Dyskeratosis Congenita: Squamous Cell Carcinoma of the Buccal Mucosa on a Background of Lichen Planus – A Case Report During COVID-19 Pandemic
A B S T R A C T
Dyskeratosis Congenita is a severe disorder that involves several systems in the human body and is well-known for its transmission hereditarily through generations. The triad of a) reticular atrophy of the skin with pigmentation b) dystrophy of the nails c) leukoplakia of the oral mucosa characterises this uncommon disorder, which most commonly affects men. One of the key reasons for premature death in this disease is malignant transformation within areas of mucosal hyperkeratosis. In Dyskeratosis Congenita, the simultaneous existence of squamous cell carcinoma (SCC) with lichen planus and or lichenoid reactions in the mucosa of the oral cavity is a rare, stated condition. This report discusses a case of a Libyan male patient affected by Dyskeratosis Congenita that ended with buccal mucosal squamous cell carcinoma developed on a lichen planus background after thirty years of suffering at the age of 64.
Keywords
Carcinoma, congenita, dyskeratosis, oral, mucosa, squamous
Introduction
Dyskeratosis Congenita is a rare genodermatosis that was first reported in 1906. It is a severe inherited, multisystem disorder which is also called as Zinsser-Engman-Cole syndrome, after its first documenters [1-3]. It frequently occurs in males and, in its conventional pattern, is hallmarked by the triad of a) reticular atrophy of the skin with pigmentation, b) dystrophy of the nails and c) leukoplakia of oral mucosa [1-3]. While most cases are X-linked, there have been reports of autosomal dominant and recessive patterns. The importance of the disorder lies in early mortality resulting from the failure of the bone marrow or malignant transformation within mucosal hyperkeratosis areas. There have been reports of different mucocutaneous and non-mucocutaneous features, but progressive bone marrow failure is the principal cause of early mortality. The development of squamous cell carcinoma (SCC) in the buccal mucosa in association with the oral lichen planus/lichenoid reaction is a previously uncommon reported finding seen in dyskeratosis congenita.
SCC is one of the most frequent cancers of the skin and oral mucosa. Internationally, it is considered to be the sixth most common intra-oral malignant tumors, constituting approximately 3% of all cancer cases that are newly diagnosed throughout the world, with a predominant increase in its incidence [4, 5]. SCC usually occurs in elderly patients from age 50 to 80, with a propensity towards men rather than women reported as the commonest malignancy among men (17%) and the third commonest malignancy in women (11%) [6, 7]. Among the different malignancies of the oral cavity, SCC of the buccal mucosa is seen more frequently [5-7]. Buccal mucosa was accounted to be the fourth more prevalent location after the lower jaw, tongue, and upper jaw representing around 10% of all oral malignant growths [8-10].
Squamous cell carcinoma (SCC) of buccal mucosa principally arises by the side of the occlusal plane and is marked by pain and the presence of an ulcerated lesion which is generally associated with a buccal mass. In comparison with the various SCCs that appear at different locations in the oral cavity, buccal mucosa SCC is characterised by its ability to progress faster and invade aggressively to adjacent and deeper structures with a high rate of local-regional relapse after treatment. The buccal site often has an early metastasis to lymph nodes rendering it difficult to achieve negative surgical margins. In this way, the SCC of buccal mucosa necessitates vigilant treatment even in its initial onset. From an anatomical perspective, buccal mucosa is associated with different structures and regions in the head, such as the vestibule of the mouth, retromolar trigone, and the masseter muscle. In this way, buccal SCC can penetrate to contiguous structures, for example, mandible and maxilla, masticatory muscles and cheeks. As a result, the SCC of the buccal mucosa has slight or no physical siege to arrest its expansion, complicating surgical removal and the following reconstruction of damaged tissues and structures, particularly when the malignancy attacks different spaces in the region such as the masticatory space. It is important to adequately reconstruct the defects that result from surgical resection of the malignant growth to reduce functional and aesthetic problems.
Lichen planus is a chronic mucocutaneous condition that can appear on the skin and/or oral mucosa. Oral lichen planus is a common oral mucosal inflammatory disease that implicates anomalous immune response mediated by mucosal cells [11, 12]. Oral lichen planus frequency has been described as 1.3% occurring mainly in mid-life with more female predilection [13, 14]. Clinically, Oral lichen planus manifests in different patterns, including ulcerative (erosive), atrophic, papular, plaque, and reticular forms [11]. Lesions occur mostly on both sides of the oral cavity and are more regularly observed on the buccal mucosa compared with other oral sites such as gingiva, tongue, and mucobuccal fold. Oral lichen planus is therefore categorised as a potentially malignant disorder affecting the oral mucosa [15].
Grinspan’s syndrome was reported as having a symptomatological triad-arterial hypertension, diabetes mellitus and erosive oral lichen planus with malignant transformation [16]. It should also be borne in mind that the erosive and atrophic types may have the same image of carcinoma [17]. Malignant transformation into carcinoma in situ (28.5%) and to microinvasive carcinoma (30-38%) is reported in the majority of patients with a lower incidence of stage I and II carcinoma [18, 19]. Oral lichen planus predisposes to the occurrence of numerous primary metachronous tumors of the oral cavity and of lymph node metastases. Thus, clinical check-ups are recommended to be performed for oral lichen planus every two months for the first 6-9 months since the time of initial diagnosis and then the lesion should be examined 3 times a year thereafter [19].
We report a case of a Libyan male patient who was diagnosed by his dermatologist in 1987 as a case of Rothmund-Thomson disease at age 32. The patient at that time complained of erythematous rough skin lesions on the sun-exposed areas along with some lesions on legs, thighs and buttocks, since childhood. Skin examination showed erythemo-squamous lesions on the face with telangiectasia more marked on the sides of the nose. The neck, particularly the nape of the neck showed an erythematous, reticulate pattern with atrophic changes, telangiectasia and slight pigmentation (poikilodermal changes). Actinic cheilitis was also presented. The dorsum of both hands showed erythematous atrophic changes with telangiectasia. Also, the legs showed xerosis with follicular hypertrophy and plugging of follicles on the thighs and buttocks. While the initial impression was Rothmund-Thomson disease, additional follow-up suggested the diagnosis of dyskeratosis congenita due to positive family history. Furthermore, the report demonstrated the details of how his condition evolved, culminating with his presentation at the age of 64 with buccal mucosal SCC developing on the background of lichen planus located on the inner side of the left cheek. The importance of this case report lies in several areas, the first of which is the occurrence of this carcinoma as a regional recurrence on a background of lichen planus after 30 years since the last lower lip tumor resection. Secondly, management of such cases under the climate of COVID-19 is challenging. Finally, this is a case of a medically compromised patient with long-standing skin disease and Grinspan’s syndrome.
