Potential Novel Ovarian Cancer Treatment Targeting Myeloid-Derived Suppressor Cells
A B S T R A C T
Diagnosis by biopsy is difficult in the ovary, since it is located deep in the abdomen. As a result, ovarian cancer is mostly found insidiously during exploratory laparotomy. Consequently, early diagnosis of ovarian cancer is often difficult. The likelihood of peritoneal dissemination increases with the progress of ovarian cancer. With further progression, ovarian cancer metastasizes to the omentum, retroperitoneal lymph nodes, large intestine, small intestine, diaphragm, spleen, and other organs. Ovarian cancer has been considered a tumor that has a favourable response to chemotherapy, but more effective treatments are still being explored. Tumors use their own immune escape mechanism to evade host immunity. The immune checkpoint (IC) mechanism, one of the immune escape mechanisms, is established by programmed cell death-1 (PD-1)/PD-ligand-1 (PD-L1) communication. It has been shown that inhibiting PD-1/PD-L1 communication in various malignancies produces antitumor effects. However, the antitumor effect of ICI monotherapy on ovarian cancer is limited in actual clinical practice. In this review, we describe a novel cancer immunotherapeutic agent that targets myeloid-derived suppressor cells (MDSCs).
Keywords
Ovarian cancer, anti-VEGF, anti-GM-CSF, MDSC, CTL
Ovarian Cancer Histopathology
Ovarian cancer has been thought to respond well to chemotherapy, but more effective treatments are still being explored [1]. Histologically, ovarian cancer is classified into serous adenocarcinoma, mucinous adenocarcinoma, endometrioid adenocarcinoma, clear cell adenocarcinoma, malignant Brenner tumor, and transitional cell carcinoma. Recent studies in clinical pathology and molecular biology have revealed that these subtypes differ in histogenesis and anticancer drug sensitivity. In Japan, the frequency of histological type occurrence is serous adenocarcinoma 39%, mucinous adenocarcinoma 12%, endometrioid adenocarcinoma 16%, and clear cell adenocarcinoma 23% (Table 1) [2].
Ovarian Cancer Requires Activation of a Pathway Other Than the IC
There are four subtypes of gene expression in high-grade serous ovarian cancer (Appendix 5): immunoreactive, mesenchymal, differentiated, and proliferative. The prognosis for immunoreactive cancer is good; however, the prognosis for mesenchymal cancer is poor [3]. In spite of this, gene expression analysis tests are not performed in routine clinical practice. High-grade serous ovarian cancer was classified by pathological examination into four subtypes: immune reactive (IR), mesenchymal transition (MT), papilloglandular (PG), solid and proliferative (SP) [4]. From the results of the research to date, it has been confirmed that among the four pathological subtypes of ovarian cancer, the prognosis is favourable in IR with significant lymphocyte infiltration in and around the tumor. However, MT, with a clearly confirmed stromal reaction and a labyrinth-like infiltration of tumor cells, has a poor prognosis. In other words, it seems that prognosis is influenced by the condition of the stroma rather than the degree of tumor differentiation.
Table 1: Classification, origin, and mechanism of epithelial ovarian cancer.
