Cavernous Hemangiomas of the Temporal Bone
A B S T R A C T
Objective: We report two cases of cavernous hemangioma of the temporal bone and review the relevant literature.
Design: Retrospective report of two cases and review of the English literature concerning cavernous hemangiomas of the ear via PubMed.
Results: A 46-year-old male was found to have a large external auditory canal vascular mass. Excision of mass with endaural approach was taken and diagnosis was confirmed with histopathology. Also, a 35-year-old female was found to have an internal auditory canal vascular mass. Retrosigmoid craniotomy was performed for resection and diagnosis was confirmed with histopathology. Less than 120 case reports of cavernous hemangiomas of the ear were identified and analyzed.
Conclusion: Cavernous hemangiomas of the ear are rare vascular tumors of the head and neck that can cause symptoms requiring histopathological diagnosis and surgical treatment.
Keywords
Cavernous hemangioma, temporal bone, vascular tumor, external ear, middle ear
Introduction
Hemangiomas are benign hamartomas of vasculature with variable proliferation indices. There are two main types of hemangiomas, and they are classified as either capillary or cavernous hemangiomas based on histology. Capillary hemangiomas exhibit clusters of capillary-like channels. Although capillary hemangiomas are the most common tumor of the orbit and periorbital areas in childhood, they may also occur in the skin, subcutaneous tissue, mucous membranes, liver, spleen, and kidneys [1]. Cavernous hemangiomas (CH) are also found in skin, mucous membranes, and internal organs, but consist primarily of large cavernous vascular spaces. Unlike capillary hemangiomas, CHs are seen more commonly in adolescents and adults and occur in deeper anatomical layers and structures [2]. CHs of the temporal bone are rare and reported mostly as individual cases in literature. They are generally delineated by the site of vascular pedicle and anatomical origin of the tumor [3]. The initial symptoms of CHs of the temporal bone are usually hearing loss and tinnitus, but patients can also display vertigo, imbalance, as well as facial palsy or spasm. Patients typically undergo high-resolution computed tomography (CT) and gadolinium-enhanced magnetic resonance imaging (MRI) to characterize the tumor, which will eventually guide surgical approach [3, 4]. Depending on the size and location of the tumor, the age and comorbidities of the patient, and severity of symptoms, treatment can range from wait-and-scan or surgical removal of the tumor [4]. We describe two interesting cases of vascular anomalies of the temporal bone and discuss the available literature for CHs of the ear.
Case Descriptions
Case 1
A 46 year old male with an extensive 15-year history of bilateral ear infections presented to the emergency department with a mass emerging from the right ear canal. He reported 3-weeks of right sided otorrhea and pulsating otalgia that did not improve with ciprofloxacin and dexamethasone otic drops. He also reported right ear pulsatile tinnitus and hearing loss as well as intermittent dizziness and imbalance. He denied fevers or facial weakness.
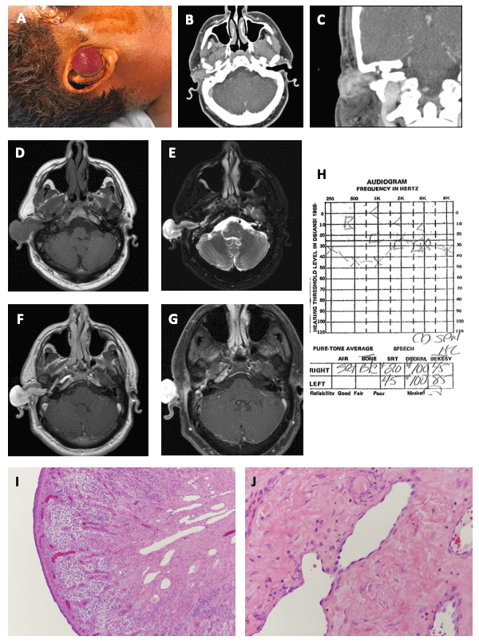
Figure 1: Presentation and work up of Cavernous Hemangioma in Case 1. Patient presented with a right, reddish spherical mass extending out of the external auditory canal (A). CT Temporal Bone with intravenous contrast shows an enhancing mass involving the right external auditory canal on axial (B) and coronal (C) sections. MRI Brain with and without contrast demonstrates a polypoid mass expanding the right external auditory canal, entering into the middle ear through the tympanic membrane, and originating from the antrum. The tumor is T1 isointense (D), T2 hyperintense (E), and enhances with gadolinium (F) on axial cuts. The tumor is seen originating in the mastoid antrum, near the lateral semicircular canal (G). The mass lacks a salt-and-pepper appearance on T1 and T2 images, which is an imaging characteristic typical of flow voids in paraganglioma tumors. Pre-operative audiogram shows a right-sided conductive hearing loss (H). Representative images of tumor sections stained with hematoxylin and eosin show a (I) surface epithelial lining and active granulation tissue underneath the lining (10X). Below the granulation tissue, dilated blood vessel proliferation is seen with dermal fibrosis and hyalinization. The tumor also demonstrates (J) dilated vessels with irregular lumen and flat endothelial lining. There are scant red blood cells seen inside the lumen as well as stromal fibrosis and hyalinization between blood vessels (40X).
