is not considered as a main factor responsible for the antitumor activity of RNases.
The supposed antitumoral and immunosuppressive mechanism of ONC involves apoptosis in the case of the majority of neoplastic cells [32]. The cytostatic effects of ONC and Amph are manifested as cell arrest in the G1 cell cycle phase and followed by apoptosis, involving activation of endonucleases such as caspases, serine proteases and transglutaminase. In previous studies, it was observed that ONC selectively alters the expression of genes involved in apoptosis with decreased expression of anti-apoptotic Bcl-2 as well as increased expression of pro-apoptotic Bax [33]. Interestingly, opposite to the majority of well-known chemotherapeutic agents that act rather rapidly, the cytostatic and cytotoxic effects of ONC was observed after a 24-48 h delay [34]. The induced apoptosis is probably enhanced by mild hyperthermia [35].
Antiviral Effect
Onconase is investigated as a potential antiviral drug due to its degradational action on RNA, both in the viral genome and in viral replication in host cells [36]. In studies with human immunodeficiency virus (HIV) and CD4 positive lymphocytes (H9), onconase inhibited viral replication, which was explained as the result of targeting tRNA and further impaired viral protein synthesis [37, 38]. In view of the gastrointestinal tract became the main space of HIV penetration, onconase was also tested as a topical drug preventing this infection [3]. in vitro, onconase inhibited six of nine activated by HIV biomarkers necessary to initiation of HIV infection [39]. Therefore, onconase seems to be a potential preventing agent.
Hodge et al. revealed the effectiveness of onconase against Ebola virus infection in vitro [40]. Then the study in mice models was performed. The results showed that most of the mice were protected from the consequences of Ebola infection by treatment with onconase, and the only observed adverse event was weight loss. In study with Rabies virus, onconase was efficient in vitro; however, this action was not confirmed in vivo [41]. As a result of onconase effectiveness against human papillomavirus (HPV) infection in vitro, phase I study was performed [42]. 42 patients were enrolled in this trial, and 30 of them were evaluated after 8 weeks of topical use of 1mg onconase. 83,3% (25 patients) achieved clinical healing, and the rest of them (5 patients, 16,7%) presented 50% reduction of lesions. The median time to curing was significantly shorter in comparison to spontaneous healing (30 days vs. 6-7 months). The most commonly observed adverse event was itching.
Bactericidal Activity
Wild type onconase does not exhibit antimicrobial effect in contrast to human RNase 3 (EDN) also belongs to RNase A family. Torrent et al. engineered variants of onconase with determinants homologous to RNase 3, which are described as responsible for bactericidal activity [43]. These new molecules were equipped in antimicrobial influence against Gram-negative bacteria especially K. pneumoniae. Each performed modification resulted in the decrease of ribonucleolytic action. The absence of cytotoxic activity did not remove bactericidal effect completely; however, the ribonucleolytic activity had an important impact for antimicrobial effect of engineered onconases.
Therapeutic Applications
Several RNases from different sources were proved to be cytotoxic against cancer cells, whereas the application of the other, closely related RNases had no antitumor activity. It was shown that ribonucleolytic activity is not sufficient to render RNase cytotoxicity. Among various RNases described so far, anticancer potential was observed in RNases derived from bacteria (binase), fungi (streptomycete ribonuclease), mammals (bovine seminal ribonuclease) and the most important from reptiles (ONC, Amph). ONC and Amph are widely investigated in many clinical trials, due to the discovery of their remarkable and complex biological activities [44]. Cytotoxic effect of in vitro ONC alone and in combination with other antitumor agents was already investigated in several clinical studies. It was observed that ONC acts strongly synergistic when combined with numerous well-known chemotherapeutic drugs. Synergism was reported for a combination with either tamoxifen or trifluoperazine on human lung carcinoma or pancreatic adenocarcinoma cell lines [45].
After this study, numerous subsequent investigations were performed. In all of them, synergism or additive effects of ONC was revealed in combination with a wide variety routinely used agents such as lovastatin, tumor necrosis factor α, interferon, vincristine, cepharanthine or rituximab, mafosfamide, vincristine, doxorubicin, and dexamethasone [46-51]. The striking feature is that the discovered synergism was in combinations with various antitumor agents with completely different mechanisms of action. Fagagnini et al. investigated that onconase is able to form a dimer which retains activity of its native monomer [52]. They also treated human pancreatic adenocarcinoma cells (PaCa44) with onconase-dimer (ONC-D) and onconase-monomer (ONC-M) alone or in combination with gemcitabine. The results showed that ONC-D was significantly more cytotoxic in comparison with ONC-M. This antitumor activity was further strengthened by combination with gemcitabine.