|
Tumor type |
Serous |
Endometrioid |
Clear cell |
Mucinous |
|
|
High-grade (Appendix 1) |
Low-grade |
||||
|
Incidence |
34% |
5% |
16% |
23% |
12% |
|
Tissue origin |
Fallopian tube epithelium |
Fallopian tube epithelium Ovarian surface epithelium |
Endometrial cells |
Endometrial cells |
Ovarian surface epithelium Brenner tumor (Appendix 4) |
|
Precancerous lesion |
STIC (Appendix 2) |
Borderline malignant tumor (Appendix 3) |
Atypical endometriosis |
Atypical endometriosis |
Borderline malignant tumor (Appendix 3) |
|
Molecular biological abnormalities |
p53 mutation BRCA1/2 mutation Chromosome instability |
KRAS mutation BRAF mutation (BRCA1/2 mutation) |
ERa high expression PI3KCA mutation CTNNB1 mutation ARID1A mutation BRCA1/2 mutation Microsatellite instability |
HNF-1b high expression PI3KCA mutation PTEN mutation ARID1A mutation |
KRAS mutation HER2 high expression |
|
Sensitivity to chemotherapy |
High |
Middle |
High |
Low |
Low |
|
HBOC (Appendix 5) |
+++ |
++ |
+ |
− |
− |
|
Type I/II |
Type II |
Type I |
|||
|
Incidence |
Europe, USA > Asia |
Europe, USA < Asia |
|||
STIC (Appendix 2): serous tubal intraepithelial carcinoma, Borderline malignant tumor (Appendix 3): Borderline ovarian tumors are abnormal cells that form in the tissue overlying the ovary. They are not cancerous and are generally treated surgically. Approximately 15 out of 100 ovarian tumors (15%) are borderline tumors. They are also described as atypical proliferative tumors and were previously known as tumors of low malignant potential. They are different from ovarian cancer because they do not grow into the supportive tissue of the ovary (the stroma). Their growth is gradual and more controlled manner than cancer cells. Borderline tumors generally affect women aged between 20 and 40 years. They are usually diagnosed at an early stage when abnormal cells are still within the ovary.
In other words, IR is a cancer that has a good immune response, and it is believed that “the cancer is cured when the immune system is activated.” In this light, Hamanishi et al. investigated the antitumor effect of the anti-PD-1 antibody nivolumab on serous ovarian cancer. A clinical trial was performed involving 20 patients with platinum-resistant ovarian cancer. In this trial, 2 patients showed a complete response and had no recurrence for >5 years. However, the effect was insufficient in 90% of cases [5]. Therefore, Hamanishi et al. suggested that ovarian cancer requires an immunotherapy strategy different from ICI. As a new immunotherapy, we considered a treatment that targets MDSC.
Suppression of Myeloid-Derived Suppressor Cells (MDSC) Generation and Migration by Anti-Vascular Endothelial Growth Factor (VEGF) Antibody
Normally, immature myeloid cells differentiate and mature into macrophages, granulocytes, and dendritic cells. However, due to inflammation such as in cancer or trauma, immature myeloid cells are induced into peripheral tissues to become myeloid-derived suppressor cells (MDSCs). MDSCs have a high immunosuppressive effect. Prognosis is poor in high MDSC ovarian cancer. Abiko et al. reported that VEGF promotes MDSC generation and migration and reduces cytotoxic T cells (CTL). Furthermore, anti-VEGF antibodies have been shown to suppress MDSC production and migration [6]. In addition, the administration of anti-Gr-1 antibodies to a mouse ovarian cancer model in which VEGF was highly expressed decreased intratumoral MDSC and increased CTL, producing an antitumor effect. Based on these results, Abiko et al. reported that “VEGF has an immunosuppressive action in addition to its angiogenic action.” When VEGF is expressed in ovarian cancer, MDSCs are generated and migrate, and CTLs are suppressed promoting tumor growth and progression. Furthermore, it has been shown that administration of an anti-VEGF antibody such as bevacizumab can suppress MDSC generation/migration.
