Biochemical and Molecular Diagnostic Indicators for Cancer
A B S T R A C T
Biomarkers are substances that are either secreted by the tumor or produced by the body in response to the presence of cancer. Biomarkers serve as an objective measure for evaluation of normal and pathological processes as well as pharmacological responses to a therapeutic intervention. Cancer studies are usually difficult to interpret, especially based on the contemporary medical diagnosis. In this circumstance, biomarkers are developing as reliable diagnostic metabolites, which have many promising applications in oncological screening, differential diagnosis, risk assessment, response to treatment, and examining the progression of disease. Genome or protein based prognostic biomarkers are available, for numerous cancer types, for potential inclusion into clinical prognostic staging methods. However, there lies difficulty in translating these biomarkers into clinical outcomes. This review concerns important biomarkers related to wide varieties of cancer and also elucidates mode of action of few major biomarkers.
Keywords
Cancer, biomarkers, apoptosis, hypoxia, angiogenesis
Introduction
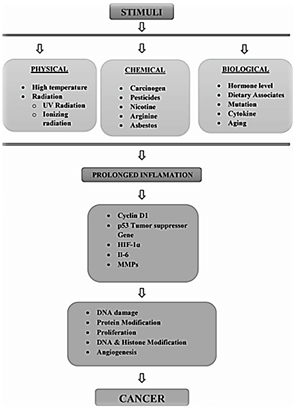
Cancer constitutes an immense weight on society in both progressively and less monetarily created nations alike. The disease is spreading as a result of population growth and an increasing percentage of risk factors such as smoking, being overweight, being physically inactive, and changing regenerative examples associated with urbanization and financial advancement. Globally, 14.1 million new illness cases and 8.2 million fatalities are expected in 2022 [1]. Because cancer is an aberrant type of tissue development in which cells divide uncontrollably, usually in a self-sustaining form, resulting in a dynamic increase in the number of dividing cells, novel treatments and screening technology are critical (Figure 1) [2].
A biomarker is an equitably estimated chemical molecule that portrays an ordinary or unusual natural state in a living being by breaking down biomolecules, for example, DNA, RNA, protein, peptide, and biomolecule create changes (Tables 1 & 2). According to the World Health Organization, a biomarker is any chemical, structure, or process that can be assessed in the body or its items and influences or predicts the occurrence of a result or illness. All the more particularly as far as clinical utility, a cancer biomarker may quantify the danger of creating growth in a particular tissue or, then again, may indicate danger of disease movement or potential reaction to therapy [3, 4].
Biomarkers have a wide range of potential uses in oncology, including risk assessment, screening, differential conclusion, visualization assurance, therapy response prediction, and tracking the progression of illness. Because biomarkers play such an important role in all stages of disease, it's critical that they go through a rigorous evaluation, which includes informational approval, clinical approval, and clinical utility evaluation, before being integrated into normal clinical care [5, 6]. Biomarker for cancer is allocated into five groups. They are,
i. Proliferation of cells in the cell cycle
ii. Apoptosis and tumor suppression
iii. Hypoxia
iv. Angiogenesis
v. Matrix degradation and cell adhesion
Figure 1: Types of stimuli that induce tumor.
Table 1: Detection and
quantification of the biomarkers.
|
SAMPLE |
BIOMARKERS |
DETECTION & QUANTIFICATION METHODS |
|
Serum / Plasma / Saliva / Urine |
·
IL-6 ·
Matrix
Metalloproteases (MMPs) ·
Cyclin
D1 ·
HIF
1α ·
p53 tumor suppressor gene |
·
Antibody
based technique ·
Mass
spectroscopy ·
ELISA ·
Western
Blotting Technique ·
Fluorescence
Techniques |
Table 2: Expression of
biomarkers in human cancer.
|
BIOMARKERS EXPRESSED |
TYPES OF CANCER |
|
Cyclin D1 |
Breast cancer Oral cancer Lung cancer Bladder cancer Liver cancer Melanoma cancer Mantle cell lymphoma |
|
p53
Tumor Suppressor Gene |
Ovarian cancer Liver cancer Prostate cancer Bladder cancer adenosarcoma |
|
HIF-1α |
Brain cancer Bladder cancer Colon cancer Ovarian cancer Pancreatic cancer Renal cancer Cervical cancer |
|
IL-6 |
Gastric cancer Head & neck cancer Colon cancer Renal cancer Lung cancer Breast cancer |
|
MMPs |
Prostate cancer Squamous cancer Colorectal cancer Pancreatic cancer Brain cancer Bladder cancer |
Uses of Biomarkers
i. To recognize the disease
ii. To determine the prognostic condition of the patient
iii. To predict the effectiveness of the treatment.
I Cell Proliferation Marker
i Cyclin D1
The CCND1 gene produces Cyclin D1, which is now considered a well-known human oncogene [7]. Cyclin D1 is a transcriptional co-regulator and the most significant regulator of cell cycle progression [8]. In cancer cases, the gene and its product have been intensively researched. This could be useful as a biomarker for breast, lung, colon, bladder, and liver cancers, as well as melanoma, oral squamous cell carcinomas, and mantle cell lymphoma [9]. Overexpression of cyclin D1 has been associated to cancer formation and progression. Deregulated cyclin D1 breakdown appears to be the cause of increased cyclin D1 levels in many cancers [10].