ONC has already been studied in phase II and phase III clinical trials. In phase II ONC as a single agent was investigated in patients with non-small-cell lung cancer, renal cell cancer, breast cancer and also in combination with tamoxifen in prostate cancer and pancreatic cancer [53-57]. ONC proved to be superior to doxorubicin in a phase III trial in malignant mesothelioma patients, while preliminary results of another large, phase III trial in malignant mesothelioma patients suggest that the combination of ONC with doxorubicin may be more beneficial than doxorubicin used alone [58]. In all studies mentioned above, ONC seems to act mainly like cytostatic rather than a cytotoxic agent, stabilizing progressive disease and potentially offer survival benefit, while having a favorable safety profile. ONC, in combination with doxorubicin, may become a second-line therapy for unresectable malignant mesothelioma, where so far no standard of care exists. Unfortunately, during the course of this trial, the combination of pemetrexed and platinum emerged as the standard first-line treatment, which as a result led to a slowing of the recruitment and reduced the interest for its use.
It is crucial that ONC appears to be generally well tolerated agent with very few serious adverse events. Majority of side effects are predictable and reversible. The most important side effect is renal toxicity, which seems to be caused mainly by the unusual stability of the enzyme [59]. What is more, dose modifications are required only in patients with observed changes in renal function. Clinical trials also demonstrated that onconase does not cause immunological sensitization. Furthermore, Raineri et al., in study with human melanoma cells (A375), showed that onconase entailed similar cytotoxicity in both parental and dabrafenib-resistance cell lines [60]. They also explored how onconase reduced the cell mobility and creating cell’s colony, which suggests its potential to inhibit forming metastases. So far, the use of RNases as adjunct agents to increase the effectiveness of treatment of different malignancies with chemotherapy or radiotherapy appears to be the most promising direction in its clinical applications.
In turn, Lee et al. revealed sensitizing to radiotherapy effect of onconase in vitro on human lung cancer cells (A549) [61]. Next they confirmed this action in mice models with the best results if the onconase was given 2h prior radiation. This sensitizing action of onconase was explained by increasing concentration of oxygen in tumor cells and decreasing tumor intestinal fluid pressure (TIFP).
Targeted Therapeutic RNases (ImmunoRNases)
Increasing the selectivity of onconase on cancer cells and facilitating its internalization is possible due to conjugation with tumor-targeting monoclonal antibodies. Fusion with humanized antibody directed to CD22 antigen specific to B-cells was tested in vitro on human lymphoma cell lines (Daudi and CA-46) and in animal models [62, 63]. in vitro, onconase was significantly faster internalized, and its cytotoxicity enhanced 10 000-fold when joined to antibody. in vivo , mice bearing Daudi lymphoma cells treated with onconase coupled to anti-CD22 antibody had the longest life span in comparison to onconase alone, antibody alone and their mixture treatment. It was also evidently less toxicity. Weber et al. performed and studied fusion anti-CD22 antibody with one and more molecules of onconase [64]. Cytotoxic effect in in vitro trial was the highest for conjugation with the number of three onconase. However, when 4-12 molecules were attached, the cytotoxicity decreased significantly.
Presenting on the surface of most T and NK cells but overexpress on T-cell acute lymphoblastic leukemia (A-LL), rapidly internalized CD7 antigen was also chosen for the purpose of target treatment. Onconase attached to antibody directed to CD7 antigen was studied in vitro on T-lymphoblastic leukemia cells (Molt 3) [65]. The results showed that this fusion caused 1000-fold increased activity of onconase. Another conjugation with CD74-directed antibody was targeted for treatment non-Hodgkin lymphomas (NHL) and multiple myeloma [66]. It was successfully tested in vitro reached high and specific cytotoxicity. Mice bearing NHL cells (Daudi and Raji) cured with this immuno-onconase achieved longer median survival time and improved therapeutic effect in comparison to saline and mixture of anti-CD74 antibody with onconase treatment. An observed adverse event was liver toxicity.
MRK16 is an antibody directed to membrane protein named Pgp, which is responsible for multidrug resistance (MDR) in cancer cells. It is known that using this molecule enables the cytotoxic action of vincristine. Newton et al. investigated whether fusion MRK16 with onconase modulated the activity of MRK16 [67]. Tested immuno-onconase was rapidly internalized (after 1h cure) in in vitro multidrug resistance human colon carcinoma cell line (HT-29) trial and more efficient potentialized action of vincristine. In mice model, treatment with onconase conjugated to MRK16 combined with vincristine was more potent than single onconase and vincristine but less effective than single onconase with MRK16 and vincristine.