Case Report
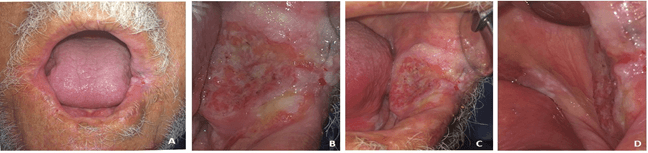
A 64-year aged Libyan man in apparently good health reported to our dental clinic at National Cancer Institute (NCI) in Misrata city on 20/02/2020 complaining of pain related to a lesion in the inner side of his left cheek that failed to heal over a year. This was accompanied by a continuous and deep pain pricking in nature, aggravated when chewing and at night with no alleviating factors. The patient had approached a general practitioner who prescribed anti-inflammatory sedative tablets along with analgesic ointment, but there was no reduction in pain nor in the lesion size. No history of adverse habits was reported. Intraoral examination revealed the presence of an oval-shaped, large exophytic ulcerative lesion of approximately 4cm (anteroposterioly) x 3cm (height) in size with granulating surfaces and well-defined, hard margins positioned at the anterior and middle of the left buccal mucosa. Overall, the peripheries of this ulcerative lesion extend as a thick white plaque, anteriorly to the left angle of the mouth, posteriorly the retromolar area and floor of the mouth surrounded with slightly erythematous mucosa (Figure 1). On extraoral examination, a white lesion was observed on the left commissure of the mouth. The skin over the lesion was normal and not tender on palpation. There was no evidence of palpable cervical lymphadenopathy.
Figure 1: Clinical pictures of the patient’s mouth. A) Previously operated lower lip with left commissure affected by the lesion. B) Ulceroproliferative growth in the left buccal mucosa. C) & D) Extension of the lesion as a thick white plaque to the floor of the mouth and left retromolar area.
Past Medical History
On the 13th of May 1987, the patient was diagnosed with Chronic Hereditary Actinic Dermatoses, (Rothmund-Thomson disease); Autosomal Dominant with Poikilodermal changes (Precancerous). On the 7th of July 1990, the patient was examined in the department of oral and maxillofacial surgery for his chief complaints of pain and a burning sensation relating to the lower lip. Intraoral examination of the lower lip revealed an ulcerative lesion measuring approximately 1x1 cm, involving the right lower lip mucosa and vermilion border. The ulcer was indurated and would bleed on touch. There was no involvement of local lymph nodes. An incisional biopsy was performed on the 7th of July 1990 and the subsequent histopathological report was conclusive for verrucous carcinoma lower lip. On the 9th of August 1990, wedge resection of the lower lip was performed along with the lesion with wide safety margins. A scar of the previous surgical operation was visible on the lower lip (Figure 1A). Postoperatively, the patient recovered well, and the healing of the surgical site was satisfactory. Histopathological diagnosis of the excised specimen reconfirmed the previous diagnosis of verrucous carcinoma. Subsequently, he was referred to a radiotherapist who advised an interstitial implant or deep X-ray therapy for the lower lip. It is likely that the patient received the above treatment in abroad (November 1990). While the details of the radiotherapy are not available, the patient had constant follow-up appointments and showed no signs of recurrence for approximately three years.
In January 1994, a patch of leukoplakia over the left lower lip mucosa and both corners of the mouth were discovered. This white lesion was excised and as per the biopsy report on 8th of January 1994, there were no signs of malignancy. Subsequent follow-ups on the 8th of September 1994 and the 26th of July 1995 did not reveal any signs of recurrence. However, the patient was advised for frequent follow-ups in the near future.
On the 15th of November 1997, surgery was required to remove a small nodule that appeared in the labial mucosa of the lower lip along with leukoplakia-like patches related to attached gingiva interdental papilla between the lower lateral incisor and canine on the right side. The histopathological diagnosis was nonspecific inflammation and epithelial dysplasia, respectively. On the 9th of June 2010, the patient was examined in the department of oral and maxillofacial surgery at NCI of Misrata for his primary complaint of a growth that appeared on the left corner of his mouth. Examination revealed the presence of an oral commissural fissure lesion on the left side. An excisional biopsy was performed on the same day and histopathological investigation concluded sebaceous hyperplasia.
On the 21st of January 2013, the patient reported to the dental clinic at National Cancer Institute in Misrata city with a recurrent ulcer on his lower lip and a skin punch biopsy specimen was taken from the lesion. Microscopic evaluation revealed heavy focal, mostly mononuclear cell infiltration and fibrosis of the dermis and prominent acanthosis with severe hyperparakeratosis. The histological diagnosis was chronic nonspecific inflammation with acanthosis and parakeratosis. On the 10
Figure 2: Highly differentiated squamous cell carcinoma of upper face skin.
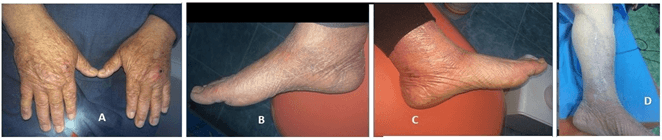
The patient was a heavy smoker since a young age with a history of withdrawal from 1990. He is diabetic, hypertensive since 1997 and experienced coronary artery thrombosis in 2019, for which he was prescribed medication. In addition to these diseases, he is suffering from long-standing photodermatitis (Figure 3) also undergoing Herniotomy and Mayo’s repair of paraumbilical hernia (PUH) in 2009.
Figure 3: Clinical pictures A) Dermatitis. B) & C) Oedema was affecting the right and left legs and feet. D) Scars of previously operated left leg to treat the cardiovascular attack.
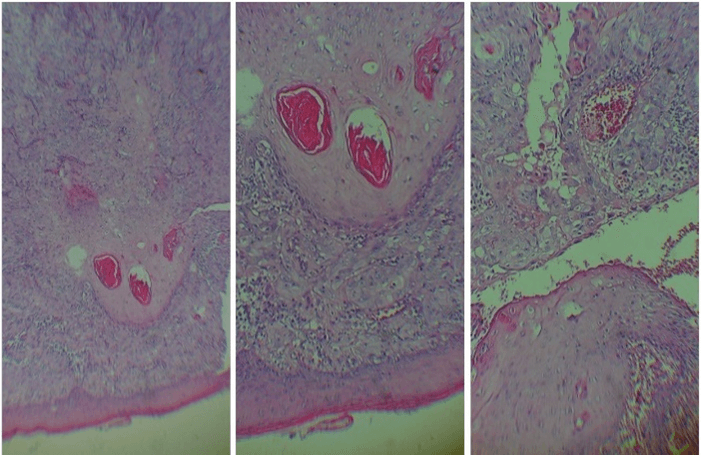
On the 20th of February 2020, because malignancy of the lesion was suspected, an incisional biopsy of the left buccal mucosa was performed under local anaesthesia. Histopathology sections of the lesion stained with haematoxylin and eosin showed invasive nests of malignant squamous cells with moderate pleomorphic nuclei, prominent nucleoli and mitosis, some of the nests present keratin pearls. The case was diagnosed as moderately differentiated invasive SCC (G2) (Figure 4). Regarding blood investigation, a complete hemogram and a carcinoembryonic antigen (CEA) tumor marker test were carried out. All the results were in the normal range except the fasting blood sugar and creatinine that showed an increase in value of 147 mg/dl and 1.81 mg/dl, respectively. The tumor marker CEA value was 4.69 ng/ml.
