Induction Chemotherapy Docetaxel, Cisplatin, 5-Fluorouracil Followed by Concurrent Chemoradiotherapy in Stage IVA Nasopharyngeal Carcinoma
A B S T R A C T
Objectives: Two phase III studies using docetaxel, cisplatin and 5-fluorouracil (TPF) followed by concurrent chemoradiotherapy (CCRT) have reported survival benefit compared to CCRT. However, TPF was used at 80% of the conventional dose in the Chinese study due to the concern of tolerability. We aim to explore the tolerability of TPF using standard dose in Chinese patients with stage IVA (AJCC 8th edition) nasopharyngeal carcinoma and report on its preliminary efficacy.
Methods: Consecutive patients treated with induction chemotherapy (IC) TPF followed by CCRT between 2017 and 2020 were reviewed. TPF regimen consisted of three cycles of Docetaxel 75 mg/m2 D1; Cisplatin 75 mg/m2 D1; 5-fluorouracil 750 mg/m2 D1-5 every 3 weeks; with G-CSF D7-11. Radiotherapy consisted of 70 Gy in 35 fractions with concomitant cisplatin 100 mg/m2 every 3 weeks.
Results: 28 patients were identified. 92.8% patients completed three cycles of IC. 79% patients received concomitant cisplatin of ≥ 200 mg/m2. The most common grade 3 or 4 acute toxicity was diarrhea (25%). Grade 3 or 4 neutropenia occurred in 10.7% of patients, of which only 3.6% resulted in febrile neutropenia. Median follow-up was 2 years. The 2-year overall survival, locoregional failure-free survival and distant failure-free survival were 100%, 96.4% and 91.3% respectively.
Conclusion: IC using standard dose TPF was well tolerated in Chinese patients with manageable toxicities and allowed adequate delivery of subsequent concurrent cisplatin. Survival outcomes were encouraging and unnecessary dose reduction of TPF should be avoided.
Keywords
Nasopharyngeal carcinoma, induction chemotherapy, concurrent chemoradiotherapy, toxicity
Introduction
Approximately 129,079 new cases of nasopharyngeal carcinoma (NPC) are diagnosed each year worldwide. It has a unique endemic distribution affecting mainly Southeast Asia, in particular the southern regions of China [1]. Due to the radiosensitive behaviour of NPC, radiotherapy (RT) is the cornerstone treatment. A meta-analysis on the role of chemotherapy in NPC (MAC-NPC) reported in 2006 confirmed an absolute survival benefit of 6% and event free survival benefit of 10% at 5 years respectively, with the addition of chemotherapy [2]. The updated report of MAC-NPC in 2015 echoed the advantage of adding chemotherapy. Despite the absence of significant overall survival benefit (hazard ratio (HR) 0.96, 95% confidence interval (CI) 0.8-1.16) with the use of induction chemotherapy (IC) alone, distant failure-free survival (DFFS) was significantly improved (HR 0.67, 95% CI 0.59-0.75) [3].
Up to one in four patients with NPC present with non-metastatic stage IV disease, for which standard treatment produces distinct failure patterns. Patients with T4 disease carry high local and distant failure risk, with 5-year locoregional failure-free survival (LRFFS) and DFFS of 73%. Patients with N3 disease are at high risk of distant metastasis, with 5-year DFFS of 66%. The 5-year overall survival (OS) is only 65% after routine adoption of concurrent chemoradiotherapy (CCRT) with intensity modulated radiotherapy (IMRT) and adjunctive chemotherapy (i.e. induction or adjuvant chemotherapy (AC)) [4]. New treatment directions to tackle these failure patterns are warranted [5, 6].
The triplet combination consisting of docetaxel, cisplatin and 5-fluorouracil (TPF) is a well-established IC regimen in locoregionally advanced head and neck cancer. TAX 323 and TAX 324 were landmark studies that demonstrated superior progression free survival (PFS) and OS using TPF compared to the PF regimen [7-9]. Two randomized phase III studies of IC TPF on locoregionally advanced NPC have also been reported recently. Albeit with two different dose regimens, both studies reported statistically significant OS and PFS gain with the addition of induction TPF over CCRT alone. Of note, the GORTEC study utilized the conventional dose regimen (Docetaxel 75 mg/m2 D1; cisplatin 75 mg/m2 D1; 5FU 750 mg/m2 D1-5), while in the Chinese study by Sun et al. (“Sun study”), the starting dose was only 80% of the conventional TPF regimen (Docetaxel 60 mg/m2 D1; cisplatin 60 mg/m2 D1; 5FU 600 mg/m2 D1-5) [10, 11]. This dose-reduced regimen was based on two phase 1 studies done at Sun Yat-sen University Cancer Center [12, 13]. Zhang et al. found the maximum tolerated dose of 5FU was 550 mg/m2 D1-5 when combined with docetaxel and cisplatin at 60 mg/m2 each [12]. Guo et al. investigated the tolerance of Chinese patients to a similar regimen, except 5FU was escalated to 600 mg/m2 [13]. Good compliance of 88% and fewer G3/4 toxicities were reported compared to other studies.
Since the publications of these two phase III studies in 2017, IC TPF was adopted in our institution for the treatment of AJCC/UICC stage IVA (T4 or N3) NPC patients under the age of 60 and without significant medical co-morbidities. We employed the conventional dose of TPF as reported in the TAX 323 and GORTEC study [7, 10]. Our objectives are to study the tolerability of using this conventional dose TPF in Chinese patients and its preliminary efficacy in the treatment of locoregionally advanced NPC.
Patients and Methods
Patients treated with TPF followed by CCRT from Jan 2017 to Jan 2020 were retrospectively reviewed. Total 28 consecutive patients were identified. All patients had stage IVA (T4 or N3; AJCC 8th edition) disease. Pre-treatment evaluation included the following: detailed history, flexible nasopharyngoscopy with biopsy, magnetic resonance imaging (MRI), positron-emission tomography (PET) and plasma Epstein-Barr virus (EBV) DNA.
All patients except one were under the age of 60 and the median age was 48 years (range 33-61). There was male predominance, with male-to-female ratio of 3:1. 26 (92.9%) patients had undifferentiated carcinoma. Two patients had keratinized, squamous and non-keratinized squamous cell carcinoma respectively. Almost all patients had performance status (PS) of 0 or 1, and only one patient with PS 2 was included. 60.7% of patients had T4 disease, while 42.9% of patients had N3 disease. Median plasma EBV DNA was 1096 copies (range 0-28750). Baseline patient demographics are summarized in (Table 1).