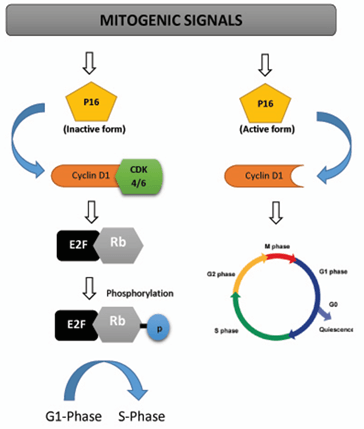
Figure 2: Mechanism and action of cyclin D1 in tumor cell cycle.
Mechanism
It forms active complexes (Figure 2) with cyclin dependent kinases 4 and 6 (CDK4 and CDK6), which phosphorylate and inactivate the retinoblastoma protein to enhance cell cycle progression (RB). According to new study, cyclin D1 regulates the activity of various transcription factors, including histone deacetylase 3 (HDAC3), which acts as a transcriptional modulator. These actions do not necessitate CDK4 activation. The cyclin D1 protein has a short half-life (24 minutes) and is largely degraded by the 26S proteasome in an ubiquitin-dependent manner [11]. Overexpression of cyclin D1 has also been linked to the development of endocrine resistance in breast cancer cells. Overexpression of cyclin D1 is prevalent in cancer, but it isn't always due to gene amplification. Instead, high amounts of cyclin D1 frequently lead to improper post-translational regulation [12].
II Hypoxic Marker
i HIF-1 Alpha
In 1991, Semenza et al. found HIF-1 [13]. They observed that binding of a particular protein produced under hypoxic conditions to the HRE triggers transcriptional activation of numerous regulatory genes. HIF-1 was later discovered to be the protein in question. HIF-1 (or its analogues HIF-2 and HIF-3) is a heterodimeric transcription factor made up of two subunits: HIF-1 (or its analogues HIF-2 and HIF-3), and HIF-1 (or its analogues HIF-2 and HIF-3) (or its analogues HIF-2 and HIF-3). In hypoxic settings, the expression of HIF-1, an oxygen-sensitive subunit, increases. HIF-1, on the other hand, has a constitutive expression pattern. HIF-1 is also known as aryl hydrocarbon nuclear translocator (ARNT) because it was found before HIF-1 and was identified as a heterodimeric partner of aryl hydrocarbon receptor (AhR). HIF-1 binds to AhR, making it easier for it to reach the nucleus. These two components belong to the bHLH-PAS protein family because their structures are similar to two nuclear proteins found in Drosophila (Per and Sim, PAS) that comprise a basic-helix-loop-helix (bHLH) motif. Identifiable domains (b, HLH, PAS, and TAD) distinguish the bHLH proteins, which can control their own transcription as well as the expression of other members of the family. The bHLH-PAS motifs are necessary for the formation of heterodimers between HIF-1 and HIF-1 subunits as well as binding to the HRE-DNA sequence on target genes in general. The ability of base domains to bind DNA was determined to be required for binding the HRE to the gene, whereas the HLH motif is where dimerization with other proteins takes place [14-17].
a Mechanism
Early transformed cancer cells can grow and form small tumors in the absence of neoangiogenesis. Oxygen and nutrients can seep from the host's ordinary tissue vessels over a distance of more than 200 metres. When a small tumor expands to a diameter of more than 400 m, especially in the tumor's center, a hypoxic environment is generated [18]. Hypoxia causes two basic reactions: a) necrosis of cells far from host tissue vessels; b) activation of HIF1 in surviving tumor cells that are closer to the vessels and have been sub fatally damaged; HIF1-driven gene expression allows them to survive and grow, increasing their commitment to malignancy [19]. Hundreds of genes are activated when HIF1 is activated. Many of them offer a reliable early sign of tumor progression. Neoangiogenesis and the ability to expand beyond the 400-m diameter limit are caused by VEGFs and their receptors; telomerase activation increases proliferative potential and the number of possible cycles; and the most well-known metabolic effects of this adaptation are changes in intermediate and energy metabolism [20, 21].
Necrosis is characterised by plasma membrane fragmentation and the release of intracellular molecules, some of which are alarmins or Damage-Associated Molecular Patterns (DAMPs). Alarmin receptors induced by hypoxia interact with released alarmins to trigger the expression of pro-inflammatory genes in a variety of cell types, including resident innate immunity cells or leukocytes, which commonly express a number of alarmin receptors in their plasma membrane, and tumor cells with alarmin receptors. Alarmin receptor signalling triggers NF-kB, which causes pro-inflammatory genes to be produced. A pro-inflammatory microenvironment may encourage tumor growth (Figure 3) [22-25].