Onconase was also attached to monoclonal antibody directed to epidermal growth factor receptor (EGFR), whose increased presentation worsened the prognosis in different cancers (head and neck, bladder, ovarian, cervical) [68]. In in vitro study, Kiesgen et al. noted significantly faster internalization and higher antitumor activity of onconase when fused to antibody in comparison to native onconase (1-2h vs. 24h) in EGFR positive cancer cells lines (human head and neck squamous cell lines and human epidermoid carcinoma cell line). These results were also confirmed in tumor-bearing mice models. Besides, mice treated with immuno-onconase lived significantly longer and did not present renal failure. To improve its efficiency of cytosolic translocation, EGFR-directed antibody-onconase connection was equipped in sequence responsible for the endosomal escape mechanism of the dengue virus [69]. This fusion exhibited increased cytotoxicity in vitro. Likewise, amphinase was joined with TNF-α to targeting EGFR [70]. in vitro, this fused molecule demonstrated increased pro-apoptotic efficiency in few EGFR-positive tumor cells lines in comparison to native-amphinase. Mice bearing breast carcinoma (MDA-MB-468) treated with amphinase attached to TNF- α showed significantly lower tumor volume than mice received amphinase alone.
It was investigated that the Trop-2 played an important role in tumorigenesis. This calcium signal transducer has accelerated expression in many cancers. in vitro, onconase attached to antibody-directed Trop-2 showed most antitumor impact for breast (T-74D, MDA-MB-468), lung adenocarcinoma (Calu-3) and cervical (ME-180) cancer cell lines [71]. It was also quickly internalized. In mice-bearing lung adenocarcinoma, significantly inhibit tumor growth was achieved for treatment with immuno-onconase in comparison to untreated control. The idea treatment targeting Trop-2 has been expanded by targeting to antibody more than one copy of onconase [72]. The immunotoxin contains four onconase molecules turned out to be 100-fold more effective than standard immuno-onconase and 3000-fold more potent in comparison to native onconase. This conjugation also increased life mice bearing breast cancer cells (MDA-MB-468).
Chemokine receptor (CXCR) is overexpressed in many types of cancers like colon, small-cell lung cancer, breast, ovarian, pancreatic, renal, prostate, glioma and haematological malignancies (melanoma, non-Hodgkin’s lymphoma, chronic lymphocytic leukemia, acute myeloid leukemia). Sequence binding CXCR was combined with onconase and tested in various cancer cell lines [73, 74]. Results showed increased internalization and cytotoxicity this treatment in comparison to native onconase. However, on the normal cell, there was no effect.
Chitosan is a specific targeting malignant gliomas peptide obtained from scorpion venom. However, it is deprived of antitumor activity. For this reason, conjugation with cytotoxic molecule became necessary to exploit it’s potential. Wang and Guo attached onconase to chitosan and subjected to testing [75]. In in vitro trial with human glioma cells (U251 and SHG-44) fusion of onconase and chitosan demonstrated significant superior cytotoxicity in comparison to onconase and chitosan mixture. Mice bearing human glioma cells treated with conjugated onconase and chitosan presented lower tumor weight due to higher inhibition tumor increase when compared to treatment with both saline and mixture of onconase and chitosan. Dose-dependent anti-glioma effect did not prove in vivo .
Following target of oncological treatment became the transferrin receptor (TfR), which is overexpressed in numerously cancers. Connection onconase and TfR-targeting antibody was performed to increase efficacy RNAse [76]. This assumption was confirmed in trial with TfR rich cancer cells (HepG2 and Hela). The results showed dose-depend cytotoxicity of onconase attached to TfR-antibody, which was significantly higher than single onconase activity. One of the reasons of the high glomerular filtration of onconase, which results in kidney injury and short circulating half time, is small molecular weight of this RNase [77]. To avoid it, the conjugations increasing the molecular size were performed. As a result of fusion onconase with albumin-chitosan polymer (hybrid nanocarrier) cationic molecule (named ONC-HNC) was created and tested in vitro with the human mesothelioma REN cells [78]. In comparison to onconase, ONC-HNC was more effectively delivered to cancer cells and significantly increased inhibition of cell viability. These results was explained by controlled release of onconase in the cytosol and positively charged ONC-HNC which facilitate endocytosis by negatively charged cancer-cell membrane. Also, conjugation onconase with both albumin and tumor-targeting antibody increased effectiveness of its delivery to tumor and cytotoxicity [44].