Figure 4: Biopsy of the left buccal mucosa. Haematoxylin and eosin showed invasive nests of malignant squamous cells with moderate pleomorphic nuclei, prominent nucleoli and mitosis. There were also some nests with keratin pearls.
To avoid waiting for an operation in Libya, the patient preferred to complete his treatment at a private clinic in abroad, although a workup had been arranged for the patient to be conducted at the NCI. Another incisional biopsy was performed there with a diagnosis confirming the previous biopsy results. Histopathology sections of the lesion using haematoxylin and eosin, May-Grünwald-Giemsa (MGG) and Periodic acid-Schiff (PAS) staining indicated that the lesion constitutes a malignant epidermoid proliferation moderately differentiated with images of coiling and cornification and dyskeratosis. Tumor cells arranged in clumps or clusters with moderate atypia sometimes stained and numerous mitoses. The stroma was the site of mild inflammatory fibrosis. This carcinoma developed on a background of lichen planus, infiltrating the muscular plane, which confirmed the diagnosis of SCC. Moreover, other investigations such as a complete body CT scan, face and brain CT scan, cardiac ultrasound and complete blood investigations were performed. A date had been fixed for operating on the lesion, but because of the COVID-19 pandemic, the appointment was cancelled, and the patient was returned home and underwent a self-isolation period of 3 weeks. After the necessary investigations against COVID-19 were performed, surgical removal of the cancer was conducted at NCI in Misrata.
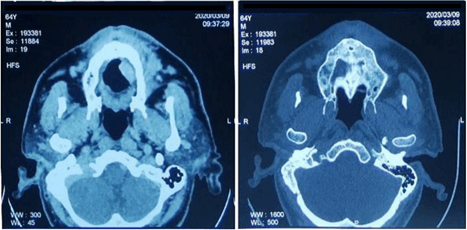
Figure 5: Patient CT scan.
The CT scan revealed a hypodense tissue process slightly enhanced after injection, related to the tumor lesion of the inner side of the left cheek, which contained some air bubbles likely ulcerated with involvement of the labial commissure and extension of the skin, 1 cm high. This mass measured 4 cm in anteroposterior, 2.4 cm in height and 1.7 cm in width, with no evidence of cervical lymph node metastasis (Figure 5). No evidence of regional or distant metastasis was found based on tomography-CT. Other CT scan findings included a subacute subdural hematoma of the left frontoparietal seat, 8 mm thick, exerting a discreet mass effect on the cortex without deviation from the median structures, sequelae of left anterior Sylvian stroke, left capsulocaudal micro-gap, sequelae of right external capsular stroke and calcifications of carotid siphons (Figure 5). At the level of thoraco-abdominal-pelvic CT scan, the following observations were made cystic dilation of the bronchi (Bronchiectasis) with signs of secondary infection, ventilation disorders in the inner segment of the middle lobe, moderate atheromatous infiltration of the mesenteric arteries, celiac trunk, renal arteries, abdominal aorta and iliacs, draft bilateral inguinal hernia with fatty content degenerative changes in the dorsolumbar spine. There was no evidence of malignant metastasis to the bone. Thus, the clinical staging was T3N0M0 (stage III). Cardiac ultrasound findings were left ventricular kinetics, discrete hypokinesia of the anteroseptal wall, type II mitral profile, pseudo normal, slightly thickened mitral valve, IM grade II, sclerotic tricuspid aortic sigmoid, medium G VG-Ao = 2 mm Hg.
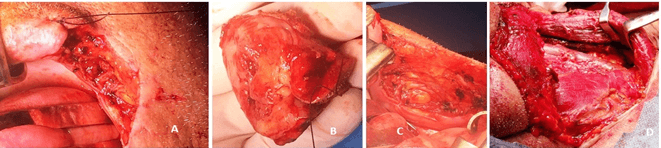
Figure 6: Tumor section. A) Resection with 1 cm margins was performed, including left commissure and left buccal region. B) Resected malignant tumor. C) Surgical site after tumor removal. D) Left neck dissection.
Surgery was performed with radical resection of the tumor with concurrent unilateral neck dissection (Figure 6). Dissection of the neck was performed as a prophylactic measure, although there was no proof of metastasis of the cervical lymph node on preoperative imaging. The surgical approach was a wide excision through the buccal mucosa to eradicate the whole tumor. The internal lining of the buccal surgical site was achieved with cutaneous grafts.
Post-Surgery
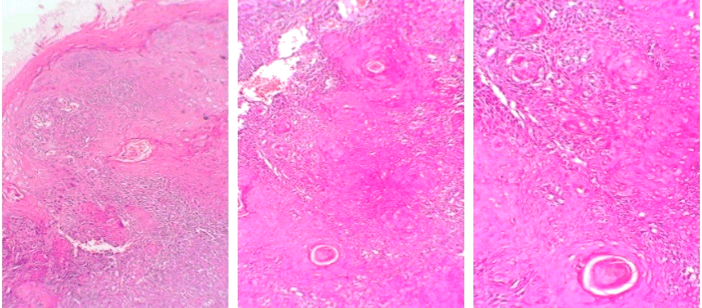
Histopathological examination of several biopsy specimens revealed a hyperplastic and dysplastic squamous epithelium showing invasive nests of malignant squamous epithelium with keratin pearls exhibiting nuclear pleomorphism, hyperchromatic nuclei, frequent mitosis, stroma contains chronic inflammatory cells (Figures 7 & 8). The medial and inferior margins were positive for malignancy, other surgical margins were negative for malignancy.
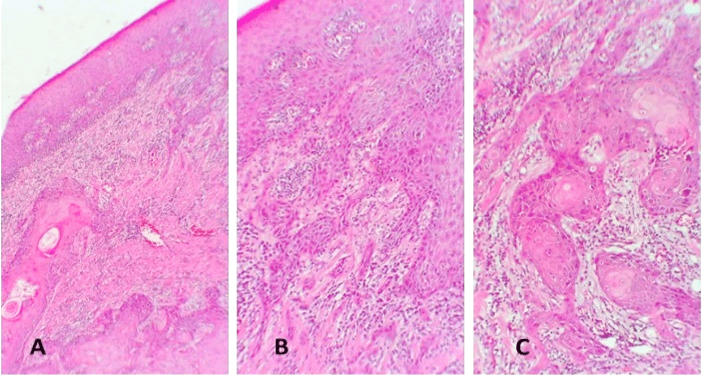
Figure 7: Hematoxylin-eosin stain sections from the buccal mucosa SCC revealed inflammation of lichen planus type. A) Hyperkeratinising stratified squamous epithelium with irregular rete ridges. The connective tissue is notable for a band-like infiltrate of lymphocytes. B) & C) A disrupted basilar area is seen in the insets. Original magnification for A) x5, insets B & C) x10.