Anti-GM-CSF Antibody Improves Hypoxia and Overcomes the Resistance to Anti-VEGF Antibody Treatment
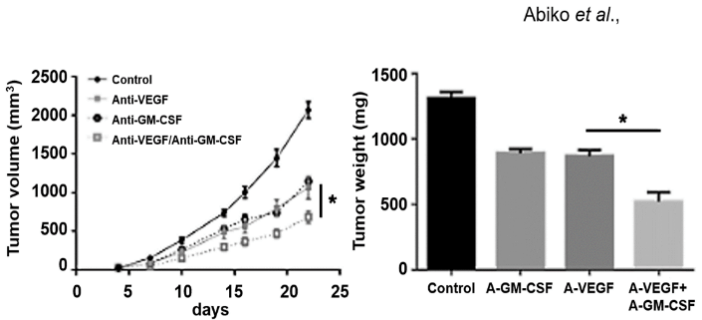
Conversely, the antitumor effect of the anti-VEGF antibody is gradually attenuated by the acquired resistance. Abiko et al. found that MDSCs increased and CTLs decreased in ovarian cancer that was resistant to anti-VEGF antibody therapy. In ovarian cancer resistant to anti-VEGF antibody therapy in mice, granulocyte macrophage colony-stimulating factor (GM-CSF), a cytokine that differentiates pluripotent hematopoietic stem cells into myeloid progenitor cells, was increased. In addition, GM-CSF seems to promote MDSC migration. Therefore, anti-VEGF antibody and the anti-GM-CSF antibody combination therapy enhances the antitumor effect (Figure 1). This combination therapy was shown to suppress infiltration of MDSC in ovarian cancer resistant to anti-VEGF antibody therapy (Figure 2) [7]. In clinical cases of bevacizumab resistance, the hypoxic markers HIF-1α, GM-CSF, and MDSC were increased.
Figure 1: Enhancement of the antitumor effect by the anti-VEGF antibody and the anti-GM-CSF antibody combination therapy.
In vivo treatment of human serous ovarian cancer cell line, OV2944-HM-1 subcutaneous tumors with anti-VEGF antibody, anti-GM-CSF, combination of anti-VEGF/anti-GM-CSF or normal IgG as a control (n = 5). Tested antibodies were administered 150 μg/mouse intraperitoneally twice a week from 4 days after tumor inoculation. Left panel: Xenografted OV2944-HM-1 tumors treated with normal IgG were collected at day 18, while Other xenografted OV2944-HM-1 tumors were collected at day 28, when the average diameter of tumors reached >10 mm. *P < 0.05. Right panel: Tumor weights were also measured on the day of tumor collection using digital scales. The modified figure is adapted from doi: 10.1038/s41416-019-0725-x.
Figure 2: Schematic representation of the antitumor effect by anti-VEGF antibody and anti-GM-CSF antibody combination therapy.
Vascular Endothelial Growth Factor (VEGF) promotes MDSC generation and migration and reduces cytotoxic T cells (CTL). Therefore, anti-VEGF antibody has been shown to suppress MDSC generation and migration. Granulocyte macrophage colony-stimulating factor (GM-CSF), a cytokine that differentiates pluripotent hematopoietic stem cells into myeloid progenitor cells, is increased in ovarian cancer resistant to anti-VEGF antibody therapy. In addition, GM-CSF seems to promote MDSC migration. Therefore, anti-VEGF antibody and anti-GM-CSF antibody combination therapy enhances the antitumor effect.
Ovarian cancer cells secrete GM-CSF and S100 calcium-binding protein A4 (S100A4) under hypoxic conditions, leading to a promotion in MDSC migration and a suppression of CTL migration into ovarian cancer cells [8]. These biological responses are considered to be the basic skeleton of the mechanism of resistance to angiogenesis inhibitors. Combination therapy with anti-GM-CSF antibody and anti-VEGF antibody is a potential treatment option for ovarian cancer resistant to anti-VEGF antibody therapy.