To further increase tumor-delivery onconase, Fernandez-Ulibarri et al. exploited adenovirus with transgene of connection onconase and antibody targeting EGFR [79]. Its cytotoxicity was revealed in vitro for oral adenosquamous carcinoma (Cal27), pancreatic carcinoma (Panc-1) and head and neck cancer (HNO210). In mice bearing epidermoid carcinoma (A431), this therapy after intratumoral injection significantly decreased tumor size. Another approach to internalization of onconase was to use photodynamic therapy [80]. This method proved effective in studies with squamous cell carcinoma of head and neck.
Conclusions/Future Perspectives
Although a major advancement in antitumor treatment has been observed, still several human malignancies remain incurable. A promising approach can be the one that involves targeting RNA. Of particular interest are numerous observations that ONC acts strongly synergistic while combined with one of the several different antitumor drugs. This was observed in vitro in combination with the agents used during standard treatment such as tamoxifen or trifluoperazine in solid tumors. Interestingly, in combination with vincristine, ONC has shown high toxicity even against multidrug resistant cells. Recently, there were attempts to bind ONC with antibody in order to increase toxicity selectively towards tumor cells. Fusion of a human monoclonal antibody with ONC will increase binding to tumor cells. Moreover, this protein displayed enhanced specificity, potency and decreased systemic toxicity in experiments on mice. The further direction of anticancer therapy is thought to involve aggressive protocols of chemotherapy in combination with targeted therapies. As discussed above, there is preclinical evidence that immunoRNases might be a new class of adjunct drugs, that will significantly enhance the therapeutic activity of standard chemotherapeutic treatments of various tumors. Such an approach may be a promising direction in further multidrug clinical applications. The full therapeutic potential and detailed mechanism of action still remain to be elucidated.
Acknowledgements
This work was supported by the grant from Ministry of Science/National Science Centre, Poland No 507-18-010 and in part, by the grant from Medical University of Lodz, Poland No 503/8-093-01/503-01.
Conflicts of Interest
None.
Article Info
Article Type
Review ArticlePublication history
Received: Tue 07, Jul 2020Accepted: Wed 22, Jul 2020
Published: Thu 10, Sep 2020
Copyright
© 2023 Piotr Smolewski. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.DDA.2020.01.04
Author Info
Monika Kowalik Magdalena Witkowska Piotr Smolewski
Corresponding Author
Piotr SmolewskiDepartment of Experimental Hematology, Medical University of Lodz, Poland
Figures & Tables
Table 1: Examples for cytotoxic and non-cytotoxic members of the RNase A superfamily.
|
Toxicity |
RNase |
Source |
|
toxic |
BS-RNase |
bovine seminal fluid |
|
toxic |
ONC |
Rana pipiens |
|
toxic |
sialic acid binding lectin (SBL-J / RJL) |
Rana japonica |
|
toxic |
sialic acid binding lectin (SBL-C / RCL) |
Rana catesbeiana |
|
toxic |
Amph 1-4 |
Rana pipens |
|
non-toxic |
RNase A |
bovine pancreas |
|
non-toxic |
RNase 1 |
human pancreas |
|
non-toxic |
eosinophil-derived neurotoxin (EDN) |
human eosinophils |
|
non-toxic |
angiogenin (ANG) |
human serum |
References
- Ardelt B, Ardelt W, Pozarowski P, Kunicki J, Shogen K et al. (2007) Cytostatic and cytotoxic properties of Amphinase, a novel cytotoxic ribonuclease from Rana pipiens oocytes. Cell Cycle 6: 3097-3102. [Crossref]
- Arnold U (2008) Aspects of the Cytotoxic Action of Ribonucleases. Curr Pharm Biotechnol 9: 161-168. [Crossref]
- Ardelt W, Shogen K, Darzynkiewicz Z (2008) Onconase and Amphinase, the antitumor ribonucleases from Rana pipiens oocytes. Curr Pharm Biotech 9: 215-225. [Crossref]
- Turcotte RF, Raines RT (2008) Design and Characterization of an HIV-Specific Ribonuclease Zymogen. AIDS Res Hum Retroviruses 24: 1357-1363. [Crossref]
- De Lorenzo C, D’Alessio G (2008) From immunotoxins to immmunoRNases. Curr Pharm Biotech 9: 210-214.