The patient displayed a good postoperative condition without any serious abnormal clinical and laboratory outcomes until the discharge three days after the operation. Pronunciation and mastication were normal, and the patient could start a regular diet. However, a transparent/white drainage oozing externally through the left upper face skin was observed from the fifth postoperative day and continued for several days before it stopped spontaneously with regular cleaning of the related area. This was associated with a dull pain in the left preauricular region, but clinically the whole left face skin and the flap seemed well with no signs of suspicious postoperative inflammation. We considered this to be a result of the damage that took place in the salivary duct and consequently drainage of saliva extra orally. Also, the associated pain was considered to be a consequence of salivary retention and digestion of tissue by the leaked saliva oozing from the damaged Stensen’s duct. Fortunately, the patient’s pain resolved, and the transparent/white discharge stopped.
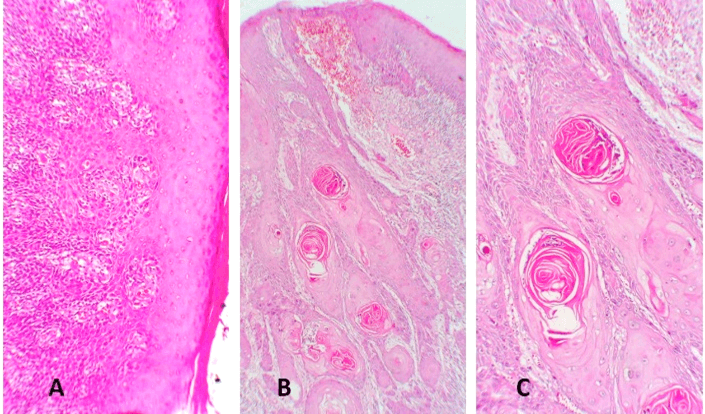
Figure 8: Hematoxylin-eosin stain sections from the buccal mucosa SCC. A) Parakeratinised stratified squamous epithelium with areas of dysplasia. The superficial underlying stroma showed several nests and islands of malignant epithelial cells. B) The neoplastic cells exhibit cellular pleomorphism, prominent nucleoli, and increased mitotic activity is seen. C) is a magnification of B). Original magnification x5 and x10 respectively.
The patient also showed the development of external submandibular swelling related to the dissected neck region on the left side. This swelling started small and soft, then became firm with fluctuation in size until it occupied a large area of the neck but did not cross the midline. The neck ultrasound conducted on the 28th of May 2020 revealed the presence of 9 x 4 cm ovoid shape focal lesion centered at the subcutaneous tissue that was hyperechoic predominantly solid with a small area of necrosis, no internal flow and no internal calcification could be detected. Consequently, the patient is re-operated on the 4th of June 2020 for further resection of the residual lesion as well as to explore and treat the neck-related swelling. Currently, the patient is well after the one-month postoperative course without any unusual clinical or abnormal laboratory findings. There are no clinical signs of relapse or new lesions.
Discussion
Dyskeratosis congenita most commonly develops between the ages 5 and 10 years. It is a progressive disease with features that appear gradually in a matter of years [20]. The sequence of occurrence of the triad of signs and symptoms is inconstant, yet the oral lesions or ulcers, leukoplakia and dental caries look to appear early [21]. The mucocutaneous characteristics of this disorder frequently manifest between ages 5 and 10. Various other anomalies have been reported in patients with dyskeratosis congenita, including involvement in dental, gastrointestinal, genitourinary, neurological, ophthalmic, pulmonary and skeletal aspects [22-25]. One of the severe and mortal consequences of this condition include the development of malignant tumors [26].
Malignant transformations have a tendency to develop beyond age 30. It has been reported that 9% of male cases developed malignancies such as myelodysplasia, Hodgkin's lymphoma and carcinomas and 6% die due to these malignancies [27]. Oral leukoplakia constitutes between 70% and 80% of three characteristic dyskeratosis congenita-related features. The buccal mucosa is the second most frequent site affected after the dorsum of the tongue, followed by other sites such as the palate, the lips and the gingiva [21, 22, 28-37]. A small number of these cases have been biopsied, so it is unknown if the lesions were already dysplastic when these patients were being examined. These areas of leukoplakia are predisposed to transform into SCCs [31, 33, 35, 38-42]. A small number of cases show a rapid reappearance of lesions and/or widespread metastasis to the lymph nodes and lung [35, 41, 42]. Findings from previous studies reported that in addition to leukoplakia, blister-like lesions that form ulcers and then leukoplakias are also present, which possibly attributed to the fact that these patients have a poor immune function that may relate to herpes virus infections [21, 31, 35, 43]. A variety of other oral abnormalities have been documented in those patients and many of them are not specific to dyskeratosis congenita [21, 22, 26, 28, 30, 31, 33-37, 40, 43-46]. It is highly possible that oral findings such as advanced periodontitis, tooth mobility, tooth loss, early exfoliation, candida infections, and impetigo are manifestations of secondary infections that occur as a result of an impaired immune response.
However, cases of oral lichen planus and lichenoid inflammatory reaction to epithelial changes also have been reported [43]. Rarely, anomalies of bone marrow may appear before the typical mucocutaneous presentations, with the marrow abnormalities manifesting at age 4-5 years, which is proposed to be the most severe phenotype of the disease [25, 45, 47-50]. In our case, no marrow abnormalities have been reported suggesting that this case may represent an incomplete expression (forme fruste) phenotype of this condition that had been reported by Bryan & Nixon, 1965 [51]. Skin involvement was not clearly identified until age 32 years on the 6th of July 1987, although it is unclear how long this had been present and to what extent seen at an earlier age. In some cases, solid malignant tumors arise in pre-existing leukoplakia and the time interval for the change varies from 10-27 years [35].
Clinical data of our patient is mostly consistent with the literature where the first malignancy appeared on his lower lip as verrucous carcinoma at the age of 34 years on the 30th of July 1990, followed by successive white lesions and ulcers developing throughout a period of thirty years. Interestingly, in our case, examination of histopathological sections of incisional biopsy and resection samples performed on the 7th of March 2020 and on the 13th of April 2020, respectively, revealed the presence of a lichen planus/lichenoid inflammatory infiltrate.
The patient presented to us on the 20th of February 2020, on the day of the first examination, with edentulous jaws. Dental involvement had not been recorded adequately in this case and the cause behind the extraction of the absent teeth is not understood. In particular, there was no documentation of the morphological and histological features of the affected teeth. However, an old dental chart dated from 2001, when the patient was aged 45, indicated that he had chronic adult periodontitis where the following teeth were present: upper and lower anterior teeth and lower posterior teeth except for the third molars. Upper premolars on the left side were present. It is reasonable to speculate that this condition would afflict the ectodermal and ectomesenchymal derivatives of the tooth. Periodontal involvement may be related to these changes in addition to the difficulty in maintaining good oral health in view of the painful ulcers in the mouth. Moreover, by age 37, this patient had developed other systemic complications such as hypertension and diabetes mellitus.