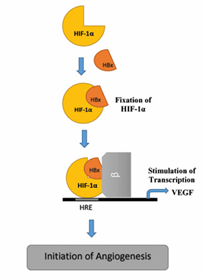
Figure 3: Role of HIF-1α in neoplastic cells.
b Role of HIF-1 Alpha
Investigating the functions of HIF1-activated genes reveals that a number of these genes or gene families are important in moving a transformed cell toward certain cancer markers. Overexpression of VEGFs and their receptors, in particular, stimulates tumor-specific neo-angiogenesis, allowing the early tumor to grow beyond the confines of basic oxygen and nutrition delivery (200-300 m). Telomerase (TERT) activation lengthens telomeres and increases proliferative potential, immortalising tumor cells. HIF1-dependent activation of proliferative genes including c-myc and cyclin D1 boosts proliferative ability even more. HIF1 also activates OCT4 and Notch, which promotes stem cell renewal and immortalization. Chemotherapy resistance is conferred by overexpression of ABC transporters. Overexpression of a number of key molecules, such as ALDA, PGK, and GLUT-1, elucidates the reprogramming of tumor energy metabolism [26-28].
The majority of invasion and metastasis genes are likewise regulated by HIF1 and NFkB. In a hypoxic environment, a range of cell types, including cancer and normal stem cells, express de novo or overexpress unique alarmin receptors such as CD45+ cell, RAGE, P2X7, TLRs, and others by activating the alarmins produced by necrotic cells. The activation of the nuclear factor kappa B (NFkB) is connected to a high degree of pro-inflammatory gene expression. This is the pivotal event that links hypoxia adaptation to the expression of hundreds of genes involved in the Inflammatory Reparative Response (IRR) and, most importantly, the development of malignant phenotypic characteristics. Genes or gene families that play a role in hypoxia adaptation and contribute to cancer progression [29-31].
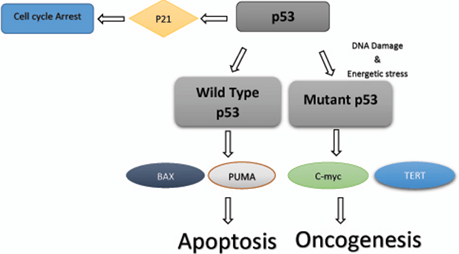
Figure 4: Mechanism of wild type & mutant p53 in cell cycle.
III Tumor Suppression Marker p53
The TP53 gene in humans genes for the tumor suppressor protein p53 (also known as protein 53 or tumor protein 53). p53 is required for multicellular animals because it regulates the cell cycle and hence works as a tumor suppressor, preventing cancer. As a result, p53 has earned the moniker “the genome's guardian,” referring to its role in preserving genomic integrity by preventing mutation. In humans, the TP53 gene on the short arm of chromosome 17 encodes p53. The TP53 gene, which codes for the protein p53, is one of the most frequently altered genes in human cancers. In around half of all cancers, p53 is thought to be inactive. A number of anticancer mechanisms are activated by p53 (Figure 4) [32-35].
However, the mechanism is interrupted in malignancies that maintain the wild-type p53 protein, leading in aberrant p53 inactivation and tumor suppressor response avoidance. Inactivation is caused by increased expression of the p53-negative regulators MDM2 and MDM4, as well as loss or epigenetic inactivation of the p53-positive regulator and MDM2 inhibitor ARF (Alternate Reading Frame tumor suppressor protein). The mutant protein has a dominant negative activity over the remaining wild-type allele in tumors with a mutated p53 protein, which is usually found in the DNA binding domain, resulting in diminished p53 transcriptional function. In addition, many p53 mutations acquire novel oncogenic features [36-38].
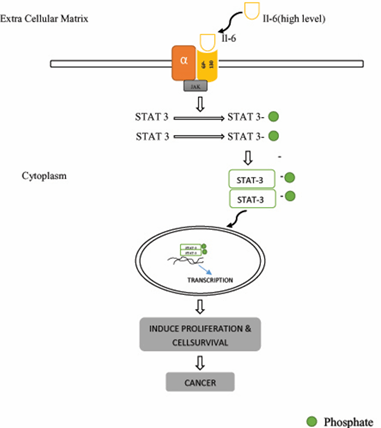
IV Angiogenesis Marker IL-6
Interleukin-6 (IL-6) is a multifunctional cytokine that influences immune function, metabolism, hematopoiesis, angiogenesis, and oncogenesis. IL-6 has both pro- and anti-inflammatory characteristics, depending on the context. Because circulating IL-6 levels often rise with inflammatory disorders like infection, diabetes, and obesity, it was formerly thought to be pro-inflammatory. On the other hand, transient IL-6 production after exercise may have anti-inflammatory and insulin-sensitizing properties. Despite its confusing biochemistry, IL-6 has been widely investigated for its function in the progression of cancer and cancer-associated cachexia. IL-6 affects these processes via affecting metabolism and endocrine function by targeting a variety of systems both directly and indirectly. IL-6 is required for VEGF production and angiogenesis in gastric cancer. VEGF is a very powerful angiogenic factor [39-42].