- Matousek J (1975) Embryotoxic effect of bull seminal ribonuclease and tissue absorption studies in rats. J Reprod Fertil 43: 171-174. [Crossref]
- Slavik T, Matousek J, Fulka J, Raines RT (2000) Effect of bovine seminal ribonuclease and bovine pancreatic ribonuclease A on bovine oocyte maturation. J Exp Zool 287: 394-399. [Crossref]
- Matousek J (1994) Aspermatogenic effect of the bull seminal ribonuclease (BS RNase) in the presence of anti BS RNase antibodies in mice. Anim Genet 25: 45-50. [Crossref]
- Castro J, Ribó M, Puig T, Colomer R, Vilanova M et al. (2012) A cytotoxic ribonuclease reduces the expression level of P-glycoprotein in multidrug-resistant cell lines. Invest New Drugs 30: 880-888. [Crossref]
- Leland PA, Schultz LW, Kim BM, Raines RT (1998) Ribonuclease A variants with potent cytotoxic activity. Proc Natl Acad Sci USA 95: 10407-10412. [Crossref]
- Goo SM, Cho S (2013) The expansion and functional diversification of the mammalian ribonuclease a superfamily epitomizes the efficiency of multigene families at generating biological novelty. Genome Biol Evol 5: 2124-2140. [Crossref]
- D’Alessio G, Di Donato A, Mazzarella L (1997) Seminal ribonuclease: the importance of diversity. In: D’Alessio G, Riordan JF, editors. Ribonucleases: structures and functions. New York, NY, USA: Academic Press 383-423.
- Shogen K, Yoan WK (1973) Antitumor activity in extracts of leopard frog (Rana pipiens) embryos. 27th Annual Eastern Colleges Science Conference; The Pennsylvania State University, State College.
- Darzynkiewicz Z, Carter SP, Mikulski SM, Ardelt WJ, Shogen K (1988) Cytostatic and cytotoxic effects of Pannon (P-30 Protein), a novel anticancer agent. Cell Tissue Kinet 21: 169-182. [Crossref]
- Dyer KD, Rosenberg HF (2006) The RNase A superfamily: generation of diversity and innate host defense. Mol Divers 10: 585-597. [Crossref]
- Ardelt W, Mikulski SM, Shogen K (1991) Amino acid sequence of an anti-tumor protein from Rana pipiens oocytes and early embryos. Homology to pancreatic ribonucleases. Biol Chem 266: 245-251. [Crossref]
- Singh UP, Ardelt W, Saxena SK, Holloway DE, Vidunas E et al. (2007) Enzymatic and structural characterisation of amphinase, a novel cytotoxic ribonuclease from Rana pipiens oocytes. J Mol Biol 371: 93-111. [Crossref]
- Leland PA, Raines RT (2001) Cancer chemotherapy: ribonucleases to the rescue. Chem Biol 8: 405-413. [Crossref]
- Titani K, Takio K, Kuwada M, Nitta K, Sakakibara F et al. (1987) Amino acid sequence of sialic acid binding lectin from frog (Rana catesbeiana) eggs. Biochemistry 26: 2189-2194. [Crossref]
- Kawauchi H, Sakakibara F, Watanabe K (1975) Agglutinins from frog eggs: a new class of proteins causing preferential agglutination of tumor cells. Experientia 31: 364-365. [Crossref]
- Nitta R, Katayama N, Okabe Y, Iwama M, Watanabe H et al. (1989) Primary structure of a ribonuclease from Bullfrog (Rana catesbeiana) liver. J Biochem 106: 729-735. [Crossref]
- Liao YD, Huang HC, Leu YJ, Wei CW, Tang PC et al. (2000) Purification and cloning of cytotoxic ribonucleases from Rana catesbeiana (bullfrog). Nucleic Acids Res 28: 4097-4104. [Crossref]
- James AM, Ambrose EJ, Lowick JH (1956) Differences between the electrical charge carried by normal and homologous tumour cells. Nature 177: 576-577. [Crossref]
- Fredman P (1993) Glycosphingolipid tumor antigens. Adv Lipid Res 25: 213-234. [Crossref]
- Haigis MC, Raines RT (2003) Secretory ribonucleases are internalized by a dynamin-independent endocytic pathway. J Cell Sci 116: 313-324. [Crossref]
- Rodriguez M, Torrent G, Bosch M, Rayne F, Dubremetz J et al. (2007) Intracellular pathway of Onconase that enables its delivery to the cytosol. J Cell Sci 120: 1405-1411. [Crossref]