Dyskeratosis congenita and chronic Rothmund-Thomson syndrome share many general manifestations, including anomalies of the skin and nails. Diagnosis toward dyskeratosis congenita should be expected in cases where the chronological pattern is incompatible with chronic Rothmund-Thomson syndrome, where mucosal leukoplakias instead of lichenoid lesions are observed. Mapping, characterisation and identification of the X-linked dyskeratosis congenita, DKC1 gene and its corresponding protein, dyskerin, on chromosome Xq2835 is an important consideration. Dyskerin is a multifunctional protein that may be implicated in the synthesis of RNA, assembly of ribosomal subunits, and binding of centromeres/microtubules [52]. A variety of mutations have now been recognised in DKC1, which is linked with various degrees of severity of the dyskeratosis congenita phenotype. Nevertheless, when tested not all cases display mutations in DKC1. Mutations in the gene encoding for telomerase RNA (hTR) have also been described and more genes could potentially be identified over the upcoming years [53, 54]. Unfortunately, in Libya such technology for DKC1 gene mutation analysis is not currently available, which negatively impacts diagnosis and management of dyskeratosis congenita cases.
Plaque-type oral lichen planus has low incidence. However, it necessitates special consideration to cautiously differentiate it from leukoplakia. Oral lichen planus is at present categorised as a potentially malignant disorder (PMD), and several investigations have been conducted regarding the appearance of SCC in these patients [15]. The result of these investigations shows that malignant transformation ranges from 0-12.5% in oral lichen planus lesions spanning from 3 months to 24 years [55]. Although the pathogenesis of the malignant oral lichen planus transformation has not been elucidated, a relationship has been described between transformation and immunosuppressive agents such as tacrolimus, used in the treatment of this disease [56, 57]. With this particular case, we documented the possible malignant potential of oral lichen planus to developing invasive SCC. The review of the past medical history of this patient did not suggest he received any type of immunosuppressive therapy, suggesting that the buccal mucosal SCC that appeared on the inner side of his left cheek is a recurrence of lower lip malignancy.
In general, the buccal mucosal malignant tumors can be widely classified into two categories. The first category usually includes stage T1 and T2 carcinomas without spread to the buccinator muscle and accordingly have not extensively invaded the surrounding soft tissues. The second category usually comprises T3 and T4 carcinomas, which have extensive spread to the tissues of the cheek and may also invade the overlying skin [58]. Thus, buccal mucosal SCC typically presents with the local disease that manifests with ulcers and can infiltrate deep into underlying tissue involving adjacent bone, causing decreased mobility to the teeth and severe pain [59, 60]. At the advanced late stage, SCCs extend locoregionally to involve draining lymph nodes and this generally takes place as a result of permeability of lymphatic vessels by lymphatics. Therefore, the malignancy spreads to areas that are difficult to treat effectively [61]. The poor prognosis of oral cancer correlates with late diagnosis and presentation of the patient with carcinomas in their advanced stage [61, 62].
Buccal carcinomas are regarded as very destructive malignant tumors if not treated early, this destruction can be expressed in different clinical manifestations such as skin ulceration, bone invasion, invasion of pharyngomaxillary fossa and posterior growth [63]. The absence of hard tissues, which act as anatomic barriers against cancer invasion as soon as it penetrates the buccal fat, can also explain why the SCC of the oral mucosa as a whole and the buccal mucosa, in particular, are associated with poor prognosis. This renders the elimination of SCC of buccal mucosa in a number of cases highly challenging because of the tight anatomical relationship with some critical structures (muscles, lymph and blood vessels), indicating the necessity for postoperative radiotherapy [5].
The prognosis can also be affected in elderly patients by a number of factors. The majority of elderly patients usually suffer from compromising medical conditions related to pulmonary, cardiovascular, renal, and/or endocrine functions. The standard intense modalities of cancer therapy, in particular, may not be well tolerated by the patient and may actually diminish survival. Therefore, the treatment strategy for this group of patients may be limited by the previously mentioned factors [64]. No standard strategies are available for treating SCC of buccal mucosa that are directed by randomized clinical trials [65]. Treatment strategies are determined largely by the clinical staging given to the cancer at the initial clinical examination. Tumors of buccal mucosa of large size will often necessitate cheek excision (a through and through excision), accompanied by removal of a segment of nearby upper or lower jaw. The buccal branches of the facial nerve are often required to be sacrificed. Surgical treatment is known to result in obvious disfigurement even with the availability of reconstructive surgery, especially when corners of the mouth/lips are sacrificed [66].
In general, cancers with stages III and IV (late-stage) are associated with a relatively poor prognosis and often receive a combination of different approaches of therapy [67]. Adjuvant radiotherapy next to surgical resection is usually the treatment strategy of choice for more advanced malignancies that can be operated. Clinically, increasing the T stage is associated with an increase in the incidence of lymph node enlargement [65]. The surgical procedure mainly implicates a radical removal of the cancerous lesion and dissection of cervical lymph nodes. Reconstruction based on free-flap is now considered an important procedure among other techniques used for rebuilding substantial and challenging defects at surgical sites. A broadly radical resection could be approached with the support from free-flap-based reconstruction [5]. It has been reported that buccal mucosal SCC has an incidence of systemic dissemination that ranges from 0-23%, where the lungs were the most frequent sites [68, 69]. All types of buccal mucosal SCC have a high rate of recurrence with a very poor prognosis for the posterior-third type. Metastasis, most commonly associated with regional lymph nodes, will recur in approximately 56% of cases and around 35% of patients with nodal enlargement. A five-year cure rate next to surgical resection alone ranges from 77% and 65% in early-stage (I and II) to 27% and 18% in those with advanced-stage (III and IV) cancer, respectively [63]. Prognosis is affected to a large extent by the stage of cancer, this stresses the significance of early detection of buccal mucosal SCC and consequently, aggressive treatment in cases diagnosed as poorly/moderately differentiated cancer [68, 69].
In our case, a malignant neoplasm developing in the buccal mucosa was staged clinically as T3N0M0 (stage III) and no evidence of regional or distant metastasis was found based on tomography-CT. The diagnosis was accompanied by a group of risk factors linked to the patient’s general medical status (dyskeratosis congenita), age and compromised medical conditions related to cardiovascular and endocrine functions, making the surgical approach more challenging. In advanced-stage cancer, preoperative chemo-radiation is another option, but this was a resectable case. Therefore, we preferred surgical removal of the lesion and postoperative chemo-radiation.
Additionally, and as with many cancer surgical cases, the COVID-19 pandemic has resulted in limited access for many public and private medical services and facilities and creating havoc within the medical system in Libya. At the present time, Libya is ill-prepared to control any pandemic and govern its consequences as a result of the chaos and absence of central administration. Moreover, Libya's healthcare infrastructure progresses to worsen because of internal conflict, insufficient human and financial support, and poor facilities for healthcare. Libya registered an average of 47% in what concerns the readiness of health emergency services [70]. The World Health Organization and Ministry of Health of Libya reported that through its Service Availability and Readiness Assessment (SARA-2017), the majority of Libyan hospitals operate at a low level of performance. These high levels of risks and deficiency of healthcare supplies will complicate the ability of the health system to react effectively to this outbreak, rendering the residents in Libya exposed to COVID-19.