Mechanism
The membrane-bound receptor gp130 is used by the IL-6 family to communicate (glycoprotein 130). The JAK (Janus Kinase) tyrosine kinase is activated when the gp130 receptor binds. Tyrosines in gp130's cytoplasmic domain are phosphorylated when JAK-1 is activated, allowing proteins with Src homology domain-2 (SH2) and phosphotyrosine-binding proteins to join. These proteins include the STATs, which are required for IL-6 signalling. STAT proteins are activated when a single tyrosine residue is phosphorylated, resulting in the formation of STAT dimmers [43, 44]. STAT dimmers can then travel to the nucleus, where they collaborate with other factors to increase transcription of genes with STAT-responsive promoters. Although IL-6 can activate both STAT-1 and STAT-3, STAT-3 has been discovered to play a larger role in the IL-6 signalling pathway. STAT-3 transcriptional targets regulate a variety of physiological functions, including immunological function, cell proliferation and growth, differentiation, and possibly apoptosis. Because the gp130 receptor is found in practically every tissue in the body, IL-6 can have a wide range of effects, particularly in high-expression situations like cancer and cancer-related cachexia [45, 46].
One of the main cytokines found to be prominent in the serum samples of patients with head and neck cancer when compared to healthy controls or patients with non-threatening diseases is interleukin 6 (IL-6), which is capable of independently forecasting tumor metastasis through the regulation of cytokine and protease mediated complex networks. IL-6 has been demonstrated to stimulate tumor cell growth in a variety of tumor cell lines, including human cervical cancer. In addition to its traditional role in stimulating proliferation, IL-6 has been shown to stimulate hyper-methylation of the promoter via DNMT3b, leading to tumor metastasis in pancreatic ductal adenocarcinoma, demonstrating that IL-6 can induce epigenetic alterations. One of the most recent research, published in 2017, supports the existing data on IL-6-mediated hypermethylation [47]. The study concluded that IL-6-mediated inflammation caused hyper-methylation of LINE-1 regions in the oral cavity, resulting in chromosomal instability and cancer. Because IL-6 is one of the key mediators in the development of oral cancer, it has been linked to the early detection of premalignant lesions (Figure 5) [48-50].
Figure 5: Action of IL-6 in malignancy.
V Matrix Metalloproteases (MMPs)
Matrix metalloproteases are a multigene family of zinc-dependent endopeptidases with a comparable structure and the ability to degrade practically every extracellular matrix component collectively (ECM). Matrix metalloproteinases (MMPs) use their endopeptidase activity to destroy proteins in the extracellular matrix, and there are currently 23 MMPs known. The many types of MMPs include collagenases (MMP-1, -8, and -13), gelatinases (MMP-2 and -9), stromelysins (MMP-3, -10, and -11), matrilysins (MMP-7 and -26), enamelysin (MMP-20), membrane-bound MMPs (MMP-14 to -17, MMP-24 and -25), and others (MMP- 19, -21, -23, -27 and -28). Matrix metalloproteinases, which are up-regulated in almost every type of cancer, are involved in proliferation, invasion, epithelial-to-mesenchymal transition, metastasis, and angiogenesis. MMPs are usually associated to a poor patient prognosis. MMPs (MMP-2, -7, -9, -10, -11, -13, -14, and -15) have been implicated in the progression and metastasis of breast cancer (21-25). MMP-1, -2, -3, -7, -9, and -14 are particularly important in tumor invasion, metastasis, and angiogenesis. Cancer cell proliferation, tumor development, and epithelial-to-mesenchymal transition have all been linked to MMP-1 and 9. By ELISA, BC tissue has a greater concentration of MMP-1and -9 proteins than normal breast tissue [51-53].
MMPs in conjunction with a patient's clinical status can aid in predicting disease risk, diagnosis, and progression. MMP-9 serum levels, for example, do not appear to be associated to the risk of stroke, but the MMP-9 C (-1562) T polymorphism has been discovered to be highly linked to the risk of stroke in both type 2 diabetes and non-diabetic patients. MMPs that are capable of predicting clinical status are still present. MMP-7, MMP-10, and MMP-12 overexpression in the sera of colon cancer patients has been connected to a bad prognosis, whereas a high serum MMP-1 level has been linked to a shorter overall survival in patients with non-small cell lung cancer. MMPs are sensitive and easy to detect, despite the fact that, due to their broad existence, they are not tissue specific. Pre-analytical conditions must be examined before initiating MMPs analysis in body fluids. While the amount of MMPs released during clotting may influence their quantity, several calcium-chelating anticoagulants may interfere with MMP function [54-56].
i Role of MMPs in Angiogenesis
MMPs facilitate angiogenesis by releasing ECM-bound proangiogenic substances and dissolving basement membrane and other ECM components, allowing endothelial cells to detach and move to new tissue (bFGF, VEGF, and TGF). Angiogenic chemicals generated by inflammatory or tumor cells, such as VEGF and bFGF (coloured triangles), bind to endothelial cells' appropriate receptors (Y-shaped receptors). Endothelial cells secrete MMPs, which alter the expression of integrins (T-shaped receptors) and cause them to proliferate. As the MMPs breakdown the ECM, additional matrix-bound VEGF and bFGF, as well as TGF, are released, and buried integrin-binding sites on the ECM fragments are exposed, integrin signalling is triggered, which potentiates growth factor receptor signalling. By cleaving cell-cell and cell-matrix receptors and removing adhesion sites, MMPs enhance endothelial cell movement. TGF causes endothelial cells to secrete MMP, which works as a chemoattractant for inflammatory cells, causing pericytes to flock to newly formed capillaries. Furthermore, MMP breakdown of ECM components produces fragments with newly accessible integrin binding sites, resulting in integrin intracellular signalling. MMPs can also make endogenous angiogenesis inhibitors from larger precursors: MMPs can make angiostatin by cleaving plasminogen; endostatin is the COOH terminal fragment of basement membrane collagen XVIII that can be made by cleavage by cathepsins and MMPs; and autocatalysis can make the hemopexin domain of MMP-2 from MMP-2. MMPs have both pro- and antiangiogenic characteristics as a result. MMPs are required for angiogenesis in general, and MMP-1 has been shown to inhibit it in animal models [57-59].