Conclusion
In the “The National Comprehensive Cancer Network Guidelines”, which are the global guidelines for cancer treatment, there is insufficient evidence for surgical treatment of recurrent ovarian cancer. Therefore, surgical treatment is not recommended as the gold standard. However, in cases where the interval between initial treatment and recurrence is a certain period (6 months) or more and complete resection of ovarian cancer is possible, surgical treatment may improve prognosis. In most cases, 6 courses of postoperative adjuvant chemotherapy (3-4 courses in NeoAdjuvant Chemotherapy (NAC) cases). TC therapy (paclitaxel and carboplatin) is given as adjuvant chemotherapy. Furthermore, TC therapy is often combined with bevacizumab in clinical stage III and above. After chemotherapy, germline variants in the breast cancer 1 (BRCA) gene are tested. If the test results are positive, the poly (ADP-ribose) polymerase (PARP) inhibitor olaparib is taken orally for up to 2 years as maintenance therapy. If the test is negative or if the BRCA test is not done, every 3 weeks, bevacizumab therapy is given until 15 doses. Chemotherapy is relatively effective for recurrent ovarian cancer. Therefore, chemotherapy is the first-line treatment for recurrent disease. However, chemotherapy is not always the only recommended treatment for recurrent ovarian cancer. It is also important to consider a combination of chemotherapy and surgical treatment.
Appendices
I Serous Carcinoma
Serous carcinoma is divided into high-grade and low-grade, the former accounts for the overwhelming majority. High-grade serous carcinoma is the most common histological type of ovarian cancer, with its clinicopathological features presenting many advanced forms of ovarian cancer. High-grade serous carcinoma reflects the classic clinical picture of ovarian cancer that is highly sensitive to anticancer drugs. More than 90% of cases have TP53 mutations, and about half are thought to originate from the fallopian tube [9, 10]. On the other hand, low-grade serous carcinoma is thought to occur in the background of serous borderline malignant tumors, with few cases of advanced disease; however, proliferative activity and anticancer drug sensitivity are low. Low-grade serous carcinoma has been reported to have many cases with KRAS and BRAF somatic mutations [9, 10]. Although the estrogen receptor is positive, the effectiveness of hormonal therapy has not been verified in clinical studies [9, 10].
II STIC
Serous tubal intraepithelial carcinoma. Frequent detection of occult STIC among pathogenic BRCA1 or BRCA2 mutation carriers during risk-reducing surgery prompted the hypothesis that many adnexal high-grade serous carcinomas arise from the fallopian tube, rather than the ovary, as previously supposed [11-13].
III Borderline Malignant Tumor
Borderline ovarian tumors are abnormal cells that form in the tissue overlying the ovary. They are not cancer and are usually treated surgically. Around 15 out of 100 ovarian tumors (15%) are borderline tumors. They are also described as atypical proliferative tumors and used to be known as tumors of low malignant potential. They are different from ovarian cancer because they do not grow into the supportive tissue of the ovary (the stroma). They tend to grow slowly and in a more controlled manner than cancer cells. Borderline tumors usually affect women aged between 20 and 40 years. They are usually diagnosed at an early stage, when the abnormal cells are still within the ovary.
IV Brenner Tumor
Brenner tumors are an uncommon subtype of the surface epithelial-stromal tumor group of ovarian neoplasms. The majority are benign, but some can be malignant [11]. They are frequently found insidiously on pelvic examination or at laparotomy [12]. Brenner tumors very rarely occur in other locations, including the testes [13].
V HBOC
Hereditary breast and ovarian cancer syndrome (HBOC syndrome) is a susceptibility syndrome for cancers such as breast cancer and ovarian cancer caused by germline mutations in BRCA. HBOC is an autosomal dominant form of inheritance [14]. Hereditary tumors associated with susceptibility due to single gene mutations account for 7-10% of breast cancers [15]. In the United States, BRCA mutations are found in about 5% of women with breast cancer aged 35 to 64 years [16]. On the other hand, it has been reported that 15% of ovarian cancer cases overseas have BRCA mutations [17].
Conflicts of Interest
The authors declare no potential conflicts of interest. The funders had no role in study design, data collection, and analysis; decision to publish; or preparation of the manuscript.
Author Contributions
K.A., T.H. and K.Y. performed most of the experiments and coordinated the project; K.A., T.H. and K.Y. conceived the study and wrote the manuscript. M.M. carefully reviewed this manuscript and reviewed the medical science. I.K. provided information on clinical medicine and supervised the entire study.