- Turcotte RF, Lavis LD, Raines RT (2009) Onconase cytotoxicity relies on the distribution of its positive charge. FEBS J 276: 3846-3857. [Crossref]
- Benito A, Ribó M, Vilanova M (2005) On the track of antitumor ribonucleases. Mol Biosyst 1: 294-302. [Crossref]
- Dickson KA, Haigis MC, Raines RT (2005) Ribonuclease inhibitor: Structure and function. Prog Nucleic Acid Res Mol Biol 80: 349-374. [Crossref]
- Johnson RJ, McCoy JG, Bingman CA, Phillips GN Jr, Raines RT (2007) Inhibition of human pancreatic ribonuclease by the human ribonuclease inhibitor protein. J Mol Biol 367: 434-449. [Crossref]
- Leland PA, Staniszewski KE, Kim BM, Raines RT (2001) Endowing human pancreatic ribonuclease with toxicity for cancer cells. J Biol Chem 276: 43095-43102. [Crossref]
- Iordanov MS, Ryabinina OP, Wong J, Dinh TH, Newton DL et al. (2000) Molecular determinants of apoptosis induced by the cytotoxic ribonuclease onconase: Evidence for cytotoxic mechanisms different from inhibition of protein synthesis. Cancer Res 60: 1983-1994. [Crossref]
- Ardelt B, Juan G, Burfeind P, Salomon T, Wu JM et al. (2007) Onconase, an anti-tumor ribonuclease suppresses intracellular oxidative stress. Int J Oncol 31: 663-669. [Crossref]
- Zwolińska M, Smolewski P (2010) Onconase: a ribonuclease with antitumor activity. Postepy Hig Med Dosw 64: 58-66. [Crossref]
- Halicka HD, Ardelt B, Shogen K, Darzynkiewicz Z (2007) Mild hyperthermia predisposes tumor cells to undergo apoptosis upon treatment with onconase. Int J Oncol 30: 841-847. [Crossref]
- Vert A, Castro J, Ribó M, Benito A, Vilanova M (2017) Activating transcription factor 3 is crucial for antitumor activity and to strengthen the antiviral properties of Onconase. Oncotarget 8: 11692-11707. [Crossref]
- Youle RJ, Wu YN, Mikulski SM, Shogen K, Hamilton RS et al. (1994) RNase inhibition of human immunodeficiency virus infection of H9 cells. Proc Natl Acad Sci U S A 91: 6012-6016. [Crossref]
- Suhasini AN, Sirdeshmukh R (2007) Onconase action on tRNA(Lys3), the primer for HIV-1 reverse transcription. Biochem Biophys Res Commun 363: 304-309. [Crossref]
- Brand RM, Siegel A, Myerski A, Metter EJ, Engstrom J et al. (2018) Ranpirnase Reduces HIV-1 Infection and Associated Inflammatory Changes in a Human Colorectal Explant Model. AIDS Res Hum Retroviruses 34: 838-848. [Crossref]
- Hodge T, Draper K, Brasel T, Freiberg A, Squiquera L et al. (2016) Antiviral effect of ranpirnase against Ebola virus. Antiviral Res 132: 210-218. [Crossref]
- Smith TG, Jackson FR, Morgan CN, Carson WC, Martin BE et al. (2020) Antiviral Ranpirnase TMR-001 Inhibits Rabies Virus Release and Cell-to-Cell Infection In Vitro. Viruses 12: 177. [Crossref]
- Squiquera L, Taxman DJ, Brendle SA, Torres R, Sulley J et al. (2017) Ranpirnase eradicates human papillomavirus in cultured cells and heals anogenital warts in a Phase I study. Antivir Ther 22: 247-255. [Crossref]
- Torrent G, Ribó M, Benito A, Vilanova M (2009) Bactericidal activity engineered on human pancreatic ribonuclease and onconase. Mol Pharm 6: 531-542. [Crossref]
- Zhao HL, Xue C, Du JL, Ren M, Xia S et al. (2012) Sustained and cancer cell targeted cytosolic delivery of Onconase results in potent antitumor effects. J Control Release 159: 346-352. [Crossref]
- Mikulski SM, Viera A, Ardelt W, Menduke H, Shogen K (1990) Tamoxifen and trifluoroperazine (Stelazine) potentiate cytostatic/cytotoxic effects of P-30 protein, a novel protein possessing anti-tumor activity. Cell Tissue Kinet 23: 237-246. [Crossref]