ii Role of MMPs in Cancer
MMP expression has been linked to advanced stage, invasive and metastatic properties, and a poor prognosis in general in almost every kind of human cancer. Early MMP synthesis, whether by tumor cells or surrounding stromal cells, contributes in the remodelling of the ECM and the release of ECM- and/or membrane bound growth factors, resulting in a favourable environment for the development of the primary tumor (Figure 1). The balance of proangiogenic factors (e.g., bFGF and VEGF) overcomes the synthesis of angiogenic inhibitors (perhaps due to hypoxia) as the tumor advances, resulting in an angiogenic switch (e.g., thrombospondins, angiostatin, and IFNs). Both MMP-2 and MMP-9 have been implicated in the induction of the angiogenic switch in several model systems [60, 61]. In additional tumor models, activating mutations of the protooncogenes K-ras and H-ras up-regulate VEGF expression while down-regulate thrombospondin expression, whereas oncogenic erbB2 signaling down-regulate. Thrombospondin is downregulated, and other proangiogenic factors are expressed. The angiogenic switch can occur extremely early in some cancers, even before malignant progression, due to increased artery density present in precancerous lesions. Upregulation of MMP synthesis, particularly gelatinases, which may degrade basement membrane components, allows tumor cells to penetrate the stroma and break down the basement membranes of capillaries and lymphatic arteries, allowing tumor cells to enter the circulation (intravasation). MMPs also aid cell migration by removing adhesion sites, exposing new binding sites, cleaving cell-cell or cell-matrix receptors, and releasing chemoattractants from the extracellular matrix, among other things (ECM). Similarly, to intravasation, MMPs are required for circulating tumor cells to escape blood vessels (extravasation), albeit this step does not appear to be rate limiting for metastasis formation. Local migration, the creation of a favourable environment for metastatic growth, and angiogenesis for long-term growth at the distant site all require MMPs. As a result, MMPs play a function in several stages of the carcinogenic process [62-64].
Conclusion
The current understanding of tumor initiation, movement, and metastasis pathways provides distinguishing proof of potential protein-based biomarkers, and their use in clinical practice is certain to not only plan a treatment to anticipate growth, but also to propel our understanding of carcinogenesis in this manner, allowing us to improve. Furthermore, the recognizable proof of protein-based biomarkers for tumor recognition is a great incentive because it allows for the early detection and conclusion of malignancy in a non-invasive manner. Nonetheless, there are still concerns such as a lack of institutionalization and others that need be addressed by additional research.
Article Info
Article Type
Review ArticlePublication history
Received: Mon 15, Feb 2021Accepted: Sat 02, Oct 2021
Published: Fri 15, Oct 2021
Copyright
© 2023 N. Baskaran. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.COR.2021.08.11
Author Info
N. Baskaran S Vignesh V Chandrasekar
Corresponding Author
N. BaskaranDepartment of Food Processing Business Incubation Center, Indian Institute of Food Processing Technology (IIFPT), Tamil Nadu, India
Figures & Tables
Table 1: Detection and
quantification of the biomarkers.
|
SAMPLE |
BIOMARKERS |
DETECTION & QUANTIFICATION METHODS |
|
Serum / Plasma / Saliva / Urine |
·
IL-6 ·
Matrix
Metalloproteases (MMPs) ·
Cyclin
D1 ·
HIF
1α ·
p53 tumor suppressor gene |
·
Antibody
based technique ·
Mass
spectroscopy ·
ELISA ·
Western
Blotting Technique ·
Fluorescence
Techniques |
Table 2: Expression of
biomarkers in human cancer.
|
BIOMARKERS EXPRESSED |
TYPES OF CANCER |
|
Cyclin D1 |
Breast cancer Oral cancer Lung cancer Bladder cancer Liver cancer Melanoma cancer Mantle cell lymphoma |
|
p53
Tumor Suppressor Gene |
Ovarian cancer Liver cancer Prostate cancer Bladder cancer adenosarcoma |
|
HIF-1α |
Brain cancer Bladder cancer Colon cancer Ovarian cancer Pancreatic cancer Renal cancer Cervical cancer |
|
IL-6 |
Gastric cancer Head & neck cancer Colon cancer Renal cancer Lung cancer Breast cancer |
|
MMPs |
Prostate cancer Squamous cancer Colorectal cancer Pancreatic cancer Brain cancer Bladder cancer |
References
1. Siegel RL, Miller
KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70: 7-30. [Crossref]
2. Kendall EJ, Barnett
MG, Praznik KC (2013) Automatic detection of anomalies in screening mammograms.