Lack of intraoperative frozen tissue analysis in operation theatres at NCI in Misrata has had a considerable negative impact on the treatment outcome of this case that needed to be operated for the second time because of the presence of positive margins in the resected tumor. Histology of frozen sections is a method of rapid intraoperative assessment of surgical specimens and has been in practice for over 100 years. Generally, this technique provides useful information for optimising the treatment procedure and is routinely used by oncosurgeons across specialties including head and neck cancer surgeries, for the assessment of surgical margins, which has an impact on final surgical margin status as well as long-term oncological outcomes. Therefore, we adopted limited surgical approaches consisting of palliative surgical removal of the lesion without resection of deep anatomical structures and this was accompanied by unilateral neck dissection of the left side.
Conclusion
This report demonstrates a case of dyskeratosis congenita in a 64-year-old male, where the mucocutaneous signs and symptoms preceded the occurrence of a variety of oral lesions. To our knowledge, the patient suffered from these lesions for more than three decades that culminated in buccal mucosal SCC associated with a lichen planus background. In comparison to different types of oral cancer, SCC that occurs in buccal mucosa is characterised as being a highly aggressive tumor with worse survival and a higher recurrence rate. It was described that early-stage SCC of buccal mucosa has poor local control and 5-year cause-specific survival rates as opposed to oral cancers at other sites such as the tongue and floor of the mouth [71]. The rate of recurrence of such types of oral SCC ranges between 30% and 80%. Therefore, it is very important at the time of surgery to provide an adequate margin of surgical resection [72-77]. There is a high possibility that advanced carcinomas of buccal mucosa give rise to latent metastasis. Thus, prophylactic neck dissection is recommended even for tumors with a size between 2 and 4 cm and without clinical evidence of regional metastasis of the lymph node [78]. In the early phase of buccal mucosa SCC, local control has been described to increase with radiotherapy after surgery [79].
This patient illustrates a number of important issues related to the diagnosis of dyskeratosis congenita:
i. Critical elements should be considered in the management of dyskeratosis congenita, including a comprehensive clinical examination, clarification of family history and close monitoring with regular follow-up.
ii. There is a need for genetic counselling and the testing for mutations in DKC1 for the purpose of early detection of the condition.
iii. All oral white lesions and ulcers, even if they appeared in patients with dyskeratosis congenita in their early ages, should be biopsied frequently and assessed for presence/absence of epithelial dysplasia, leukoplakia and premalignant changes. Wide resection of these lesions will diminish the possibility of SCC formation. Thus, appropriate surgical and/or radio-therapeutic measures should be taken.
iv. Abnormalities related to dentition are most likely associated with this multisystem disease. However, most other oral findings that have been described are likely the result of poor immune function and pancytopenia.
v. SCC of buccal mucosa has an aggressive nature and is fast expanding with a high rate of recurrence. Thus, it necessitates vigilant management even if the malignant tumor is at an early stage. It is recommended to apply prophylactic neck dissection when a malignant tumor has a size of T2 or more, and radiotherapy received after surgery may be useful for local control.
Careful monitoring is highly recommended for patients with oral lichen planus, especially old patients with plaque-like oral lichen planus that presents like leukoplakia to a large extent. Consequently, examination of the lesion tissue sections by professional oral pathologists is obligatory in order to approve the clinical diagnosis and rule out dysplasia and the presence of malignant transformation [80, 81].
Article Info
Article Type
Case ReportPublication history
Received: Fri 02, Apr 2021Accepted: Tue 20, Apr 2021
Published: Mon 03, May 2021
Copyright
© 2023 Khaled Saleh Ben Salah. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.COR.2021.05.02
Author Info
Khaled Saleh Ben Salah Fatma M. Emaetig Ebrahim H. El Mahjoubi Abdalla O. Jebriel Nada A. Oshah Firas K. Abdulmalik Mohamed A. Elfagieh Wafa A. Baba Mohamed O. Altaweel Naji Zoubia Alla Shoaoba Monir Abdelmollah Yaser O. Howayw Abdouldaim Ukwas Abdulhakim W Zaggut
Corresponding Author
Khaled Saleh Ben SalahDepartment of Pathology, National Cancer Institute (NCI), Misrata, Libya
Figures & Tables
References
1. Zinsser F (1906) Atropha cutis reticularis cum pigmentatione, dystrophia ungiumet leukoplakia oris. Ikonogr Dermatol (Hyoto) 5: 219-223.
2. Engman MF (1926) A unique case of reticular pigmentation of the skin with atrophy. Arch Bleg Dermatol Syphiligr 13: 685-687.
3. Cole HN, Rauschkolb J, Toomey J (1930) Dyskeratosis congenital with pigmentation, dystrophia unguis and leukokeratosis oris. Arch Bleg Dermatol Syphiligr 21: 71-95.
4. Fang KH, Kao HK, Cheng MH, Chang YL, Tsang NM et al. (2009) Histological differentiation of primary oral squamous cell carcinomas in an area of betel quid chewing prevalence. Otolaryngol Head Neck Surg 141: 743-749. [Crossref]
5. Coppen C, Wilde PCM, Pop LA, van den Hoogen FJA, Merkx MAW (2006) Treatment results of patients with a squamous cell carcinoma of the buccal mucosa. Oral Oncol 42: 795-799. [Crossref]
6. Iype EM, Pandey M, Mathew A, Thomas G, Sebastian P et al. (2001) Oral cancer among patients under the age of 35 years. J Postgrad Med 47: 171-176. [Crossref]
7. Iype EM, Pandey M, Mathew A, Thomas G, Nair MK(2004) Squamous cell cancer of the buccal mucosa in young adults. Br J Oral Maxillofac Surg 42: 185-189. [Crossref]
8. Kuk SK, Kim BK, Yoon HJ, Hong SD (2015) Investigation on the Age and Location of Oral Squamous Cell Carcinoma Incidence in Korea. Korean J Oral Maxillofacial Path 39: 393-402.
9. Shah JP, Cendon RA, Farr HW, Strong EW (1976) Carcinoma of the oral cavity. factors affecting treatment failure at the primary site and neck. Am J Surg 132: 504-507. [Crossref]
10. Vegers JW, Snow GB, van der Waal I (1979) Squamous cell carcinoma of the buccal mucosa. A review of 85 cases. Arch Otolaryngol 105: 192-195. [Crossref]
11. Lodi G, Scully C, Carrozzo M, Griffiths M, Sugerman PB et al. (2005) Current controversies in oral lichen planus: report of an international consensus meeting. Part 1. Viral infections and etiopathogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 100: 40-51. [Crossref]
12. Thornhill MH (2001) Immune mechanisms in oral lichen planus. Acta Odontol Scand 59: 174-177. [Crossref]
13. McCartan BE, Healy CM (2008) The reported prevalence of oral lichen planus: a review and critique. J Oral Pathol Med 37: 447-453. [Crossref]
14. Li C, Tang X, Zheng X, Ge S, Wen H et al. (2020) Global Prevalence and Incidence Estimates of Oral Lichen Planus: A Systematic Review and Meta-analysis. JAMA Dermatology 156: 172-181. [Crossref]
15. Warnakulasuriya S, Johnson NW, van der Waal I (2007) Nomenclature and classification of potentially malignant disorders of the oral mucosa. J Oral Pathol Med 36: 575-580. [Crossref]
16. Colella G, Itro A, Corvo G (1992) A case of a carcinoma arising in lichen planus in a subject with diabetes mellitus and arterial hypertension (Grinspan's syndrome). Minerva Stomatol 41: 417-420. [Crossref]
17. Collins SL (1993) Carcinoma squamoso di cavità orale, orofaringe e pareti faringee. In: Ballenger JJ. Patologia otorinolaringoiatrica Prima edizione Italiana. Milan: ABA Scientifica 346-347.