Table 1: Patient demographics.
|
Baseline characteristics |
No. of patients (%), N=28 |
|
Age Mean (range) |
49 y (33-61) |
|
Sex Male/Female |
21 (75%) / 7 (25%) |
|
ECOG 0 1 2 |
10 (35.7%) 17 (60.7%) 1 (3.57%) |
|
Histology Keratinized, squamous Non-keratinized squamous Undifferentiated |
1 (3.57%) 1 (3.57%) 26 (92.9%) |
|
T category 1 2 3 4 |
2 (7.14%) 2 (7.14%) 7 (25%) 17 (60.7%) |
|
N category 0 1 2 3 |
3 (10.7%) 9 (32.1%) 4 (14.3%) 12 (42.9%) |
|
Radiation dose 70 Gy 30 Gy (palliative dose) |
27 (96.4%) 1 (3.57%) |
|
Baseline EBV DNA Mean (range) |
3683 copies/ml (0-28750) |
|
Post radical RT |
No of patients (%), N=27 |
|
6 weeks post-treatment NP biopsy No residue Residue |
26 (96.2%) 1 (3.7%) |
|
8 weeks post-treatment EBV DNA 0 copy >0 copy Missing data |
24 (88.9%) 3 (11.1%) 0 |
|
26 weeks post-treatment EBV DNA 0 copy >0 copy Missing data |
23 (85.2%) 2 (7.4%) 2 (7.4%) |
|
16 weeks post-treatment MRI or PET Complete remission Partial response Missing data |
26 (96.2%) 1 (3.7%) 0 |
|
Median follow up Median (range) |
2 years (8.6-39.5 months) |
Chemotherapy
The IC TPF regimen consisted of three cycles of Docetaxel 75 mg/m2 D1, cisplatin 75 mg/m2 D1 and 5-fluorouracil 750 mg/m2 as continuous infusion D1-5 every 3 weeks. All patients received prophylactic granulocyte colony stimulating factor (G-CSF) support D7-11 during IC. For CCRT, cisplatin 100 mg/m2 was administered on D1, D22 and D43. During the induction and concurrent phases, chemotherapy was withheld until nadir values were ≥1.5 (x 109/L) for neutrophils and ≥100 (x 109/L) for platelets. Dose modifications were based on nadir blood counts and toxicities of the proceeding cycles.
Radiotherapy
All patients were treated with IMRT. RT consisted of 70 Gy in 35 fractions over 7 weeks. RT protocols are available in previous publications [14]. CCRT was commenced within 21 days after the first day of the last cycle of IC.
Assessment and Follow Up Policy
Tumors were assessed with the use of flexible nasopharyngoscopy and biopsy at 8 weeks, and MRI at 16 weeks after completion of CCRT. MRI responses were graded according to Response Evaluation Criteria in Solid Tumors (RECIST). Plasma EBV DNA was checked at 8 weeks and 26 weeks after completion of CCRT. In the first 3 years of follow-up, all patients underwent assessment with detailed history, physical examination, flexible nasopharyngoscopy every 3 months and then every 6 months thereafter until death. A follow up MRI was performed at 2 years, while additional EBV DNA and imaging were also done based on the clinician’s judgment. The median follow-up of this study was 2 years (range: 8.6-39.5 months).
Statistical Analysis
The primary objective of this study was to report on the acute toxicities from IC and CCRT. Late toxicities were also recorded. All toxicities were reviewed from medical records and graded according to the Common Terminology Criteria for Adverse Events (version 5.0). Survival outcomes were analysed. OS, LRFFS and DFFS were defined as the time interval between biopsy diagnosis and death, locoregional failure and distant metastasis respectively. Data was censored at the date of last follow-up. Time to event data was described using Kaplan-Meier curves. All analyses were performed using SPSS (version 26) and R (version 3.6.1).
Results
I Compliance and Tolerance of Induction TPF
26 patients (92.8%) completed 3 cycles of TPF. All patients received full dose TPF during the 1st cycle. 6 patients (21.4%) required dose reduction to 80% TPF dose at cycle 2. Among these 6 patients, one patient received re-escalation of docetaxel and cisplatin to full dose in the last cycle. Therefore, in the 3rd cycle only 17.9% patients had 80% docetaxel and 5FU dose, while 21.4% patients had 80% cisplatin. 96.4 % patients received the scheduled TPF without delay. Only one patient had a delay of 0.5 week at cycle 2 due to G3 diarrhea. Among the 2 patients who did not proceed with the last cycle TPF, one patient developed ST-elevation myocardial infarction (STEMI). While the other patient had received one cycle of IC PF in another hospital before commencing TPF in our unit, thus the last cycle was not given. Table 2 summarizes the compliance to TPF.
Table 2: Compliance and tolerance to conventional dose TPF, N=28 patients.
|
Dose/cycle |
1st TPF |
2nd cycle Docetaxel |
2nd cycle Cisplatin |
2nd cycle 5 fluorouracil |
3rd cycle Docetaxel |
3rd cycle Cisplatin |
3rd cycle 5-fluorouracil |
|
100% |
28 (100%) |
22 (78.6%) |
22 (78.6%) |
22 (78.6%) |
21 (75%) |
20 (71.4%) |
20 (71.4%) |
|
80% |
0 |
6 (21.4%) |
6 (21.4%) |
6 (21.4%) |
5 (17.9%) |
6 (21.4%) |
5 (17.9%) |
|
0% |
0 |
0 |
0 |
0 |
2 (7.14%)* |
2 (7.14%)* |
2 (7.14%)* |
*One patient received 1st cycle chemotherapy (consisting of cisplatin and 5 fluorouracil) at another institution; another developed ST-elevation myocardial infarction.
II Acute Toxicities During Induction TPF
G3/4 toxicities were not common. The most common G3 toxicity was diarrhea (25%). 2 patients (7.1%) and 1 patient (3.6%) had G4 and G3 neutropenia respectively. Only one patient (3.6%) developed febrile neutropenia. G3 anemia was found in 7.1% of patients. G3/4 hyponatremia occurred in 10.7% of patients. 7.1% of patients had G3 vomiting. One patient developed STEMI after 2 cycles of TPF and was further complicated with gastrointestinal bleeding. Due to unsatisfactory general condition, this patient received palliative RT with 30 Gy in 10 fractions over 2 weeks instead of the preplanned 70 Gy radical RT. This patient is currently alive on best supportive care with no evidence of disease on clinical assessment. There was no treatment related death. Table 3 summarizes the acute toxicities during TPF.