BMC Med Imaging 13: 43. [Crossref]
3. Chang L, Chang M,
Chang HM, Chang F (2018) Microsatellite Instability: A Predictive Biomarker for
Cancer Immunotherapy. Appl Immunohistochem Mol Morphol 26: e15-e21. [Crossref]
4. Hristova VA, Chan
DW (2019) Cancer biomarker discovery and translation: proteomics and beyond. Expert
Rev Proteomics 16: 93-103. [Crossref]
5. Wu L, Qu X (2015)
Cancer biomarker detection: recent achievements and challenges. Chem Soc Rev
44: 2963-2997. [Crossref]
6. Lu M, Zhang J,
Zhang L (2017) Emerging Concepts and Methodologies in Cancer Biomarker
Discovery. Crit Rev Oncog 22: 371-388. [Crossref]
7. John RR, Malathi N,
Ravindran C, Anandan S (2017) Mini review: Multifaceted role played by cyclin
D1 in tumor behavior. Indian J Dent Res 28: 187-192. [Crossref]
8. Fu XF, Yao K, Du X, Li Y, Yang XY et al. (2016) PGC-1α regulates the cell
cycle through ATP and ROS in CH1 cells. J Zhejiang Univ Sci B 17:
136-146. [Crossref]
9. Huang J, Ji EH, Zhao X, Cui L, Misuno K et al. (2019) Sox11
promotes head and neck cancer progression via the regulation of SDCCAG8. J
Exp Clin Cancer Res 38: 138. [Crossref]
10. Baskaran N,
Manoharan S, Kowsalya R, Silvan S, Palanimuthu D (2012) Coumarin modulates
apoptotic, cell proliferative, inflammatory and angiogenic markers in favor of
tumor inhibition during 7,12-dimethylbenz[a]anthracene-induced hamster buccal
pouch carcinogenesis. Int J Res Pharm Sci 3: 1-9.
11. Lee KA, Shepherd
ST, Johnston SR (2019) Abemaciclib, a potent cyclin-dependent kinase 4 and 6
inhibitor, for treatment of ER-positive metastatic breast cancer. Future
Oncol 15: 3309-3326. [Crossref]
12. Butt AJ, Caldon CE,
McNeil CM, Swarbrick A, Musgrove EA et al. (2008) Cell cycle machinery: links
with genesis and treatment of breast cancer. Adv Exp Med Biol 630:
189-205. [Crossref]
13. Semenza GL, Koury
ST, Nejfelt MK, Gearhart JD, Antonarakis SE (1991) Cell-type-specific and hypoxia-inducible
expression of the human erythropoietin gene in transgenic mice. Proc Natl
Acad Sci U S A 88: 8725-8729. [Crossref]
14. Masoud GN, Li W
(2015) HIF-1α pathway: role, regulation and intervention for cancer therapy. Acta
Pharm Sin B 5 :378-389. [Crossref]
15. Wolff M, Jelkmann
W, Dunst J, Depping R (2013) The Aryl Hydrocarbon Receptor Nuclear Translocator
(ARNT/HIF-1beta) is influenced by hypoxia and hypoxia-mimetics. Cell Physiol
Biochem 32: 849-858. [Crossref]
16. Minet E, Mottet D, Michel G, Roland I, Raes M et al. (1999)
Hypoxia-induced activation of HIF-1: role of HIF-1alpha-Hsp90 interaction. FEBS
Lett 460: 251-256. [Crossref]
17. Drutel G, Kathmann
M, Héron A, Gros C, Macé S et al. (2000) Two splice variants of the
hypoxia-inducible factor HIF-1alpha as potential dimerization partners of ARNT2
in neurons. Eur J Neurosci 12: 3701-3708. [Crossref]
18. Balamurugan K
(2016) HIF-1 at the crossroads of hypoxia, inflammation, and cancer. Int J Cancer
138: 1058-1066. [Crossref]
19. Pezzuto A, Carico E
(2018) Role of HIF-1 in Cancer Progression: Novel Insights. A Review. Curr Mol Med 18: 343-351. [Crossref]
20. Liu H, Ren X, Ma C
(2018) Effect of Berberine on Angiogenesis and HIF-1α / VEGF Signal
Transduction Pathway in Rats with Cerebral Ischemia - Reperfusion Injury. J
Coll Physicians Surg Pak 28: 753-757. [Crossref]
21. Cheng CY, Ho TY,
Hsiang CY, Tang NY, Hsieh CL et al. (2017) Angelica sinensis Exerts Angiogenic
and Anti-apoptotic Effects Against Cerebral Ischemia-Reperfusion Injury by
Activating p38MAPK/HIF-1[Formula: see text]/VEGF-A Signaling in Rats. Am J
Chin Med 45: 1683-1708. [Crossref]
22. Voronov E,
Dinarello CA, Apte RN (2018) Interleukin-1alpha as an intracellular alarmin in
cancer biology. Semin Immunol 38: 3-14. [Crossref]
23. Roh JS, Sohn DH
(2018) Damage-Associated Molecular Patterns in Inflammatory Diseases. Immune
Netw 18: e27. [Crossref]
24. Bergami PL, Barbero
G (2020) The emerging role of Wnt5a in the promotion of a pro-inflammatory and
immunosuppressive tumor microenvironment. Cancer Metastasis Rev 39:
933-952. [Crossref]
25. Fabre J, Giustiniani J, Garbar C, Antonicelli F, Merrouche Y et al. (2016) Targeting
the Tumor Microenvironment: The Protumor Effects of IL-17 Related to Cancer
Type. Int J Mol Sci 17: 1433. [Crossref]
26. Núñez MS,
Bouthelier A, Somavilla AS, Hernández RM, Adrados M et al. (2020) LAT-1 and
GLUT-1 Carrier Expression and Its Prognostic Value in Gastroenteropancreatic
Neuroendocrine Tumors. Cancers (Basel) 12: 2968. [Crossref]
27. Sharen G, Peng Y,
Cheng H, Liu Y, Shi Y et al. (2017) Prognostic value of GLUT-1 expression in
pancreatic cancer: results from 538 patients. Oncotarget 8: 19760-19767.