On physical examination, the patient had a round, red-colored mass protruding out of the right external auditory canal (EAC) accompanied by white-yellow discharge (Figure 1A). The mass blanched with palpation. The right tympanic membrane (TM) was not visible due to occlusion of the EAC by the mass. The contralateral left ear was unremarkable, and the remainder of the head and neck was otherwise grossly normal.
The CT temporal bone (Figure 1B & 1C) showed a right-sided 2.4 cm x 3.8 cm x 2.1 cm heterogeneous enhancing soft tissue mass extending from the middle ear to the pinna with evidence of ossicular and scutal erosion. The otic capsule, carotid canal and jugular bulb appeared grossly intact. A follow-up MRI was also obtained (Figure 1D-1G), which demonstrated a T1-isointense, T2-hyperintense, and enhancing polypoidal mass expanding the right EAC and without significant flow voids. There was also invasion through the TM medially, extension along the middle ear space and into the eustachian tube. Bilateral mastoid effusions were also present with post obstructive changes on the right. Our differential diagnosis included infectious polyp, paraganglioma, hemangioma, or other vascular lesion.
A culture of the right EAC was performed and revealed Corynebacterium colonization. A outpatient trial of oral antibiotics was performed but failed to have significant improvements in symptoms. Thus, a superficial biopsy of the EAC mass was obtained showing granulation tissue with acute and chronic inflammation. An audiogram was performed and showed conductive hearing loss on the right side (Figure 1H). The patient was subsequently taken to the operating room for removal of this mass. Extensive debulking of the vascular mass was performed via an endaural approach with judicious use of bipolar cautery for hemostasis. The tumor was followed into the ear canal until the tympanic membrane could be visualized. The tumor was extending into the middle ear and antrum through an attic defect and there was associated attic cholesteatoma.
Hence, a retroauricular incision was also performed followed by a mastoidectomy and extended facial recess approach for exposure. The mastoid was poorly pneumatized with extensive granulation tissue. The incus and malleus were eroded and involved with the mass and cholesteatoma and therefore removed. The stapes superstructure was intact, and some minor granulation tissue was left at the oval window niche. The mass was traced to a vascular pedicle at the antrum involving the subarcuate artery as the dominant blood supply. There was brisk bleeding encountered upon resection of the tumor and its blood supply. Hemostasis was achieved with bipolar cautery and drilling with a diamond bur.
The scuttal and TM defects were repaired with conchal cartilage and temporalis fascia graft, respectively. The final pathology of the tumor was consistent with cavernous hemangioma with surface ulceration (Figure 1I & 1J). Specifically, below the surface epithelial lining of the tumor, there was active granulation tissue and dilated blood vessels with irregular lumens and flattening of the endothelial lining. Dermal and stromal fibrosis along with hyalinization were also observed. The patient was seen 10 days after surgery and endorsed no pain, fever, or discharge, and intact facial nerve function.
Case 2
A 35-year-old previously healthy female presented with a 4-month history of left-sided hemifacial spasms, ear pain, and facial weakness. An MRI was performed at an outside facility and was reported as unremarkable. She was then treated at an outside facility with carbamazepine for presumed vascular compression of the facial nerve as well as Botox injections for hemifacial spasms. There was no significant improvement of symptoms with these therapies. The patient also reported constant non-pulsatile left sided tinnitus and mild imbalance without subjective hearing loss. On physical examination, she was found to have facial asymmetry with House-Brackmann (HB) grade 2 facial function on the left side. Otomicroscopy and the remainder of the cranial nerve and head and neck examination were unremarkable.