III Compliance and Tolerance to CCRT
14.3% of patients received 3 cycles of cisplatin during CCRT. Most patients (82.1%) received 2 cycles of concurrent cisplatin. One patient did not receive CCRT due to STEMI, as mentioned previously. Compliance according to dose intensity was further analysed. Patients receiving 300 mg/m2, 280 mg/m2, 200 mg/m2, 180 mg/m2, 160 mg/m2 were 7.14%, 7.14%, 64.3%, 7.1% and 10.7% respectively. Overall, 79% patients received ≥ 200 mg/m2. 27 patients (96.4%) completed radical RT 70 Gy in 35 fractions over 7 weeks without any delay. Due to STEMI, one patient received palliative RT with 30 Gy in 10 fractions over 2 weeks instead.
IV Acute Toxicities During CCRT
G3/4 toxicities were not frequent. The most common G3/4 toxicity was myelosuppression. 25.9% and 3.7% of patients had G3 and G4 neutropenia respectively. Among them, only one patient (3.7%) developed febrile neutropenia. G3 anemia occurred in 33.7% of patients. Other G3 cisplatin-related acute toxicities included vomiting (11.1%), hyponatremia (7.4%) and non-neutropenic infection (7.4%). G3 mucositis and skin reaction were seen in 48.1% and 11.1 % patients respectively. 2 patients required nasogastric tube feeding due to significant mucositis. There was no treatment related death. Table 3 summarizes the acute toxicities during CCRT.
Table 3: Acute toxicities during TPF and CCRT.
|
|
TPF (n=28) |
CCRT (n=27) |
||||||
|
Toxicity/Grade |
1 |
2 |
3 |
4 |
1 |
2 |
3 |
4 |
|
Vomiting |
6 (21.4%) |
2 (7.14%) |
2 (7.14%) |
0 |
3 (11.1%) |
2 (7.41%) |
3 (11.1%) |
0 |
|
Diarrhea |
6 (21.4%) |
2 (7.14%) |
7 (25%) |
0 |
0 |
0 |
0 |
0 |
|
HFS |
1 (3.57%) |
0 |
0 |
0 |
0 |
0 |
1 (3.70%) |
0 |
|
Neutropenia |
1 (3.57%) |
0 |
1 (3.57%) |
2 (7.14%) |
4 (14.8%) |
5 (18.5%) |
7 (25.9%) |
1 (3.70%) |
|
Neutropenic fever |
1 (3.57%) |
0 |
1 (3.57%) |
0 |
2 (7.40%) |
0 |
1 (3.70%) |
0 |
|
Non-neutropenic infection |
0 |
2 (7.14%) |
1 (3.57%) |
0 |
0 |
0 |
2 (7.40%) |
0 |
|
Anaemia |
17 (60.7%) |
6 (21.4%) |
2 (7.14%) |
0 |
10 (37.0%) |
8 (29.6%) |
9 (33.3%) |
0 |
|
Thrombocytopenia |
7 (25%) |
0 |
0 |
0 |
11 (40.7%) |
5 (18.5%) |
0 |
0 |
|
Hearing loss |
0 |
0 |
0 |
0 |
1 (3.70%) |
0 |
0 |
0 |
|
Mucositis |
1 (3.57%) |
0 |
0 |
0 |
2 (7.41%) |
12 (44.4%) |
13 (48.1%) |
0 |
|
Neuropathy |
2 (7.14%) |
0 |
0 |
0 |
0 |
1 (3.70%) |
0 |
0 |
|
Impaired renal function |
9 (32.1%) |
0 |
0 |
0 |
14 (51.9%) |
1 (3.70%) |
0 |
0 |
|
Increased alanine transaminase |
7 (25%) |
1 (3.57%) |
0 |
0 |
2 (7.40%) |
1 (3.70%) |
0 |
0 |
|
Increased bilirubin |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
Hypokalaemia |
14 (50%) |
3 (10.7%) |
0 |
0 |
6 (22.2%) |
4 (14.8%) |
0 |
0 |
|
Hyponatremia |
13 (46.4%) |
2 (7.14%) |
2 (7.14%) |
1 (3.57%) |
13 (48.1%) |
0 |
2 (7.41%) |
0 |
|
Skin Reaction |
|
|
|
|
11 (40.7%) |
12 (44.4%) |
3 (11.1%) |
0 |
|
Tube Feeding |
|
|
|
|
3 (11.1%) |
0 |
0 |
0 |
|
Others* |
0 |
0 |
0 |
1 (3.57%)* |
|
|
|
|
CCRT: Concurrent Chemoradiotherapy; HFS: Hand Foot Syndrome; *ST- elevation myocardial infarction. *estimated 3-year OS; §estimated 3-year PFS.
V Late Toxicities
There were no G3/4 late toxicity. 4 patients (14.4%) and 1 patient (3.6%) had G1 and G2 neuropathy. 3 patients (10.7%) and 1 patient (3.6%) had G1 and G2 nephropathy respectively.
VI Treatment Response Assessment
Among the 27 patients who received radical CCRT, only one patient (3.7%) had persistent local disease confirmed by nasopharyngeal biopsy at 8 weeks upon completion of CCRT. Further stereotactic boost of 20 Gy in 8 fractions was given to this patient, which resulted in disease remission. 96.2% of patients had complete response on week 16 post-CCRT MRI. Only one patient (3.7%) had partial response, who was later confirmed with both local and distant relapse. 24 patients (88.9%) had undetectable plasma EBV DNA at 8 weeks post-CCRT. 3 patients had detectable EBV DNA (53 copies, 15 copies and 12 copies respectively). Among them, the patient with 53 copies subsequently developed distant failure. At 26 weeks post-CCRT, 23 patients (85.1%) had undetectable EBV DNA. 2 patients had detectable EBV DNA (1510 copies and 25 copies), and both were confirmed with distant metastasis. There were 2 patients with missing data.
VII Outcomes
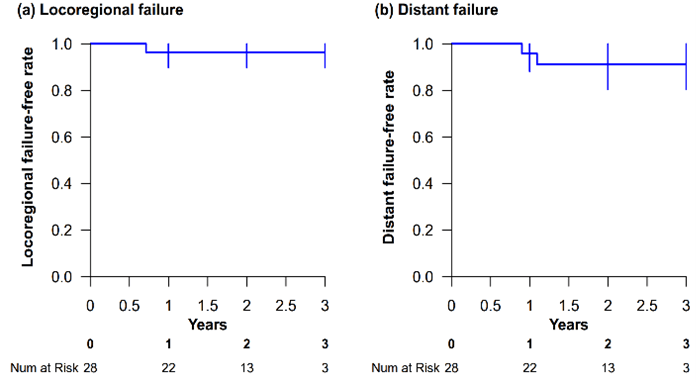
There was no patient death among the 28 patients and 2-year OS was 100%. For the 27 patients who completed radical treatment, local failure was found in 1 patient only while distant metastasis was reported in 2 patients. Overall, the 2-year LRFFS was 96.4%; the 2-year DFFS was 91.3%. Figures 1a & 1b present the survival curves.