[Crossref]
28. Boira MA, Martino
MD, Gordillo C, Adrados M, Perez EM (2020) GLUT-1 as a predictor of worse
prognosis in pancreatic adenocarcinoma: immunohistochemistry study showing the
correlation between expression and survival. BMC Cancer 20: 909. [Crossref]
29. Zahid H, Simpson
ER, Brown KA (2016) Inflammation, dysregulated metabolism and aromatase in
obesity and breast cancer. Curr Opin Pharmacol 31: 90-96. [Crossref]
30. Rajagopal C,
Lankadasari MB, Aranjani JM, Harikumar KB (2018) Targeting oncogenic
transcription factors by polyphenols: A novel approach for cancer therapy. Pharmacol
Res 130: 273-291. [Crossref]
31. Rozpedek W, Pytel
D, Mucha B, Leszczynska H, Diehl JA et al. (2016) The Role of the
PERK/eIF2α/ATF4/CHOP Signaling Pathway in Tumor Progression During Endoplasmic
Reticulum Stress. Curr Mol Med 16: 533-544. [Crossref]
32. Budanov AV (2014)
The role of tumor suppressor p53 in the antioxidant defense and metabolism. Subcell
Biochem 85: 337-358. [Crossref]
33. Reddy ND, Shoja MH,
Biswas S, Nayak PG, Kumar N et al. (2016) An appraisal of cinnamyl sulfonamide
hydroxamate derivatives (HDAC inhibitors) for anti-cancer, anti-angiogenic and
anti-metastatic activities in human cancer cells. Chem Biol Interact
253: 112-124. [Crossref]
34. Nemade H, Chaudhari
U, Acharya A, Hescheler J, Hengstler JG et al. (2018) Cell death mechanisms of
the anti-cancer drug etoposide on human cardiomyocytes isolated from
pluripotent stem cells. Arch Toxicol 92: 1507-1524. [Crossref]
35. Pabla N, Huang S,
Mi QS, Daniel R, Dong Z (2008) ATR-Chk2 signaling in p53 activation and DNA
damage response during cisplatin-induced apoptosis. J Biol Chem 283:
6572-6583. [Crossref]
36. Wang Z, Inuzuka H,
Zhong J, Fukushima H, Wan L et al. (2012) DNA damage-induced activation of ATM
promotes β-TRCP-mediated Mdm2 ubiquitination and destruction. Oncotarget 3:
1026-1035. [Crossref]
37. Colin DJ, Hain KO,
Allan LA, Clarke PR (2015) Cellular responses to a prolonged delay in mitosis
are determined by a DNA damage response controlled by Bcl-2 family proteins. Open
Biol 5: 140156. [Crossref]
38. Verkhivker GM
(2012) Simulating molecular mechanisms of the MDM2-mediated regulatory
interactions: a conformational selection model of the MDM2 lid dynamics. PLoS
One 7: e40897. [Crossref]
39. Lu Y, Qin T, Li J,
Wang L, Zhang Q et al. (2017) MicroRNA-140-5p inhibits invasion and
angiogenesis through targeting VEGF-A in breast cancer. Cancer Gene Ther
24: 386-392. [Crossref]
40. Li D, Xie K, Zhang L, Yao X, Li H et al. (2016) Dual blockade of vascular
endothelial growth factor (VEGF) and basic fibroblast growth factor (FGF-2)
exhibits potent anti-angiogenic effects. Cancer Lett 377: 164-173. [Crossref]
41. Ahluwalia A, Jones
MK, Budnik TM, Tarnawski AS (2014) VEGF and colon cancer growth beyond
angiogenesis: does VEGF directly mediate colon cancer growth via a
non-angiogenic mechanism? Curr Pharm Des 20: 1041-1044. [Crossref]
42. Dedieu S, Canron X,
Rezvani HR, Bouchecareilh M, Mazurier F et al. (2010) The cytoprotective drug
amifostine modifies both expression and activity of the pro-angiogenic factor
VEGF-A. BMC Med 8:19. [Crossref]
43. Lim CP, Cao X
(2004) Structure, function, and regulation of STAT proteins. Mol Biosyst
2: 536-550. [Crossref]
44. Turkson J (2004)
STAT proteins as novel targets for cancer drug discovery. Expert Opin Ther
Targets 8: 409-422. [Crossref]
45. Heinrich PC,
Behrmann I, Haan S, Hermanns HM, Newen GM et al. (2003) Principles of
interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J
374: 1-20. [Crossref]
46. Uciechowski P,
Dempke WCM (2020) Interleukin-6: A Masterplayer in the Cytokine Network. Oncology
98: 131-137. [Crossref]
47. Tomassi S, Tosato S