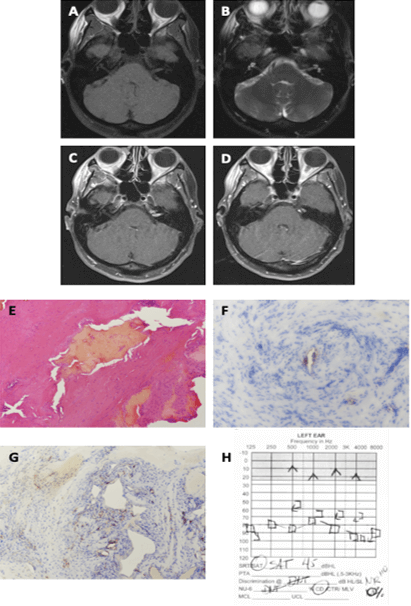
Figure 2: Presentation and work up of Cavernous Hemangioma in Case 2. MRI of the Internal Auditory Canal (IAC) and Posterior Fossa with and without gadolinium shows a mass within the left IAC that is T1 hypo-to-isointense (A) and T2-hyperintense (B) on axial cuts. The IAC mass also enhances with gadolinium (C). There are two additional tumor components, one within the petrous apex bone and the other located just anterior to the porous acousticus and within the cerebellopontine angle (D). The pathology of the tumor shows (E) stromal proliferation with irregularly shaped vascular channels. The tumor also stains with brown chromogenic reagent for blood vessel markers (F) CD31 and (G) CD34 (40X). The post-operative audiogram shows non-serviceable sensorineural hearing loss in the left ear with 0%-word discrimination (H).
Pre-operative audiogram showed normal hearing bilaterally. An MRI of the internal auditory canals (IAC) and posterior fossa (Figure 2A-2D) showed an enhancing 0.9cm mass at the left IAC that was T1 hypo-to-isointense and T2-hyperintense, suggestive of vestibular schwannoma or vascular loop. In addition, there was a separate enhancing mass with similar MRI characteristics along the posterior petrous bone, within the left cerebellopontine angle (CPA), just inferior to the cisternal segment of the left trigeminal nerve. The lesion also involved the petrous apex, and due to its location and appearance, meningioma was suspected.
The patient was taken to the operating room for a left retrosigmoid craniotomy for resection of CPA and IAC lesions using intraoperative auditory brainstem response (ABR) monitoring for hearing preservation. A large PICA-AICA loop was noted abutting the CN VII/VIII complex at the level of the porous acousticus; however, the vascular loop did not directly touch or compress the facial nerve. The root entry zone of the facial nerve was clear of any vascular loops and no gross tumor could be appreciated in the CPA. Following drilling of the posterior petrous bone and opening of the IAC dura, no tumor could be immediately identified along the superior and inferior vestibular nerves. However, after inferior displacement of the CN VII/VIII complex a vascular tumor could be visualized extending along the anterior wall of the IAC. The mass appeared to be originating from the temporal bone at the anterior IAC and petrous ridge, which required further drilling for removal of the mass. The mass was fully resected along with a skip lesion on the anterior surface of the porous acousticus, which was separate from the IAC lesion.
Although the facial nerve appeared intact at the conclusion of resection, it did not stimulate well proximally and 0.1 mAmp was required at the root entry zone for stimulation. Despite a growing delay on the ABR waveforms throughout the case, a reproducible wave was achieved after closure. Papaverine was used throughout the case. Pathology of the vascular mass was consistent with cavernous hemangioma and showed stromal proliferation with dilated vascular channels that were irregularly shaped. The vascular structures of the mass were positive for endothelial markers CD31 (also known as platelet endothelial cell adhesion molecule 1 [PECAM1]) and CD34 (a transmembrane glycoprotein expressed extensively on blood vessels) (Figure 2E-2G). The basement membrane of the vascular channels expressed type IV collagen, and the smooth muscle around the vessels show alpha-smooth muscle actin (SMA), consistent with CH. The tumor was negative for schwannoma markers, Sry-related HMg-Box gene (SOX-10), S-100, epithelial membrane antigen (EMA), and neural cell adhesion molecule (NCAM or CD56). Furthermore, the tumor did not express typical meningioma markers, such as progesterone receptor (PR) and somatostatin receptor (SSRT2).