Figure 1: a) Locoregional failure-free survival and b) distant failure-free survival.
Discussion
The Intergroup-0099 study was the landmark study to establish the current standard of care for NPC [15]. However, treatment outcomes for stage IVA (T4 or N3) disease are still unsatisfactory. 5-year OS was approximately 65%, compared with 80% for stage III disease [4]. Thus, new treatment approach is required for this high-risk group. Due to significant toxicities resulting from CCRT, compliance to AC is generally poor. Switching from AC to IC can be a solution. There are several additional advantages of using IC. Firstly, it can eradicate micrometastases. Secondly, IC can potentially downsize the tumor and facilitate RT delivery by increasing the likelihood of encompassing all viable tumor cells in the high dose irradiation field.
The role and optimal timing of chemotherapy for the treatment of locoregionally advanced NPC has always been a popular topic of debate. Emerging evidence comparing IC followed by CCRT with CCRT have been reported [10, 11, 16-22]. Most of these trials used a combination of chemotherapy such as taxane, platinum, 5FU, gemcitabine and anthracycline. Among them, IC using docetaxel seems to provide more consistent benefit when compared with gemcitabine-based IC [10, 16, 18, 21, 22]. The individual patient data (IPD) network meta-analysis by MAC-NPC published in 2017 provided further evidence and insight on these aspects [23]. Recently, the MAC-NPC collaborative group presented an updated IDP network meta-analysis in ASCO 2020 with the addition of 8 more trials [24]. Overall, 28 trials and 8,214 patients were included in this update. The median follow-up was 7.2 years. Apart from addressing the optimal treatment sequencing, a planned subgroup analysis on taxane and non-taxane IC was conducted. IC taxane-CCRT attained the highest probability of benefit in OS with HR 0.75 (95% CI 0.59-0.96), followed by IC non-taxane-CCRT HR 0.81 (95% CI 0.69-0.95), and CCRT-AC HR 0.88 (95% CI 0.75-1.04), when compared to CCRT alone. When the two types of IC (taxane and non-taxane) were merged, IC-CCRT still ranked the first, followed by CCRT-AC with a HR of 0.91 (95% CI 0.76-1.07) for IC-CCRT versus CCRT-AC.
Moreover, IC taxane also attained the highest probability of benefit in distant control with HR 0.62 (95% CI 0.45-0.84), followed by IC non-taxane CCRT HR 0.67 (95% CI 0.55-0.82) and IC-CCRT HR 0.77 (95% CI 0.59-1.01), compared to CCRT alone. This recent evidence has shed light on the optimal treatment sequence for locoregionally advanced NPC, suggesting that IC followed by CCRT may be the best sequence, while taxane-based IC may be the most potent treatment regimen. The emerging role of IC is also recognized by international guidelines. The National Comprehensive Caner network guidelines 2020 version 2.0 recommends both CCRT with AC and IC followed by CCRT as category 2A; while European Society of Medical Oncology guidelines recommends IC followed by CCRT for patients with tumor in close proximity to critical anatomical structures as category II, B [25, 26].
Taxanes are microtubule-stabilizing drugs that have been used extensively in many solid tumors. TPF is a well-established effective IC regimen used in locoregionally advanced head and neck cancer. The TAX 323 study was the first study to demonstrate a median OS improvement of 4.3 months with the addition of docetaxel to PF as induction followed by RT alone [7]. TAX 324 and its long-term update, which randomized patients to TPF vs PF, followed by concurrent carboplatin with RT also demonstrated a significant OS benefit with HR 0.74 (95% CI 0.58-0.94) [8, 9]. The GORTEC study with similar study design, echoed the above findings [27]. Of note, the TPF dose and schedule in these studies were largely comparable. Such regimen is currently recognized as the conventional standard. Details of TPF dose and schedule in various Phase III studies in head and neck cancer are summarized in (Supplementary Table 1).
With the success of IC TPF demonstrated in locoregionally advanced head and neck cancer, two Phase III studies utilizing TPF as IC in NPC patients were recently published. Both reported significant improvement in OS and PFS. However, due to concerns related to potential acute toxicities of TPF, especially in the Chinese population, the starting dose of TPF was only 80% of the conventional dose in the study by Sun et al. Despite dose reduction, the 3-year OS and 3-year PFS attained were 92% and 80% respectively in the TPF arm [11]. On the other hand, in the GORTEC study by Frikha et al., full dose of TPF was used. Comparable 3-year OS of 86% and 3-year PFS of 74% were achieved [10]. Table 4 compared different outcome parameters of GORTEC study and the study by Sun et al. with our present study.
Table 4: Characteristics of selected randomized studies on induction TPF in NPC and comparison with the present study.
|
Author |
No. of patients |
Stage IVA (%) |
Induction chemotherapy regimen |
% of patients with GCSF |
% for patients completing 3 cycles TPF |
% of patients receiving planned dose TPF |
Concurrent cisplatin compliance |
G3/4 toxicities |
Survival outcomes |
|
Sun et al. [11] |
480 |
45.4 |
Docetaxel 60 mg/m2 D1 Cisplatin 60 mg/m2 D1 5FU 600 mg/m2 D1-5 Q3 weeks |
0 |
88 |
78 |
79% received ≥ 200 mg/m2 |
Neutropenia – 35% Neutropenic fever – 2.5 % Mucositis- 6.5% Diarrhea – 7% |
3-year OS 85.6% 3-year PFS 77.4% |
|
Frikha et al. [10] |
83 |
NR |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750 mg/m2 D1-5 Q3 weeks |
15 |
95 |
75 |
86 % received ≥200 mg/m2 |
Neutropenia -27.5% Neutropenic fever – 7.5 Mucositis- 12.5% Diarrhea - |
3-year OS 86.3% 3-year PFS 73.9% |
|
Present study |
28 |
100 |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750 mg/m2 D1-5 Q3 weeks |
100 |
93 |
71 |
Mean dose of cisplatin received : 221 mg/m2 |
Neutropenia -11% Neutropenic fever – 3.6% Mucositis- 0% Diarrhea – 8% |
*3-year OS 100% §3-year PFS 92.2% |
NR: Not Reported, GCSF: Granulocyte Colony Stimulating Factor; OS: Overall Survival; PFS: Progression Free survival.