Acknowledgments
We thank Professor Richard A. Young (Whitehead Institute for Biomedical Research, Massachusetts Institute of Technology, Cambridge, MA) for his research assistance. This study was supported in part by grants from the Japan Ministry of Education, Culture, Science and Technology (No. 24592510, No. 15K1079, and No. 19K09840), the Foundation of Osaka Cancer Research, The Ichiro Kanehara Foundation for the Promotion of Medical Science and Medical Care, the Foundation for Promotion of Cancer Research, the Kanzawa Medical Research Foundation, The Shinshu Medical Foundation, and the Takeda Foundation for Medical Science.
Article Info
Article Type
Mini ReviewPublication history
Received: Mon 23, Nov 2020Accepted: Wed 23, Dec 2020
Published: Wed 30, Dec 2020
Copyright
© 2023 Takuma Hayashi. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.CROGR.2020.03.03
Author Info
Kaoru Abiko Takuma Hayashi Ken Yamaguchi Masaki Mandai Ikuo Konishi
Corresponding Author
Takuma HayashiNational Hospital Organization, Kyoto Medical Center, Kyoto, Japan
Figures & Tables
Table 1: Classification, origin, and mechanism of epithelial ovarian cancer.
|
Tumor type |
Serous |
Endometrioid |
Clear cell |
Mucinous |
|
|
High-grade (Appendix 1) |
Low-grade |
||||
|
Incidence |
34% |
5% |
16% |
23% |
12% |
|
Tissue origin |
Fallopian tube epithelium |
Fallopian tube epithelium Ovarian surface epithelium |
Endometrial cells |
Endometrial cells |
Ovarian surface epithelium Brenner tumor (Appendix 4) |
|
Precancerous lesion |
STIC (Appendix 2) |
Borderline malignant tumor (Appendix 3) |
Atypical endometriosis |
Atypical endometriosis |
Borderline malignant tumor (Appendix 3) |
|
Molecular biological abnormalities |
p53 mutation BRCA1/2 mutation Chromosome instability |
KRAS mutation BRAF mutation (BRCA1/2 mutation) |
ERa high expression PI3KCA mutation CTNNB1 mutation ARID1A mutation BRCA1/2 mutation Microsatellite instability |
HNF-1b high expression PI3KCA mutation PTEN mutation ARID1A mutation |
KRAS mutation HER2 high expression |
|
Sensitivity to chemotherapy |
High |
Middle |
High |
Low |
Low |
|
HBOC (Appendix 5) |
+++ |
++ |
+ |
− |
− |
|
Type I/II |
Type II |
Type I |
|||
|
Incidence |
Europe, USA > Asia |
Europe, USA < Asia |
|||
STIC (Appendix 2): serous tubal intraepithelial carcinoma, Borderline malignant tumor (Appendix 3): Borderline ovarian tumors are abnormal cells that form in the tissue overlying the ovary. They are not cancerous and are generally treated surgically. Approximately 15 out of 100 ovarian tumors (15%) are borderline tumors. They are also described as atypical proliferative tumors and were previously known as tumors of low malignant potential. They are different from ovarian cancer because they do not grow into the supportive tissue of the ovary (the stroma). Their growth is gradual and more controlled manner than cancer cells. Borderline tumors generally affect women aged between 20 and 40 years. They are usually diagnosed at an early stage when abnormal cells are still within the ovary.