Post-operatively, she displayed a HB grade 3 facial function, which rapidly improved at the 3-month follow-up. Postoperative audiometry showed non-serviceable hearing (Figure 2H), which did not recover with steroids or time. She had complete resolution of her hemifacial spasms with no abnormalities in other cranial nerves.
Discussion
CHs are common in the head and neck, with 60% of cavernous hemangiomas in children found in the region. They more typically involve the skin or brain [5]. Yet, cases of vascular lesions, including CHs and vascular malformations, of the temporal bone are more rare, comprising less than 1% of the tumors in the area [6, 7]. A review of English literature via PubMed (using search “cavernous hemangioma” and “ear” or “temporal bone” or “middle ear” or “tympanic membrane” or “internal auditory canal” or “external auditory canal” or “cerebellopontine angle”) identified less than 120 cases of cavernous hemangioma of the ear [4, 8-10]. Reports of these cases have generally characterized the tumor by the location occupied by the lesion (e.g. EAC, middle ear, mastoid, IAC, CPA), which gives insight on the symptoms patients with CH may demonstrate [1, 9-14]. Generally, patients with CHs of the temporal bone and ear present with hearing loss and tinnitus. Patients with IAC CH have shown symptoms of vertigo and facial spasm or weakness [10, 14]. EAC and TM CH may also present in patients as bloody otorrhea, aural pressure, and otalgia [9] (Table 1). CPA CH have been noted to cause symptoms disproportionate to their size, with profound hearing loss and facial nerve weakness [13].
Table 1: Summary of 2 Patients with Cavernous Hemangioma of the Temporal Bone.
|
Case No. |
Age (yrs) |
Sex |
Main Symptoms |
Duration of Symptoms (months) |
Surgical Approach |
Preop HB |
Postop HB |
FU HB |
Intraop Findings |
Postop Results |
|
1 |
46 |
M |
Drainage, pain, SNHL |
2 |
Endaural, Postauricular |
I |
I |
I |
FN partially dehisced, stimulated well |
No resulting symptoms |
|
2 |
35 |
F |
Hemifacial spasms, pain |
4 |
Retrosigmoid |
II |
VI |
III |
FN intact, but decreased stimulation |
Deaf |
Although our second case presentation had a skip lesion, clinical clues that may aid CH diagnosis include a solitary, unilateral vascular lesion in an adolescent or adult that has not shown regression. Other lesions that can potentially mimic a CH of the temporal bone include glomus tumor, attic cholesteatoma with aural polyp, arteriovenous malformation (AVM), granulation tissue, endolymphatic sac tumor, and metastatic tumor [4, 15]. Imaging can help narrow or broaden the differential diagnosis and thus confirmatory diagnosis often relies on histopathology. CT with and without contrast may help delineate tumor boundaries or determine whether adjacent tissues or structures are involved. On MRI, CHs are commonly seen as enhancing lesions that are T1-isointense and T2-hyperintense [16, 17], which is a pattern that can be seen in various tumors, including schwannomas and meningiomas. When the diagnosis is unclear and the size and location of the tumor allow, biopsy can be performed prior to or in conjunction with surgical excision to confirm the vascular nature of the tumor and rule out malignancy [2].
CHs sit under a larger umbrella term of vascular malformations and are sometimes difficult to differentiate from other specific vascular lesions even with histopathology. In general, CHs show thin-walled blood vessels lined by normal-appearing endothelial cells and contain red blood cells. They are distinct from capillary hemangiomas by larger diameter, blood-filled sinusoidal locules known as “caverns” [18]. Histopathologic differential diagnosis may include arteriovenous malformation (AVM), angiosarcoma, vasculitides, and Kaposi sarcoma [2]. As such, immunostaining is used in histopathological definition of vascular lesions, particularly if morphology is less than definitive. The vessels within hemangiomas should stain positive for normal blod vessel markers of the endothelial, basement membrane, and smooth muscle layers. Endothelial markers include CD31, CD34, von Willebrand factor (VWF), Ulex eurpaeus lectin I, friend leukemia virus integration 1 (Fli-1), endothelial transcription factor ETS-related gene (ERG), and vascular endothelial cadherin [19]. Blood vessel basement membrane contains mainly type IV collagen and laminin, and the smooth muscle of blood vessels contains SMA, which can be used to aid in CH diagnosis [20]. CHs can be differentiated from schwannomas by staining negatively for S-100 and SOX-10, which are well-known Schwann cell markers [21]. SOX-10 and S-100 can also be used to differentiate CHs from melanoma [22]. Some known markers can be used to rule out other primary benign or metastatic possibilities in the differential diagnoses. For example, negative staining for EMA and SSRT2 can help rule out meningioma [23]. If the tumor is positive for PR or thyroid transcription factor-1 (TTF-1), metastatic breast and lung cancer should be considered, respectively [24].