Distant metastasis is the predominant failure pattern in stage IVA NPC. Hence, it is logical to tackle this by intensifying systemic chemotherapy while maintaining treatment compliance. In the present study, we used full conventional dose of TPF to explore its tolerability in Chinese patients. Compliance to TPF was comparable to that reported in the GORTEC study. 93% patients completed 3 cycles of TPF, compared to 95% and 88% of patients in GORTEC and Sun study respectively [10, 11]. 7% of our patients received two cycles of TPF as compared to 5% patients in the two phase III studies. In fact, only one patient in our cohort did not complete the planned three cycles due to G4 toxicity of STEMI, while another patient had received one cycle of IC PF in another hospital before commencing TPF in our unit, thus the last cycle was not given. By omitting this exceptional patient, the compliance with TPF was up to 96%. Overall, 71% of our patients completed full dose TPF at cycle 3 compared with 75% and 78% of patients in the GORTEC and Sun study respectively [10, 11].
The incidence of G3/4 toxicities during TPF was much lower in our study. This was accounted by the routine use of prophylactic G-CSF in our patients. This was also in line with emerging clinical practice to incorporate prophylactic G-CSF to reduce the incidence of hematological toxicities and infection risk associated with TPF [28]. On the other hand, G-CSF was not used in the Sun study, while only 15% of patients received G-CSF in GORTEC trial [10, 11]. The incidence of G3/4 neutropenia in the current study was 11% only, compared with 27.5% and 35% in the GORTEC and Sun study. We also reported a low incidence of febrile neutropenia of 3.6%, compared to 7.5% in the GORTEC study, and a similar incidence of 2.5% in the dose reduced TPF study reported by Sun et al. G3/4 mucositis was significant in the GORTEC study (12.5%) and the Sun study (6.5%), but did not occur in our cohort. The incidence of G3/4 diarrhea in our study was comparable to the Sun study, at 7% and 8% respectively.
From the latest IPD network meta-analysis by MAC-NPC, the HR of locoregional control was seemingly inferior with the use of IC(taxane)-CCRT [HR – 0.89 (with 95% CI 0.62 – 1.28)] as compared to CCRT-AC [HR – 0.74 (CI 0.56 – 0.99)] [24]. One postulation owing to IC-CCRT inferiority is that potential toxicities resulting from IC may preclude subsequent delivery of full dose CCRT and hence jeopardize the locoregional control. Yet, conclusions could not be drawn due to the overlapping of CI in their hazard ratios. Furthermore, 6 out of 8 phase II/III studies had reduced cisplatin dose intensity during CCRT in the IC arm as compared to the CCRT alone arm [10, 17-19, 21, 22]. However, only 2 studies failed to report a positive result on survival [17, 18]. Nevertheless, the tolerance to cisplatin during CCRT is a crucial factor to consider. Recently, a phase III study of 298 patients demonstrated no additional advantage of using higher cumulative cisplatin dose, and concluded that cisplatin dose of 200 mg/m2 during CCRT would be adequate [29]. In our study, 79% patients received cisplatin dose of ≥ 200 mg/m2 during CCRT. Although slightly lower than the 86% patients reported in Sun study, the result was still satisfactory [11]. In the GORTEC study, the mean dose of cisplatin during CCRT was 221 mg/m2 [10].
TPF using Docetaxel 60 mg/m2 D1, Cisplatin 60 mg/m2 D1, 5FU 600 mg/m2 D1-5 every 3 weeks is a common regimen adopted in the Chinese population, yet there is no consensus on the standard TPF dose in Chinese. This regimen was based on two phase 1 studies done at Sun Yat-sen University Cancer Center [12, 13]. Recently, Jin et al. compared high vs. low-dose IC TPF followed by CCRT in Chinese patients with locoregionally advanced NPC and reported similar treatment efficacy [10]. However, the treatment arms were imbalanced with significantly more stage IVA-B patients (64%) receiving high dose TPF vs the low dose (36%). Such results should be interpreted with caution. Based on our retrospective study, conventional dose TPF was well tolerated in the Chinese population. With the use of prophylactic G-CSF, the incidence of G3/4 neutropenia and febrile neutropenia was low. Other acute toxicities were highly manageable and did not preclude subsequent delivery of CCRT. Our treatment outcomes were also promising. Overall, the 2-year LRFFS was 96.4% and 2-year DFFS was 91.3%. This is comparable to the phase III study on stage IVA-B NPC by Hong et al. which reported 3-year DFFS of 65% and 3-year OS of 85%. There are several limitations to our study. Firstly, this was retrospective analysis with inherent record bias. Secondly, only a small patient sample was included. Thirdly, our median follow up period of 2 years was relatively short.
Despite the above caveats, our study did inspire us on the optimal TPF dose in the Chinese population. We believe standard dose TPF followed by CCRT is well tolerated in Chinese patients with manageable toxicities and allows adequate dosage of concurrent cisplatin to be delivered subsequently. Survival outcomes are encouraging and routine dose reduction of TPF should be avoided, especially knowing that stage IVA disease carries high inherent risk of distant failure.
Financial Disclosure
None.
Conflicts of Interest
None.
Supplementary Table 1: TPF dose and schedule in various Phase III studies in head and neck cancer.
|
Study |
Induction chemotherapy regimen |
Concurrent chemotherapy |
|
TAX 323 [7] |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750mg/m2 D1-5 Q3 weeks |
Nil |
|
TAX 324 [8, 9] |
Docetaxel 75 mg/m2 D1 Cisplatin 100 mg/m2 D1 5FU 1000mg/m2 D1-4 Q3 weeks |
Carboplatin AUC x 1.5 Q1 week |
|
GORTEC 2000-01 [27] |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750mg/m2 D1-5 Q3 weeks |
Cisplatin, carboplatin, and 5-fluorouracil or a combination of two drugs *
|
|
TTCC 2002 [30] |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 1000mg/m2 D1-5 Q3 weeks |
Cisplatin 100 mg/m2 Q3 weeks |
|
DeCIDE [31] |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750 mg/m2 D1-5 Q3 weeks |
Hydroxyurea at 500 mg PO q12 hours × 6 days, 5-FU at 600 mg/m2/day × 5 days, Docetaxel D1 starting at 20 mg/m2 (increasing by 5 mg/m2 in successive dose levels, maximum 30 mg/m2) Q2W |
|
PARADIGM [32] |
Docetaxel 75 mg/m2 D1 Cisplatin 100 mg/m2 D1 5FU 1000 mg/m2 D1-4 Q3 weeks |
Weekly carboplatin AUC x 1.5 or Weekly docetaxel 20 mg/m² for 4 weeks (poor responder only) |
|
Italian trial [33] |
Docetaxel 75 mg/m2 D1 Cisplatin 80 mg/m2 D1 5FU 800 mg/m2 D1-4 Q3 weeks |
Cisplatin 20 mg/m2 D1-4 and 5FU 800 mg/m2 D1-D4 (weeks 1 and 6) or weekly cetuximab 250 mg/m2 |
AUC: Area Under the Concentration-Time-Curve. *Details (scheme and dosage) not stated.