18. Mignogna MD, Muzio LL, Russo LL, Fedele S, Ruoppo E et al. (2001) Clinical guidelines in early detection of oral squamous cell carcinoma arising in oral lichen planus: a 5-year experience. Oral Oncol 37: 262-267. [Crossref]
19. Mignogna MD, Russo LL, Fedele S, Ruoppo E, Califano L et al. (2002) Clinical behaviour of malignant transforming oral lichen planus. Eur J Surg Oncol 28: 838-843. [Crossref]
20. Bodalski J, Defecinska E, Judkiewicz L, Pacanowska M (1963) FANCONI'S ANAEMIA AND DYSKERATOSIS CONGENITA AS A SYNDROME. Dermatologica 127: 330-342. [Crossref]
21. Engman MF Jr (1935) CONGENITAL ATROPHY OF THE SKIN, WITH RETICULAR PIGMENTATION: REPORT OF TWO CASES. JAMA 105: 1252-1256.
22. Sirinavin C, Trowbridge AA (1975) Dyskeratosis congenita: clinical features and genetic aspects. Report of a family and review of the literature. J Med Genet 12: 339-354. [Crossref]
23. Drachtman RA, Alter BP (1995) Dyskeratosis congenita. Dermatol Clin 13: 33-39. [Crossref]
24. Dokal I (1996) Dyskeratosis congenita: an inherited bone marrow failure syndrome. Br J Haematol 92: 775-779. [Crossref]
25. Dokal I (1999) Dyskeratosis congenita: recent advances and future directions. J Pediatr Hematol Oncol 21: 344-350. [Crossref]
26. Milgrom H, Stoll HL Jr, Crissey JT (1964) Dyskeratosis Congenita: A Case With New Features. Archives of Dermatology 89: 345-349. [Crossref]
27. Knight S, Vulliamy T, Copplestone A, Gluckman E, Mason P et al. (1998) Dyskeratosis Congenita (DC) Registry: identification of new features of DC. Br J Haematol 103: 990-996. [Crossref]
28. Wald C, Diner H (1974) Dyskeratosis congenita with associated periodontal disease. Oral Surg Oral Med Oral Pathol 37: 736-744. [Crossref]
29. Ogden GR, Connor E, Chisholm DM (1988) Dyskeratosis congenita: report of a case and review of the literature. Oral Surg Oral Med Oral Pathol 65: 586-591. [Crossref]
30. Brown CJ (2000) Dyskeratosis congenita: report of a case. Int J Paediatr Dent 10: 328-334. [Crossref]
31. Garb J (1947) Dyskeratosis congenita with pigmentation, dystrophia unguium and leukoplakia oris; patient with evidence suggestive of Addison's disease. Arch Derm Syphilol 55: 242-250. [Crossref]
32. Milgrom H, Stoll HL Jr, Crissey JT (1964) DYSKERATOSIS CONGENITA. A CASE WITH NEW FEATURES. Arch Dermatol 89: 345-349. [Crossref]
33. Anil S, Beena VT, Raji MA, Remani P, Ankathil R et al. (1994) Oral squamous cell carcinoma in a case of dyskeratosis congenita. Ann Dent 53: 15-18. [Crossref]
34. Sorrow JM, Hitch JM Jr (1963) DYSKERATOSIS CONGENITA. FIRST REPORT OF ITS OCCURRENCE IN A FEMALE AND A REVIEW OF THE LITERATURE. Arch Dermatol 88: 340-347. [Crossref]
35. Cannell H (1971) Dyskeratosis congenita. Br J Oral Surg 9: 8-20. [Crossref]
36. Ortega JA, Swanson VL, Landing BH, Hammond GD (1972) Congenital dyskeratosis. Zinsser-Engman-Cole syndrome with thymic dysplasia and aplastic anemia. Am J Dis Child 124: 701-704. [Crossref]
37. Yavuzyilmaz E, Yamalik N, Yetgin S, Kansu O (1992) Oral-dental findings in dyskeratosis congenita. J Oral Pathol Med 21: 280-284. [Crossref]
38. Cole HN, Cole HN Jr, Lascheid WP (1957) Dyskeratosis congenita; relationship to poikiloderma atrophicans vasculare and to aplastic anemia of Fanconi. AMA Arch Derm 76: 712-719. [Crossref]
39. Addison M, Rice MS (1965) THE ASSOCIATION OF DYSKERATOSIS CONGENITA AND FANCONI'S ANAEMIA. Med J Aust 1: 797-799. [Crossref]
40. Llistosella E, Moreno A, de Moragas JM (1984) Dyskeratosis congenita with macular cutaneous amyloid deposits. Arch Dermatol 120: 1381-1382. [Crossref]
41. Hyodo M, Sadamoto A, Hinohira Y, Yumoto E (1999) Tongue cancer as a complication of dyskeratosis congenita in a woman. Am J Otolaryngol 20: 405-407. [Crossref]
42. Moretti S, Spallanzani A, Chiarugi A, Muscarella G, Battini ML (2000) Oral carcinoma in a young man: a case of dyskeratosis congenita. J Eur Acad Dermatol Venereol 14: 123-125. [Crossref]
43. Loh HS, Koh ML, Giam YC (1987) Dyskeratosis congenita in two male cousins. Br J Oral Maxillofac Surg 25: 492-499. [Crossref]
44. Steier W, Van Voolen GA, Selmanowitz VJ (1972) Dyskeratosis congenita: relationship to Fanconi's anemia. Blood 39: 510-521. [Crossref]
45. De Boeck K, Degreef H, Verwilghen R, Corbeel L, Van Daele MC (1981) Thrombocytopenia: first symptom in a patient with dyskeratosis congenita. Pediatrics 67: 898-903. [Crossref]
46. Jacobs P, Saxe N, Gordon W, Nelson M (1984) Dyskeratosis congenita. Haematologic, cytogenetic, and dermatologic studies. Scand J Haematol 32: 461-468. [Crossref]
47. Kuo CY, Lin DT, Tuu WM, Chen BW, Lin KH et al. (1989) Dyskeratosis congenita preceded by severe aplastic anemia: report of one case. Zhonghua Min Guo Xiao Er Ke Yi Xue Hui Za Zhi 30: 337-341. [Crossref]
48. Lin TC, Lin CK, Lee JY, Chau WK, Wang SY (1989) X-linked dyskeratosis congenita with aplastic anemia--genetic and hematologic studies. Zhonghua Yi Xue Za Zhi (Taipei) 43: 57-62. [Crossref]
49. Forni GL, Melevendi C, Jappelli S, Rasore-Quartino A (1993) Dyskeratosis congenita: unusual presenting features within a kindred. Pediatr Hematol Oncol 10: 145-149. [Crossref]
50. Ivker RA, Woosley J, Resnick SD (1993) Dyskeratosis congenita or chronic graft-versus-host disease? A diagnostic dilemma in a child eight years after bone marrow transplantation for aplastic anemia. Pediatr Dermatol 10: 362-365. [Crossref]