Treatment of CHs of the temporal bone is primarily surgical. While en bloc resection of CHs is curative, it may lead to serious residual effects of facial weakness and deafness. Resection of large CHs that involve the temporal bone are more difficult to perform en bloc due to the complex bony anatomy of the temporal bone and critical neurovascular structures close by. Although not ideal, piecemeal resection of large CHs may be necessary to eradicate these tumors and also preserve inner ear structures for balance and hearing, cranial nerves, and adjacent arteries and veins. Cauterizing the vascular pedicle early on may prevent significant bleeding in both en bloc and piecemeal resections. One case of recurrence of CH due to inadequate excision has been reported [25].
The surgical approach that optimizes tumor exposure and minimizes the post-operative side effect profile should be determined based on the patient’s clinical presentation and the location and size of the tumor. Retrosigmoid and translabyrinthine approaches have been described for surgical excision of IAC and CPA CHs [13]. Because translabyrinthine approaches sacrifice hearing, this approach is generally reserved for patients with unserviceable hearing loss. Depending on the size and location, EAC and TM CHs have been managed with transcanal, endaural, or postauricular excisions. Small TM CHs can be excised with a transcanal or endaural approach, while larger lesions may require a formal postauricular approach [9].
The exact mechanisms by which cavernous hemangiomas arise is unknown. Theories suggest that these tumors arise due in part to abnormal proliferation of endothelial cells expression of placental vascular epitopes angiogenesis, and/or vasodilatation of pre-existing blood vessels [26-29]. Certain factors have been implicated in these processes, including basic fibroblast growth factor (bFGF) and vascular endothelial growth factor (VEGF), though the exact role of these factors has yet to be elucidated. bFGF is a potent angiogenic factor released by mast cells that enhances proliferation and migration of fibroblasts. It also regulates the expression of fibronectin and collagen, which are important for formation of new vascular channels. bFGF levels have been shown to be elevated in serum and urine of infants with proliferating hemangiomas [30]. VEGF is another potent angiogenic factor that is involved in vascular proliferation and permeability of membranes. VEGF and its receptor flk-1 have also been shown to be expressed in orbital cavernous hemangioma samples [31]. Propranolol, a non-cardioselective beta-blocker used to treat infantile hemangioma, is thought to exert its effects through bFGF and VEGF pathways [32]. Although propranolol has not been used traditionally for CHs, isolated reports show significant reductions in the size of CH after a few months of use [33, 34]. Use of propranolol for CH may be particularly beneficial in patients with comorbid conditions that cannot undergo surgery or when surgical resection will cause cranial nerve palsies or irreversible hearing loss.
There is also some evidence for genetic causes of hemangiomas. While CCM1, CCM2, and CCM3 genes have been studied extensively for their role in autosomal dominant cerebral vascular pathology no genes have been studied specifically for CHs of the temporal bone and ear [18, 35]. One study attempted to create an animal model of infantile hemangioma and showed that transgenic mice with SV40-late-promoter-driven Polyomavirus Middle T oncogene developed hemangioma-like structures in multiple sites, including ear mucosa. Sections of hemangioma tissue from this model stained negative for Ki-67, indicating the tumors were hemangioma rather than angiosarcomas [36].
Conclusion
CH of the temporal bone have been described in recent literature, though little is known about the pathogenesis of these lesions. Although clinical signs and imaging are helpful, definitive diagnosis of CHs of the temporal bone relies on histopathological characteristics. Treatment of these lesions is centered around surgical resection, which requires detailed pre-surgical planning to avoid injury to adjacent neurovascular and inner ear structures. Propranolol may have utility in controlling CHs of the temporal bone while minimizing surgical risks; however further investigation is warranted. In this case series, we describe two tumors that involve the temporal bone to raise awareness of the possibility of CH in the differential diagnosis and methods for management.
Disclosure
Simon Angeli is a consultant for Medtronic and Advanced Bionics; Jacques Morcos owns stock in Cogent. No disclosures for all other authors.