Article Info
Article Type
Research ArticlePublication history
Received: Wed 28, Oct 2020Accepted: Wed 11, Nov 2020
Published: Fri 20, Nov 2020
Copyright
© 2023 Sarah W.M. Lee. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.COR.2020.11.05
Author Info
Sarah W.M. Lee Philip Y. Wu Tommy C.Y. So K. C. Lee Edwin C. Y. Wong Jim P. H. Lam Horace C.W. Choi Wai Tong Ng
Corresponding Author
Sarah W.M. LeeDepartment of Clinical Oncology, Pamela Youde Nethersole Eastern Hospital, Hong Kong
Figures & Tables
Table 1: Patient demographics.
|
Baseline characteristics |
No. of patients (%), N=28 |
|
Age Mean (range) |
49 y (33-61) |
|
Sex Male/Female |
21 (75%) / 7 (25%) |
|
ECOG 0 1 2 |
10 (35.7%) 17 (60.7%) 1 (3.57%) |
|
Histology Keratinized, squamous Non-keratinized squamous Undifferentiated |
1 (3.57%) 1 (3.57%) 26 (92.9%) |
|
T category 1 2 3 4 |
2 (7.14%) 2 (7.14%) 7 (25%) 17 (60.7%) |
|
N category 0 1 2 3 |
3 (10.7%) 9 (32.1%) 4 (14.3%) 12 (42.9%) |
|
Radiation dose 70 Gy 30 Gy (palliative dose) |
27 (96.4%) 1 (3.57%) |
|
Baseline EBV DNA Mean (range) |
3683 copies/ml (0-28750) |
|
Post radical RT |
No of patients (%), N=27 |
|
6 weeks post-treatment NP biopsy No residue Residue |
26 (96.2%) 1 (3.7%) |
|
8 weeks post-treatment EBV DNA 0 copy >0 copy Missing data |
24 (88.9%) 3 (11.1%) 0 |
|
26 weeks post-treatment EBV DNA 0 copy >0 copy Missing data |
23 (85.2%) 2 (7.4%) 2 (7.4%) |
|
16 weeks post-treatment MRI or PET Complete remission Partial response Missing data |
26 (96.2%) 1 (3.7%) 0 |
|
Median follow up Median (range) |
2 years (8.6-39.5 months) |
Table 2: Compliance and tolerance to conventional dose TPF, N=28 patients.
|
Dose/cycle |
1st TPF |
2nd cycle Docetaxel |
2nd cycle Cisplatin |
2nd cycle 5 fluorouracil |
3rd cycle Docetaxel |
3rd cycle Cisplatin |
3rd cycle 5-fluorouracil |
|
100% |
28 (100%) |
22 (78.6%) |
22 (78.6%) |
22 (78.6%) |
21 (75%) |
20 (71.4%) |
20 (71.4%) |
|
80% |
0 |
6 (21.4%) |
6 (21.4%) |
6 (21.4%) |
5 (17.9%) |
6 (21.4%) |
5 (17.9%) |
|
0% |
0 |
0 |
0 |
0 |
2 (7.14%)* |
2 (7.14%)* |
2 (7.14%)* |
*One patient received 1st cycle chemotherapy (consisting of cisplatin and 5 fluorouracil) at another institution; another developed ST-elevation myocardial infarction.
Table 3: Acute toxicities during TPF and CCRT.
|
|
TPF (n=28) |
CCRT (n=27) |
||||||
|
Toxicity/Grade |
1 |
2 |
3 |
4 |
1 |
2 |
3 |
4 |
|
Vomiting |
6 (21.4%) |
2 (7.14%) |
2 (7.14%) |
0 |
3 (11.1%) |
2 (7.41%) |
3 (11.1%) |
0 |
|
Diarrhea |
6 (21.4%) |
2 (7.14%) |
7 (25%) |
0 |
0 |
0 |
0 |
0 |
|
HFS |
1 (3.57%) |
0 |
0 |
0 |
0 |
0 |
1 (3.70%) |
0 |
|
Neutropenia |
1 (3.57%) |
0 |
1 (3.57%) |
2 (7.14%) |
4 (14.8%) |
5 (18.5%) |
7 (25.9%) |
1 (3.70%) |
|
Neutropenic fever |
1 (3.57%) |
0 |
1 (3.57%) |
0 |
2 (7.40%) |
0 |
1 (3.70%) |
0 |
|
Non-neutropenic infection |
0 |
2 (7.14%) |
1 (3.57%) |
0 |
0 |
0 |
2 (7.40%) |
0 |
|
Anaemia |
17 (60.7%) |
6 (21.4%) |
2 (7.14%) |
0 |
10 (37.0%) |
8 (29.6%) |
9 (33.3%) |
0 |
|
Thrombocytopenia |
7 (25%) |
0 |
0 |
0 |
11 (40.7%) |
5 (18.5%) |
0 |
0 |
|
Hearing loss |
0 |
0 |
0 |
0 |
1 (3.70%) |
0 |
0 |
0 |
|
Mucositis |
1 (3.57%) |
0 |
0 |
0 |
2 (7.41%) |
12 (44.4%) |
13 (48.1%) |
0 |
|
Neuropathy |
2 (7.14%) |
0 |
0 |
0 |
0 |
1 (3.70%) |
0 |
0 |
|
Impaired renal function |
9 (32.1%) |
0 |
0 |
0 |
14 (51.9%) |
1 (3.70%) |
0 |
0 |
|
Increased alanine transaminase |
7 (25%) |
1 (3.57%) |
0 |
0 |
2 (7.40%) |
1 (3.70%) |
0 |
0 |
|
Increased bilirubin |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
Hypokalaemia |
14 (50%) |
3 (10.7%) |
0 |
0 |
6 (22.2%) |
4 (14.8%) |
0 |
0 |
|
Hyponatremia |
13 (46.4%) |
2 (7.14%) |
2 (7.14%) |
1 (3.57%) |
13 (48.1%) |
0 |
2 (7.41%) |
0 |
|
Skin Reaction |
|
|
|
|
11 (40.7%) |
12 (44.4%) |
3 (11.1%) |
0 |
|
Tube Feeding |
|
|
|
|
3 (11.1%) |
0 |
0 |
0 |
|
Others* |
0 |
0 |
0 |
1 (3.57%)* |
|
|
|
|
CCRT: Concurrent Chemoradiotherapy; HFS: Hand Foot Syndrome; *ST- elevation myocardial infarction. *estimated 3-year OS; §estimated 3-year PFS.