- Mikulski SM, Viera A, Darzynkiewicz Z, Shogen K (1992) Synergism between a novel amphibian oocyte ribonuclease and lovastatin in inducing cytostatic and cytotoxic effects in human lung and pancreatic carcinoma cell lines. Br J Cancer 66: 304-310. [Crossref]
- Deptala A, Halicka HD, Ardelt B, Ardelt W, Mikulski SM et al. (1998) Potentiation of tumor necrosis factor induced apoptosis by onconase. Int J Oncol 13: 11-16. [Crossref]
- Vasandani VM, Castelli JC, Hott JS, Saxena S, Mikulski SM et al. (1999) Interferon enhances the activity of the anticancer ribonuclease, onconase. J Interferon Cytokine Res 19: 447-454. [Crossref]
- Rybak SM, Pearson JW, Fogler WE, Volker K, Spence SE et al. (1996) Enhancement of vincristine cytotoxicity in drug-resistant cells by simultaneous treatment with onconase, an antitumor ribonuclease. J Natl Cancer Inst 88: 747-753. [Crossref]
- Ita M, Halicka HD, Tanaka T, Kurose A, Ardelt B et al. (2008) Remarkable enhancement of cytotoxicity of onconase and cepharanthine when used in combination on various tumor cell lines. Cancer Biol Ther 7: 1104-1108. [Crossref]
- Majchrzak A, Witkowska M, Mędra A, Zwolińska M, Bogusz J et al. (2013) In vitro cytotoxicity of ranpirnase (onconase) in combination with components of R-CHOP regimen against diffuse large B cell lymphoma (DLBCL) cell line. Postepy Hig Med Dosw (Online) 67: 1166-1172. [Crossref]
- Fagagnini A, Pica A, Fasoli S, Montioli R, Donadelli M et al. (2017) Onconase dimerization through 3D domain swapping: structural investigations and increase in the apoptotic effect in cancer cells. Biochem J 474: 3767-3781. [Crossref]
- Mikulski S, Chun H, Mittelman A, Panella T, Puccio C et al. (1995) Relationship between response rate and median survival in patients with advanced nonsmall cell lung-cancer - comparison of onconase(r) with other anticancer agents. Int J Oncol 6: 889-897. [Crossref]
- Vogelzang NJ, Stadler WM (1999) Gemcitabine and other new chemotherapeutic agents for the treatment of metastatic bladder cancer. Urology 53: 243-250. [Crossref]
- Puccio C, Mittelman A, Chun H, et al. (1996) A new anticancer RNase (Onconase): clinical trial in patients (pts) with breast cancer (BC). Proc Am Soc Clin Oncol 242.
- Costanzi J, Sidransky D, Navon A, Goldsweig H (2005) Ribonucleases as a novel pro-apoptotic anticancer strategy: review of the pre-clinical and clinical data for ranpirnase. Cancer Invest 23: 643-650. [Crossref]
- Chun H, Costanzi J, Mittelman A, et al. (1995) Phase I/II trial of onconase (ONC) plus tamoxifen (TMX) in patients (pts) with advanced pancreatic carcinoma (APC). Proc Am Soc Clin Oncol 517.
- Pavlakis N, Vogelzang NJ (2006) Ranpirnase--an antitumour ribonuclease: its potential role in malignant mesothelioma. Expert Opin Biol Ther 6: 391-399. [Crossref]
- Merlino A, Mazzarella L, Carannante A, Fiore AD, Donato AD et al. (2005) The importance of dynamic effects on the enzyme activity: X-ray structure and molecular dynamics of onconase mutants. J Biol Chem 280: 17953-17960. [Crossref]
- Raineri A, Fasoli S, Campagnari R, Gotte G, Menegazzi M (2019) Onconase Restores Cytotoxicity in Dabrafenib-Resistant A375 Human Melanoma Cells and Affects Cell Migration, Invasion and Colony Formation Capability. Int J Mol Sci 20. [Crossref]
- Lee I, Kim DH, Sunar U, Magnitsky S, Shogen K (2007) The therapeutic mechanisms of ranpirnase-induced enhancement of radiation response on A549 human lung cancer. In Vivo 21: 721-728. [Crossref]
- Newton DL, Hansen HJ, Liu H, Ruby D, Iordanov MS et al. (2001) Specifically targeting the CD22 receptor of human B-cell lymphomas with RNA damaging agents. Crit Rev Oncol Hematol 39: 79-86. [Crossref]