Article Info
Article Type
Case ReportPublication history
Received: Sat 28, Dec 2019Accepted: Mon 13, Jan 2020
Published: Tue 28, Jan 2020
Copyright
© 2023 Christine Dinh. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.AJSCR.2020.01.01
Author Info
Enrique Perez Carmen Gomez-Fernandez Christine Mei Christine Dinh Harold Essenfeld Jacques Morcos Rita Bhatia Simon Angeli Stefania Goncalves
Corresponding Author
Christine DinhDepartment of Otolaryngology, University of Miami Miller School of Medicine, Miami
Figures & Tables
Table 1: Summary of 2 Patients with Cavernous Hemangioma of the Temporal Bone.
|
Case No. |
Age (yrs) |
Sex |
Main Symptoms |
Duration of Symptoms (months) |
Surgical Approach |
Preop HB |
Postop HB |
FU HB |
Intraop Findings |
Postop Results |
|
1 |
46 |
M |
Drainage, pain, SNHL |
2 |
Endaural, Postauricular |
I |
I |
I |
FN partially dehisced, stimulated well |
No resulting symptoms |
|
2 |
35 |
F |
Hemifacial spasms, pain |
4 |
Retrosigmoid |
II |
VI |
III |
FN intact, but decreased stimulation |
Deaf |
References
- Reeck JB, Yen TL, Szmit A, Cheung SW (2002) Cavernous hemangioma of the external ear canal. Laryngoscope 112: 1750-1752. [Crossref]
- Kita AE, Long JL (2016) Hemangioma. Ear Nose Throat J 95: 19-20.
- Yang Y, Guan J, Ma W, Li Y, Xing B et al. (2016) Primary Intraosseous Cavernous Hemangioma in the Skull. Medicine (Baltimore) 95: e3069. [Crossref]
- Odat H, Al-Qudah M, Al-Qudah MA (2016) Cavernous Hemangioma of the External Canal, Tympanic Membrane, and Middle Ear Cleft: A Case Report. Head Neck Pathol 10: 261-264. [Crossref]
- Zheng JW, Zhou Q, Yang XJ, Wang Ya, Fan XD et al. (2010) Treatment guideline for hemangiomas and vascular malformations of the head and neck. Head Neck 32: 1088-1098. [Crossref]
- Hecht DA, Jackson CG, Grundfast KM (2001) Management of middle ear hemangiomas. Am J Otolaryngol 22: 362-366. [Crossref]
- Mangham CA, Carberry JN, Brackmann DE (1981) Management of intratemporal vascular tumors. Laryngoscope 91: 867-876. [Crossref]
- Magliulo G, Parrotto D, Sardella B, Della Rocca C, Re M (2007) Cavernous hemangioma of the tympanic membrane and external ear canal. Am J Otolaryngol 28: 180-183. [Crossref]
- Martines F, Bentivegna D, Maira E, Marasa S, Ferrara S (2012) Cavernous haemangioma of the external auditory canal: clinical case and review of the literature. Acta Otorhinolaryngol Ital 32: 54-57. [Crossref]
- Zhu WD, Huang Q, Li XY, Chen HS, Wang ZY et al. (2016) Diagnosis and treatment of cavernous hemangioma of the internal auditory canal. J Neurosurg 124: 639-646. [Crossref]
- Yeo A, Majithia A, Kalan A (2008) Haemangioma or vascular malformation of the tympanic membrane? a case report and review of literature. Indian J Otolaryngol Head Neck Surg 60: 59-61. [Crossref]
- Watson G, McSorley A, Kaushik V (2016) Diagnosis and management of a tympanic membrane hemangioma. Ear Nose Throat J 95: E12-E16. [Crossref]
- Oldenburg MS, Carlson ML, Van Abel KM, Giannini C, Jacob J et al. (2015) Cavernous hemangiomas of the internal auditory canal and cerebellopontine angle. Otol Neurotol 36: e30-e34. [Crossref]
- Mastronardi L, Carpineta E, Cacciotti G, Di Scipio E, Roperto R (2016) Cavernous hemangioma of the internal auditory canal encasing the VII and VIII cranial nerve complexes: case report and review of the literature. Neurosurg Rev 39: 349-354. [Crossref]
- Yang TH, Chiang YC, Chao PZ, Lee FP (2006) Cavernous hemangioma of the bony external auditory canal. Otolaryngol Head Neck Surg 134: 890-891. [Crossref]
- Nair SC (2018) Vascular Anomalies of the Head and Neck Region. J Maxillofac Oral Surg 17: 1-12. [Crossref]
- Dubois J, Rypens F (2015) Diagnostics of Infantile Hemangiomas Including Visceral Hemangiomas. In: Mattassi R, Loose DA, Vaghi M, editors. Hemangiomas and vascular malformations: an atlas of diagnosis and treatment. Springer: 81-88.