Table 4: Characteristics of selected randomized studies on induction TPF in NPC and comparison with the present study.
|
Author |
No. of patients |
Stage IVA (%) |
Induction chemotherapy regimen |
% of patients with GCSF |
% for patients completing 3 cycles TPF |
% of patients receiving planned dose TPF |
Concurrent cisplatin compliance |
G3/4 toxicities |
Survival outcomes |
|
Sun et al. [11] |
480 |
45.4 |
Docetaxel 60 mg/m2 D1 Cisplatin 60 mg/m2 D1 5FU 600 mg/m2 D1-5 Q3 weeks |
0 |
88 |
78 |
79% received ≥ 200 mg/m2 |
Neutropenia – 35% Neutropenic fever – 2.5 % Mucositis- 6.5% Diarrhea – 7% |
3-year OS 85.6% 3-year PFS 77.4% |
|
Frikha et al. [10] |
83 |
NR |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750 mg/m2 D1-5 Q3 weeks |
15 |
95 |
75 |
86 % received ≥200 mg/m2 |
Neutropenia -27.5% Neutropenic fever – 7.5 Mucositis- 12.5% Diarrhea - |
3-year OS 86.3% 3-year PFS 73.9% |
|
Present study |
28 |
100 |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750 mg/m2 D1-5 Q3 weeks |
100 |
93 |
71 |
Mean dose of cisplatin received : 221 mg/m2 |
Neutropenia -11% Neutropenic fever – 3.6% Mucositis- 0% Diarrhea – 8% |
*3-year OS 100% §3-year PFS 92.2% |
NR: Not Reported, GCSF: Granulocyte Colony Stimulating Factor; OS: Overall Survival; PFS: Progression Free survival.
Supplementary Table 1: TPF dose and schedule in various Phase III studies in head and neck cancer.
|
Study |
Induction chemotherapy regimen |
Concurrent chemotherapy |
|
TAX 323 [7] |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750mg/m2 D1-5 Q3 weeks |
Nil |
|
TAX 324 [8, 9] |
Docetaxel 75 mg/m2 D1 Cisplatin 100 mg/m2 D1 5FU 1000mg/m2 D1-4 Q3 weeks |
Carboplatin AUC x 1.5 Q1 week |
|
GORTEC 2000-01 [27] |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750mg/m2 D1-5 Q3 weeks |
Cisplatin, carboplatin, and 5-fluorouracil or a combination of two drugs *
|
|
TTCC 2002 [30] |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 1000mg/m2 D1-5 Q3 weeks |
Cisplatin 100 mg/m2 Q3 weeks |
|
DeCIDE [31] |
Docetaxel 75 mg/m2 D1 Cisplatin 75 mg/m2 D1 5FU 750 mg/m2 D1-5 Q3 weeks |
Hydroxyurea at 500 mg PO q12 hours × 6 days, 5-FU at 600 mg/m2/day × 5 days, Docetaxel D1 starting at 20 mg/m2 (increasing by 5 mg/m2 in successive dose levels, maximum 30 mg/m2) Q2W |
|
PARADIGM [32] |
Docetaxel 75 mg/m2 D1 Cisplatin 100 mg/m2 D1 5FU 1000 mg/m2 D1-4 Q3 weeks |
Weekly carboplatin AUC x 1.5 or Weekly docetaxel 20 mg/m² for 4 weeks (poor responder only) |
|
Italian trial [33] |
Docetaxel 75 mg/m2 D1 Cisplatin 80 mg/m2 D1 5FU 800 mg/m2 D1-4 Q3 weeks |
Cisplatin 20 mg/m2 D1-4 and 5FU 800 mg/m2 D1-D4 (weeks 1 and 6) or weekly cetuximab 250 mg/m2 |
AUC: Area Under the Concentration-Time-Curve. *Details (scheme and dosage) not stated.
References
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA et al. (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68: 394-424. [Crossref]
- Baujat B, Audry H, Bourhis J, Chan ATC, Onat H et al. (2006) Chemotherapy in locally advanced nasopharyngeal carcinoma: An individual patient data meta-analysis of eight randomized trials and 1753 patients. Int J Radiat Oncol Biol Phys 64: 47-56. [Crossref]
- Blanchard P, Lee A, Marguet S, Leclercq L, Ng WT et al. (2015) Chemotherapy and radiotherapy in nasopharyngeal carcinoma: an update of the MAC-NPC meta-analysis. Lancet Oncol 16: 645-655. [Crossref]
- Au KH, Ngan RKC, Ng AWY, Poon DMC, Ng WT et al. (2018) Treatment outcomes of nasopharyngeal carcinoma in modern era after intensity modulated radiotherapy (IMRT) in Hong Kong: A report of 3328 patients (HKNPCSG 1301 study). Oral Oncol 77: 16-21. [Crossref]
- Lee AWM, Ngan RKC, Tung SY, Cheng A, Kwong DLW et al. (2015) Preliminary results of trial NPC-0501 evaluating the therapeutic gain by changing from concurrent-adjuvant to induction-concurrent chemoradiotherapy, changing from fluorouracil to capecitabine, and changing from conventional to accelerated radiotherapy fractionation in patients with locoregionally advanced nasopharyngeal carcinoma. Cancer 121: 1328-1338. [Crossref]
- Ng WT, Corry J, Langendijk JA, Lee AWM, Mäkitie A et al. (2020) Current management of stage IV nasopharyngeal carcinoma without distant metastasis. Cancer Treat Rev 85: 101995. [Crossref]
- Vermorken JB, Remenar E, van Herpen C, Gorlia T, Mesia R et al. (2007) Cisplatin, Fluorouracil, and Docetaxel in Unresectable Head and Neck Cancer. N Engl J Med 357: 1695-1704. [Crossref]
- Posner MR, Hershock DM, Blajman CR, Mickiewicz E, Winquist E et al. (2007) Cisplatin and fluorouracil alone or with docetaxel in head and neck cancer. N Engl J Med 357: 1705-1715. [Crossref]
- Lorch JH, Goloubeva O, Haddad RI, Cullen K, Sarlis N et al. (2011) Induction chemotherapy with cisplatin and fluorouracil alone or in combination with docetaxel in locally advanced squamous-cell cancer of the head and neck: long-term results of the TAX 324 randomised phase 3 trial. Lancet Oncol 12: 153-159. [Crossref]
- Frikha M, Auperin A, Tao Y, Elloumi F, Toum N et al. (2018) A randomized trial of induction docetaxel–cisplatin–5FU followed by concomitant cisplatin-RT versus concomitant cisplatin-RT in nasopharyngeal carcinoma (GORTEC 2006-02). Ann Oncol 29: 731-736. [Crossref]
- Sun Y, Li WF, Chen NY, Zhang N, Hu G et al. (2016) Induction chemotherapy plus concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: a phase 3, multicentre, randomised controlled trial. Lancet Oncol 17: 1509-1520. [Crossref]
- Zhang QH, Luo W, Zhou QC, Yu Z, Ma J et al. (2009) TPF induction chemotherapy followed by intensity-modulated radiotherapy and concomitant chemotherapy for locoregionally advanced nasopharyngeal carcinoma. Chin J Cancer Prevent Treat 16: 625-628.