References
- Gurung A, Hung T, Morin J, Gilks CB (2013) Molecular abnormalities in ovarian carcinoma:clinical, morphological and therapeutic correlates. Histopathology 62: 59-70. [Crossref]
- Heintz AP, Odicino F, Maisonneuve P, Quinn MA, Benedet JL et al. (2006) Carcinoma of the ovary. FIGO 26th Annual Report on the Results of Treatment in Gynecological Cancer. Int J Gynaecol Obstet 95: S161-S192. [Crossref]
- Verhaak RG, Tamayo P, Yang JY, Hubbard D, Zhang H et al. (2013) Cancer Genome Atlas Research Network. Prognostically Relevant Gene Signatures of High-Grade Serous Ovarian Carcinoma. J Clin Invest 123: 517-525. [Crossref]
- Murakami R, Matsumura N, Mandai M, Yoshihara K, Tanabe H et al. (2016) Establishment of a Novel Histopathological Classification of High-Grade Serous Ovarian Carcinoma Correlated With Prognostically Distinct Gene Expression Subtypes. Am J Pathol 186: 1103-1113. [Crossref]
- Hamanishi J, Mandai M, Ikeda T, Minami M, Kawaguchi A et al. (2015) Safety and Antitumor Activity of Anti-PD-1 Antibody, Nivolumab, in Patients With Platinum-Resistant Ovarian Cancer. J Clin Oncol 33: 4015-4022. [Crossref]
- Horikawa N, Abiko K, Matsumura N, Hamanishi J, Baba T et al. (2017) Expression of Vascular Endothelial Growth Factor in Ovarian Cancer Inhibits Tumor Immunity Through the Accumulation of Myeloid-Derived Suppressor Cells. Clin Cancer Res 23: 587-599. [Crossref]
- Horikawa N, Abiko K, Matsumura N, Baba T, Hamanishi J et al. (2020) Anti-VEGF Therapy Resistance in Ovarian Cancer Is Caused by GM-CSF-induced Myeloid-Derived Suppressor Cell Recruitment. Br J Cancer 122: 778-788. [Crossref]
- Hayahsi T, Sano K, Aburatani H, Yaegashi N, Konishi I (2020) Initialization of epithelial cells by tumor cells in a metastatic microenvironment. Oncogene 39: 2638-2640. [Crossref]
- Taha AAA, Koshiyama M, Matsumura N, Abiko K, Yamaguchi K et al. (2018) The effect of the type of dietary protein on the development of ovarian cancer. Oncotarget 9: 23987-23999. [Crossref]
- Gurung A, Hung T, Morin J, Gilks CB (2013) Molecular abnormalities in ovarian carcinoma: clinical, morphological and therapeutic correlates. Histopathology 62: 59-70. [Crossref]
- Piek JM, van Diest PJ, Zweemer RP, Jansen JW, Poort Keesom RJ et al. (2001) Dysplastic changes in prophylactically removed Fallopian tubes of women predisposed to developing ovarian cancer. J Pathol 195: 451-456. [Crossref]
- Crum CP, Drapkin R, Kindelberger D, Medeiros F, Miron A et al. (2007) Lessons from BRCA: the tubal fimbria emerges as an origin for pelvic serous cancer. Clin Med Res 5: 35-44. [Crossref]
- Kurman RJ, Shih IM (2016) The dualistic model of ovarian carcinogenesis: revisited, revised, and expanded. Am J Pathol 186: 733-747. [Crossref]
- Petrucelli N, Daly MB, Pal T (1998) RCA1- and BRCA2-Associated Hereditary Breast and Ovarian Cancer. GENEReviews. [Crossref]
- Kauff ND (2007) Management of BRCA-Negative Hereditary Breast Cancer Families. Hereditary Breast Cancer. (Claudine Isaacs C, Rebbeck TR) CRC Press 311-318.
- Malone KE, Daling JR, Doody DR, Hsu L, Bernstein L et al. (2006) Prevalence and predictors of BRCA1 and BRCA2 mutations in a population-based study of breast cancer in white and black American women ages 35 to 64 years. Cancer Res. 66: 8297-8308. [Crossref]
- Norquist BM, Harrell MI, Brady MF, Walsh T, Lee MK et al. (2016) Inherited Mutations in Women With Ovarian Carcinoma. JAMA Oncol 2: 482-490. [Crossref]