- Cox EM, Bambakidis NC, Cohen ML (2017) Pathology of cavernous malformations. Handb Clin Neurol 143: 267-277. [Crossref]
- North PE (2018) Classification and Pathology of Congenital and Perinatal Vascular Anomalies of the Head and Neck. Otolaryngol Clin North Am 51: 1-39. [Crossref]
- Hoya K, Asai A, Sasaki T, Kimura K, Kirino T (2001) Expression of smooth muscle proteins in cavernous and arteriovenous malformations. Acta Neuropathol 102: 257-263. [Crossref]
- Rodriguez FJ, Folpe AL, Giannini C, Perry A (2012) Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol 123: 295-319. [Crossref]
- Yeh I, McCalmont TH (2011) Distinguishing neurofibroma from desmoplastic melanoma: the value of the CD34 fingerprint. J Cutan Pathol 38: 625-630. [Crossref]
- Boulagnon-Rombi C, Fleury C, Fichel C, Lefour S, Marchal Bressenot A et al. (2017) Immunohistochemical Approach to the Differential Diagnosis of Meningiomas and Their Mimics. J Neuropathol Exp Neurol 76: 289-298. [Crossref]
- Pekmezci M, Perry A (2013) Neuropathology of brain metastases. Surg Neurol Int 4: S245-S255. [Crossref]
- Jackson CG, Levine SC, McKennan KX (1990) Recurrent hemangioma of the external auditory canal. Am J Otol 11: 117-118. [Crossref]
- Marchuk DA (2001) Pathogenesis of hemangioma. J Clin Invest 107: 665-666. [Crossref]
- Boye E, Yu Y, Paranya G, Mulliken JB, Olsen BR et al. (2001) Clonality and altered behavior of endothelial cells from hemangiomas. J Clin Invest 107: 745-752. [Crossref]
- North PE, Waner M, Mizeracki A, Mrak RE, Nicholas R et al. (2001) A unique microvascular phenotype shared by juvenile hemangiomas and human placenta. Arch Dermatol 137: 559-570. [Crossref]
- Matsumoto N, Tsuchiya M, Nomoto S, Matsue Y, Nishikawa Y et al. (2015) CD105 expression in oral capillary hemangiomas and cavernous hemangiomas. J Oral Sci 57: 45-53. [Crossref]
- Dosquet C, Coudert MC, Wassef M, Enjolras O, Drouet L (1998) Importance of bFGF ("basic fibroblast growth factor") for diagnosis and treatment of hemangiomas. Ann Dermatol Venereol 125: 313-316. [Crossref]
- Nagasaka M, Naganuma H, Satoh E (2007) Growth potential of orbital cavernous hemangioma suggested by vascular endothelial growth factor and its receptor flk-1. Neurol Med Chir (Tokyo) 47: 5-10. [Crossref]
- Rotter A, de Oliveira ZNP (2017) Infantile hemangioma: pathogenesis and mechanisms of action of propranolol. J Dtsch Dermatol Ges 15: 1185-1190. [Crossref]
- Albuquerque JC, Magalhaes RA, Felix JA, Bastos MV, Fontenele JB et al. (2014) Treatment of children and adolescents with hemangioma using propranolol: preliminary results from a retrospective study. Sao Paulo Med J 132: 48-54. [Crossref]
- Ramdhani D, Seedat RY (2018) Recurrent parotid cavernous haemangioma in an adolescent, successfully treated with Propranolol. Eur Ann Otorhinolaryngol Head Neck Dis 135: S117-S119. [Crossref]
- Choquet H, Pawlikowska L, Lawton MT, Kim H (2015) Genetics of cerebral cavernous malformations: current status and future prospects. J Neurosurg Sci 59: 211-220. [Crossref]
- Xu Q, Chen W, Wang Z, Zheng J, Zhang Z (2009) Mice transgenic with SV40-late-promoter-driven Polyomavirus Middle T oncogene exclusively develop hemangiomas. Transgenic Res 18: 399-406. [Crossref]