- Guo L, Lin HX, Xu M, Chen QH, Wang CT et al. (2010) Phase I study of TPF neoadjuvant chemotherapy followed by radical radiotherapy in advanced nasopharyngeal carcinoma. Chin J Cancer 29: 136-139. [Crossref]
- Ng WT, Lee MCH, Hung WM, Choi CW, Lee KC et al. (2011) Clinical Outcomes and Patterns of Failure After Intensity-Modulated Radiotherapy for Nasopharyngeal Carcinoma. Int J Rad Oncol Biol Phys 79: 420-428. [Crossref]
- Al Sarraf M, LeBlanc M, Giri PG, Fu KK, Cooper J et al. (1998) Chemoradiotherapy versus radiotherapy in patients with advanced nasopharyngeal cancer: phase III randomized Intergroup study 0099. J Clin Oncol 16: 1310-1317. [Crossref]
- Hui EP, Ma BB, Leung SF, King AD, Mo F et al. (2009) Randomized Phase II Trial of Concurrent Cisplatin-Radiotherapy With or Without Neoadjuvant Docetaxel and Cisplatin in Advanced Nasopharyngeal Carcinoma. J Clin Oncol 27: 242-249. [Crossref]
- Fountzilas G, Ciuleanu E, Bobos M, Kalogera Fountzila A, Eleftheraki AG et al. (2012) Induction chemotherapy followed by concomitant radiotherapy and weekly cisplatin versus the same concomitant chemoradiotherapy in patients with nasopharyngeal carcinoma: a randomized phase II study conducted by the Hellenic Cooperative Oncology Group (HeCOG) with biomarker evaluation. Ann Oncol 23: 427-435. [Crossref]
- Tan T, Lim WT, Fong KW, Cheah SL, Soong YL et al. (2015) Concurrent Chemo-Radiation With or Without Induction Gemcitabine, Carboplatin, and Paclitaxel: A Randomized, Phase 2/3 Trial in Locally Advanced Nasopharyngeal Carcinoma. Int J Radiat Oncol Biol Phys 91: 952-960. [Crossref]
- Hong RL, Hsiao CF, Ting LL, Ko JY, Wang CW et al. (2018) Final results of a randomized phase III trial of induction chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in patients with stage IVA and IVB nasopharyngeal carcinoma-Taiwan Cooperative Oncology Group (TCOG) 1303 Study. Ann Oncol 29: 1972-1979. [Crossref]
- Yang Q, Cao SM, Guo L, Hua YJ, Huang PY et al. (2019) Induction chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: long-term results of a phase III multicentre randomised controlled trial. Eur J Cancer 119: 87-96. [Crossref]
- Zhang Y, Chen L, Hu GQ, Zhang N, Zhu XD et al. (2019) Gemcitabine and Cisplatin Induction Chemotherapy in Nasopharyngeal Carcinoma. N Engl J Med 381: 1124-1135. [Crossref]
- Li WF, Chen NY, Zhang N, Hu GQ, Xie FY et al. (2019) Concurrent chemoradiotherapy with/without induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: Long‐term results of phase 3 randomized controlled trial. Int J Cancer 145: 295-305. [Crossref]
- Ribassin Majed L, Marguet S, Lee AWM, Ng WT, Ma J et al. (2017) What Is the Best Treatment of Locally Advanced Nasopharyngeal Carcinoma? An Individual Patient Data Network Meta-Analysis. J Clin Oncol 35: 498-505. [Crossref]
- Petit C, Lee AWM, Carmel A, Ng WT, Ma J et al. (2020) Network-meta-analysis of chemotherapy in nasopharyngeal carcinoma (MAC-NPC): An update on 8,221 patients. J Clin Oncol 38: 6523-6523.
- National Comprehensive Cancer Network (2020) NCCN Guidelines.
- Chan ATC, Grégoire V, Lefebvre JL, Licitra L, Hui EP et al. (2012) Nasopharyngeal cancer: EHNS–ESMO–ESTRO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 23: vii83-vii85. [Crossref]
- Janoray G, Pointreau Y, Garaud P, Chapet S, Alfonsi M et al. (2015) Long-Term Results of a Multicenter Randomized Phase III Trial of Induction Chemotherapy With Cisplatin, 5-fluorouracil, ± Docetaxel for Larynx Preservation. J Natl Cancer Inst 108: djv368. [Crossref]
- Haddad RI, Posner M, Hitt R, Cohen EEW, Schulten J et al. (2018) Induction chemotherapy in locally advanced squamous cell carcinoma of the head and neck: role, controversy, and future directions. Ann Oncol 29: 1130-1140. [Crossref]
- Peng H, Chen L, Zhang Y, Li WF, Mao YP et al. (2016) Prognostic Value of the Cumulative Cisplatin Dose During Concurrent Chemoradiotherapy in Locoregionally Advanced Nasopharyngeal Carcinoma: A Secondary Analysis of a Prospective Phase III Clinical Trial. Oncologist 21: 1369-1376. [Crossref]
- Hitt R, Grau JJ, López Pousa A, Berrocal A, García Girón C et al. (2014) A randomized phase III trial comparing induction chemotherapy followed by chemoradiotherapy versus chemoradiotherapy alone as treatment of unresectable head and neck cancer. Ann Oncol 25: 216-225. [Crossref]
- Cohen EEW, Karrison TG, Kocherginsky M, Mueller J, Egan R et al. (2014) Phase III Randomized Trial of Induction Chemotherapy in Patients With N2 or N3 Locally Advanced Head and Neck Cancer. J Clin Oncol 32: 2735-2743. [Crossref]
- Haddad R, O'Neill A, Rabinowits G, Tishler R, Khuri F et al. (2013) Induction chemotherapy followed by concurrent chemoradiotherapy (sequential chemoradiotherapy) versus concurrent chemoradiotherapy alone in locally advanced head and neck cancer (PARADIGM): a randomised phase 3 trial. Lancet Oncol 14: 257-264. [Crossref]
- Ghi MG, Paccagnella A, Ferrari D, Foa P, Alterio D et al. (2017) Induction TPF followed by concomitant treatment versus concomitant treatment alone in locally advanced head and neck cancer. A phase II–III trial. Ann Oncol 28: 2206-2212. [Crossref]