(2017) Epigenetics and gene expression profile in first-episode psychosis: The
role of childhood trauma. Neurosci Biobehav Rev 83: 226-237. [Crossref]
48. Wang IM, Lin H,
Goldman SJ, Kobayashi M (2004) STAT-1 is activated by IL-4 and IL-13 in
multiple cell types. Mol Immunol 41: 873-884. [Crossref]
49. Zhang Q, Lee B,
Korecka M, Li G, Weyland C et al. (1999) Differences in phosphorylation of the
IL-2R associated JAK/STAT proteins between HTLV-I(+), IL-2-independent and
IL-2-dependent cell lines and uncultured leukemic cells from patients with
adult T-cell lymphoma/leukemia. Leuk Res 23: 373-384. [Crossref]
50. Dumoutier L,
Leemans C, Lejeune D, Kotenko SV, Renauld JC (2001) Cutting edge: STAT
activation by IL-19, IL-20 and mda-7 through IL-20 receptor complexes of two
types. J Immunol 167: 3545-3549. [Crossref]
51. Frey AB, Wali A,
Pass H, Lonardo F (2007) Osteopontin is linked to p65 and MMP-9 expression in
pulmonary adenocarcinoma but not in malignant pleural mesothelioma. Histopathology
50: 720-726. [Crossref]
52. Baskaran N, Selvam
GS, Yuvaraj S, Abhishek A (2018) Parthenolide attenuates
7,12-dimethylbenz[a]anthracene induced hamster buccal pouch carcinogenesis. Mol
Cell Biochem 440: 11-22. [Crossref]
53. Martín VS, García
LJ, Herranz S, Luque A, Acebo P et al. (2019) α-Hispanolol Induces Apoptosis
and Suppresses Migration and Invasion of Glioblastoma Cells Likely via
Downregulation of MMP-2/9 Expression and p38MAPK Attenuation. Front
Pharmacol 10: 935. [Crossref]
54. Yan Y, Liang H, Li T, Li M, Li R et al. (2014) The MMP-1, MMP-2, and MMP-9 gene
polymorphisms and susceptibility to bladder cancer: a meta-analysis. Tumour
Biol 35: 3047-3052. [Crossref]
55. Luukkaa H, Klemi P,
Hirsimäki P, Vahlberg T, Kivisaari A et al. (2008) Matrix metalloproteinase
(MMP)-1, -9 and -13 as prognostic factors in salivary gland cancer. Acta
Otolaryngol 128: 482-490. [Crossref]
56. Zhang X, Jin G, Li
J, Zhang L (2015) Association between four MMP-9 polymorphisms and breast
cancer risk: a meta-analysis. Med Sci Monit 21: 1115-1123. [Crossref]
57. Eck SM, Hoopes PJ,
Petrella BL, Coon CI, Brinckerhoff CE (2009) Matrix metalloproteinase-1
promotes breast cancer angiogenesis and osteolysis in a novel in vivo model. Breast
Cancer Res Treat 116: 79-90. [Crossref]
58. Naglich JG, Kunkel MJ, Gupta E, Fargnoli J, Henderson AJ et al. (2001) Inhibition
of angiogenesis and metastasis in two murine models by the matrix
metalloproteinase inhibitor, BMS-275291. Cancer Res 61: 8480-8485. [Crossref]
59. Nozaki S, Maeda M,
Tsuda H, Sledge GW Jr (2004) Inhibition of breast cancer regrowth and pulmonary
metastasis in nude mice by anti-gastric ulcer agent, irsogladine. Breast
Cancer Res Treat 83: 195-199. [Crossref]
60. Maki H, Maekawa R,
Yoshida H, Hojo K, Uchida N et al. (1999) Antiangiogenic and antitumor effect
of BPHA, an orally-active matrix metalloproteinase inhibitor, in in vivo murine
and human tumor model. Gan To Kagaku Ryoho 26: 1599-1606. [Crossref]
61. Wei X, Shao X
(2018) Nobiletin alleviates endometriosis via down-regulating NF-κB activity in
endometriosis mouse model. Biosci Rep 38: BSR20180470. [Crossref]
62. Premaratne GU, Ma LP, Fujita M, Lin X, Bollano E et al. (2011) Stromal
vascular fraction transplantation as an alternative therapy for ischemic heart
failure: anti-inflammatory role. J Cardiothorac Surg 6: 43. [Crossref]
63. Raza QS, Vanderploeg JL, Jacobs JR (2017) Matrix Metalloproteinases are required for membrane motility and lumenogenesis during Drosophila heart development. PLoS One 12: e0171905. [Crossref]
64. Peters F, Pauly CB (2019) Role of meprin metalloproteases in metastasis and tumor microenvironment. Cancer Metastasis Rev 38: 347-356. [Crossref]