51. Bryan HG, Nixon RK (1965) DYSKERATOSIS CONGENITA AND FAMILIAL PANCYTOPENIA. JAMA 192: 203-208. [Crossref]
52. Heiss NS, Knight SW, Vulliamy TJ, Klauck SM, Wiemann S et al. (1998) X-linked dyskeratosis congenita is caused by mutations in a highly conserved gene with putative nucleolar functions. Nat Genet 19: 32-38. [Crossref]
53. Marciniak RA, Johnson FB, Guarente L (2000) Dyskeratosis congenita, telomeres and human ageing. Trends Genet 16: 193-195. [Crossref]
54. Vulliamy T, Marrone A, Goldman F, Dearlove A, Bessler M et al. (2001) The RNA component of telomerase is mutated in autosomal dominant dyskeratosis congenita. Nature 413: 432-435. [Crossref]
55. Gonzalez Moles MA, Scully C, Gil Montoya JA (2008) Oral lichen planus: controversies surrounding malignant transformation. Oral Dis 14: 229-243. [Crossref]
56. Morita M, Asoda S, Tsunoda K, Soma T, Nakagawa T et al. (2017) The onset risk of carcinoma in patients continuing tacrolimus topical treatment for oral lichen planus: a case report. Odontology 105: 262-266. [Crossref]
57. Mattsson U, Magnusson B, Jontell M (2010) Squamous cell carcinoma in a patient with oral lichen planus treated with topical application of tacrolimus. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 110: e19-e25. [Crossref]
58. Waldron J, Stafford N (1989) Management of oral cancer / [edited by] Nicholas Stafford and John Waldron., ed. O.m. publications: Oxford; New York: Oxford University Press.
59. Schwender FT, Wollner I, Kunju LP, Nakhleh RE, Chan KM (2002) Squamous cell carcinoma of the buccal mucosa with metastases to the pericardial cavity, lung and thyroid. Oral Oncol 38: 114-116. [Crossref]
60. Chen AY, Myers JN (2000) Cancer of the oral cavity. Curr Probl Surg 37: 633-731. [Crossref]
61. Sciubba JJ (2001) Oral cancer and its detection. History-taking and the diagnostic phase of management. J Am Dent Assoc 132: 12s-18s. [Crossref]
62. Kerdpon D, Sriplung H (2001) Factors related to advanced stage oral squamous cell carcinoma in southern Thailand. Oral Oncol 37: 216-221. [Crossref]
63. Shah JP, NWJ, John G, Batsakis (2003) Dunitz Martin Ltd. 480., Oral Cancer. 1st edition.
64. Nickolakakos AP, OCRA (2007) 1st edition.
65. Ghoshal S, Mallick I, Panda N, Sharma SC (2006) Carcinoma of the buccal mucosa: analysis of clinical presentation, outcome and prognostic factors. Oral Oncol 42: 533-539. [Crossref]
66. Waldron, N.S.a.J., Management of Oral Cancer. 1st edition, ed. 1989.
67. Liao CT, Wang HM, Ng SH, Yen TC, Lee LY et al. (2006) Good tumor control and survivals of squamous cell carcinoma of buccal mucosa treated with radical surgery with or without neck dissection in Taiwan. Oral Oncol 42: 800-809. [Crossref]
68. Huang CH, Chu ST, Ger LP, Hou YY, Sun CP (2007) Clinicopathologic evaluation of prognostic factors for squamous cell carcinoma of the buccal mucosa. J Chin Med Assoc 70: 164-170. [Crossref]
69. Huang CH, Chu ST, Ger LP, Hou YY, Sun CP (2007) Clinicopathologic evaluation of prognostic factors for Squamous cell carcinoma of the Buccal Mucosa. J Chin Med Assoc 70:164-170. [Crossref]
70. Organization WH (2017) Health emergencies and humanitarian response update. Cited 2020.
71. Lin CS, Jen YM, Cheng MF, Lin YS, Su WF et al. (2006) Squamous cell carcinoma of the buccal mucosa: an aggressive cancer requiring multimodality treatment. Head Neck 28: 150-157. [Crossref]
72. Bloom ND, Spiro RH (1980) Carcinoma of the cheek mucosa. A retrospective analysis. Am J Surg 140: 556-559. [Crossref]
73. Conley J, Sadoyama JA (1973) Squamous cell cancer of the buccal mucosa. A review of 90 cases. Arch Otolaryngol 97: 330-333. [Crossref]
74. Lapeyre M, Peiffert D, Malissard L, Hoffstetter S, Pernot M (1995) An original technique of brachytherapy in the treatment of epidermoid carcinomas of the buccal mucosa. Int J Radiat Oncol Biol Phys 33: 447-454. [Crossref]
75. Pop LA, Eijkenboom WM, de Boer MF, de Jong PC, Knegt P et al. (1989) Evaluation of treatment results of squamous cell carcinoma of the buccal mucosa. Int J Radiat Oncol Biol Phys 16: 483-487. [Crossref]
76. Strome SE, To W, Strawderman M, Gersten K, Devaney KO et al. (1999) Squamous cell carcinoma of the buccal mucosa. Otolaryngol Head Neck Surg 120: 375-379. [Crossref]
77. Urist MM, O'Brien CJ, Soong SJ, Visscher DW, Maddox WA (1987) Squamous cell carcinoma of the buccal mucosa: analysis of prognostic factors. Am J Surg 154: 411-414. [Crossref]
78. Hakeem AH, Pradhan SA, Tubachi J, Kannan R (2013) Outcome of per oral wide excision of T1-2 N0 localized squamous cell cancer of the buccal mucosa--analysis of 156 cases. Laryngoscope 123: 177-180. [Crossref]
79. Sieczka E, Datta R, Singh A, Loree T, Rigual N et al. (2001) Cancer of the buccal mucosa: are margins and T-stage accurate predictors of local control? Am J Otolaryngol 22: 395-399. [Crossref]
80. Eisen D, Carrozzo M, Bagan Sebastian JV, Thongprasom K et al. (2005) Number V Oral lichen planus: clinical features and management. Oral Dis 11: 338-349. [Crossref]
81. Scully C, Carrozzo M (2008) Oral mucosal disease: Lichen planus. Br J Oral Maxillofac Surg 46: 15-21. [Crossref]