- Newton DL, Hansen HJ, Mikulski SM, Goldenberg DM, Rybak SM (2001) Potent and specific antitumor effects of an anti-CD22-targeted cytotoxic ribonuclease: potential for the treatment of non-Hodgkin lymphoma. Blood 97: 528-535. [Crossref]
- Weber T, Mavratzas A, Kiesgen S, Haase S, Bötticher B et al. (2015) A Humanized Anti-CD22-Onconase Antibody-Drug Conjugate Mediates Highly Potent Destruction of Targeted Tumor Cells. J Immunol Res 2015: 561814. [Crossref]
- Erickson HA, Jund MD, Pennell CA (2006) Cytotoxicity of human RNase-based immunotoxins requires cytosolic access and resistance to ribonuclease inhibition. Protein Eng Des Sel 19: 37-45. [Crossref]
- Chang CH, Sapra P, Vanama SS, Hansen HJ, Horak ID et al. (2005) Effective therapy of human lymphoma xenografts with a novel recombinant ribonuclease/anti-CD74 humanized IgG4 antibody immunotoxin. Blood 106: 4308-4314. [Crossref]
- Newton D, Pearson J, Xue Y, Smith M, Fogler W et al. (1996) Anti-tumor ribonuclease, combined with or conjugated to monoclonal antibody MRK16, overcomes multidrug resistance to vincristine in vitro and in vivo. Int J Oncol 8: 1095-1104. [Crossref]
- Kiesgen S, Arndt MAE, Körber C, Arnold U, Weber T et al. (2015) An EGF receptor targeting Ranpirnase-diabody fusion protein mediates potent antitumour activity in vitro and in vivo. Cancer Lett 357: 364-373. [Crossref]
- Kiesgen S, Liebers N, Cremer M, Arnold U, Weber T et al. (2014) A fusogenic dengue virus-derived peptide enhances antitumor efficacy of an antibody-ribonuclease fusion protein targeting the EGF receptor. Protein Eng Des Sel 27: 331-337. [Crossref]
- Shen R, Ye D, Huang Q, Li J, Wang Q et al. (2018) An EGF receptor-targeting amphinase recombinant protein mediates anti-tumor activity in vitro and in vivo. Acta Biochim Biophys Sin (Shanghai) 50: 391-398. [Crossref]
- Chang CH, Gupta P, Michel R, Loo M, Wang Y et al. (2010) Ranpirnase (frog RNase) targeted with a humanized, internalizing, anti-Trop-2 antibody has potent cytotoxicity against diverse epithelial cancer cells. Mol Cancer Ther 9: 2276-2286. [Crossref]
- Liu D, Cardillo TM, Wang Y, Rossi EA, Goldenberg DM et al. (2014) Trop-2-targeting tetrakis-ranpirnase has potent antitumor activity against triple-negative breast cancer. Mol Cancer 13: 53. [Crossref]
- Sun M, Tang H, Gao Y, Dai X, Yuan Y et al. (2016) Constitutive expression and anticancer potency of a novel immunotoxin onconase-DV3. Oncol Rep 35: 1987-1994. [Crossref]
- Sun M, Sun L, Sun D, Zhang C, Li M (2018) Targeted delivery of immuno-RNase may improve cancer therapy. Cancer Cell Int 18: 58. [Crossref]
- Wang X, Guo Z (2015) Chlorotoxin-conjugated onconase as a potential anti-glioma drug. Oncol Lett 9: 1337-1342. [Crossref]
- Qi J, Ye X, Li L, Bai H, Xu C (2018) Improving the specific antitumor efficacy of ONC by fusion with N-terminal domain of transferrin. Biosci Biotechnol Biochem 82 : 1153-1158. [Crossref]
- Vasandani VM, Burris JA, Sung C (1999) Reversible nephrotoxicity of onconase and effect of lysine pH on renal onconase uptake. Cancer Chemother Pharmacol 44: 164-169. [Crossref]
- Tekade RK, Youngren Ortiz SR, Yang H, Haware R, Chougule MB (2014) Designing hybrid onconase nanocarriers for mesothelioma therapy: a Taguchi orthogonal array and multivariate component driven analysis. Mol Pharm 11: 3671-3683. [Crossref]
- Fernández Ulibarri I, Hammer K, Arndt MAE, Kaufmann JK, Dorer D et al. (2015) Genetic delivery of an immunoRNase by an oncolytic adenovirus enhances anticancer activity. Int J Cancer 136: 2228-2240. [Crossref]
- Liebers N, Holland Letz T, Welschof M, Høgset A, Jäger D et al. (2017) Highly efficient destruction of squamous carcinoma cells of the head and neck by photochemical internalization of Ranpirnase. J Exp Ther Oncol 12: 113-120. [Crossref]
