Appearance of 4 Degree Rash While Treating Advanced Lung Cancer with Icotinib – Whether to Stop the Drug: A Case Report
A B S T R A C T
Skin and skin adnexa toxicities are the most common side effects associated with epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs). Previous research showed that the rash appeared, and the severity of EGFR-TKIs may be a marker of clinical benefit. In this context, we report a 75-year-old man with advanced lung cancer who on receiving icotinib developed severe adverse reactions, 4 degree rash (NCI-CTC AE version 4.0 common toxicity grading standards grade), and refused to stop taking the drug; but with the anti-infection and symptomatic nursing, the patient recovered, the rash disappeared, and the patient received a better prognosis. Thus, we would like to emphasize the importance of deciding whether to stop the drug when patients developed adverse reactions of 3-4 degree rash.
Keywords
Lung cancer, icotinib, adverse reactions, rash
Key Points
I Significant Findings of the Study
A patient who received EGFR-TKIs developed 4 degree rash and refused to stop taking the drug, but through the symptomatic nursing the patient recovered, the rash disappeared, and he received a better prognosis.
II What This Study Adds
The rash appeared and the severity of EGFR-TKIs may be a marker of clinical benefit, and when patients developed adverse reactions of 3-4 degree rash, whether there is a need to stop the drug.
Introduction
Icotinib is a first-generation EGFR-TKI developed by Zhejiang Beta Pharma in China, which is the first small molecular targeted drugs and has gained international recognition [1]. The main adverse reactions of icotinib are rash and diarrhea; the incidence of adverse events and the severity of rash were lower than gefitinib and erlotinib [2-5]. In terms of efficacy, icotinib and gefitinib are equivalent, but icotinib has a better security [6]. Therefore, for patients with EGFR mutation and the elderly who cannot tolerate chemotherapy, icotinib is more appropriate [3]. We report a 75-year-old man with advanced lung cancer who received icotinib, developed severe adverse reactions, and refused to stop taking the drug, but through anti-infection the rash disappeared and the patient received a better prognosis.
Case Report
The patient, a 75-year-old male with “recurrent cough, sputum for more than 30 years, and then increased with left chest pain more than 4 months” was admitted. The chest computed tomography (CT) scan showed: left lower lobe mass shadow, more likely to consider the surrounding lung cancer, bilateral middle volume of pleural effusion; bone scan showed multiple metastatic bone lesions. He underwent a bilateral pleural effusion puncture drainage operation, and the cytology test showed malignant tumor cells. We could not further clarify the pathological type of lung cancer because the patient refused further pathological examination. Through the combination of tumor markers detection and clinical manifestations, he was considered to be diagnosed with lung adenocarcinoma and clinically staged as cT2aN0M1b, stage IV. So, this patient was given “bevacizumab 100mg and cisplatin 30mg and normal saline 20ml” pleural perfusion chemotherapy 2 times. Blood tests found mutations in EGFR; so, the patient received 125 mg oral icotinib in tablet form three times per day to maintain. This study was conducted in accordance with the declaration of Helsinki. This study was conducted with approval from the Ethics Committee of The Second Affiliated Hospital of Kunming Medical University. Written informed consent was obtained from all participants.
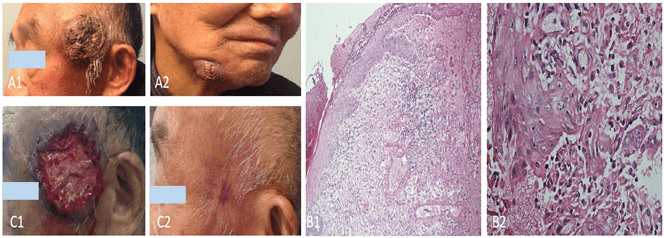
But the patient's face began to develop red papules after taking icotinib for 7 days, which were considered the side effects of drugs, but he did not receive any special treatment. Since then, the rash was significantly increased, fused into masses, canker scab, accompanied by purulent secretions (Figures 1A1 & 1A2). We recommended that patient stops taking icotinib, but he refused, worrying about rapid recurrence and metastasis. The abscess on the face of the patient was cut open and drained in surgery. The biopsy of the bump showed squamous epithelium hyperplasia, with atypical hyperplasia, chronic inflammation of the dermis and abscess formation (Figures 1B1 & 1B2). After 2 weeks of anti-infection treatment, the patient's skin rash wound secretion decreased, and the granulation rate was slow (Figures 1C1 & 1C2).
Figure 1: A1 & A2) Patient occurred adverse reactions of III-IV skin after icotinib treatment. Left temporal of the patient see about 6×8cm mass, which protruding from the surface of the skin, the boundary is unclear, mass surface ulceration, crusting, overlying purulent secretions (A1); the right lower jaw shows about 3×5cm mass, fusion, ulceration (A1) (December 29, 2014). B1 & B2) HE of the bump on the left temporal of patient. It showed squamous epithelium hyperplasia which partly accompanied by atypical hyperplasia; chronic suppurative inflammation of the dermis with abscess formation, no clear neoplastic lesions. (B1) H&E stain, original magnification (×40); (B2) H&E stain, original magnification (×400). C1 & C2) The left temporal mass purulent secretion was completely drained, exudation less in January 14, 2015 (C1); the left temporal mass of patient has disappeared in March 27, 2015 (C2).
Twenty days later, CT revealed tumor regression; the curative effect was evaluated as partial remission (PR) (Figures 2A & 2B). Two months later (Figures 2C1 & 2C2), and three months later (Figures 2D1 & 2D2), a PR was also confirmed in the chest CT scan. However, four months later, the CT scan revealed that the tumor grew larger, the efficacy was evaluated as progressive disease (PD) (Figures 2E1 & 2E2). After medication for more than 7 months, a PD was confirmed in the chest CT scan (Figures 2F1 & 2F2). In addition, the tumor markers of patient were significantly decreased (Table 1).
Table 1: Changes of tumor markers.
|
Date |
CEA (ng/ml) |
NSE (ng/ml) |
CA125 (U/ml) |
CA199 (U/ml) |
|
Oct. 21, 2014 |
54.99 |
- |
826.84 |
- |
|
Dec. 9, 2014 |
36.57 |
16.24 |
95.77 |
<2.00 |
|
Feb. 3, 2015 |
26.03 |
14.28 |
85.89 |
<2.00 |
|
Mar.27, 2015 |
19.67 |
13.36 |
75.24 |
0.01 |
CEA: Carcinoembryonic Antigen; CA: Carbohydrate Antigen; NSE: Neuron Specific Enolase; -: without detection; <: lower than.
Figure 2: A) The Lung condition before treatment with icotinib. The lower lobe of the left lung heart perivascular saw a mass shadow, about the size of 4.8×1.9cm, CT value 38-47Hu, with small spot high density shadow, the edge of the visible less light, visible shallow lobes. Double lung markings increased, multiple nodules on the dot sheets and point density in the middle lobe of the right lung distribution obviously. Mediastinal lymph node enlargement can be seen. Bilateral small pleural effusion. Pulmonary window (A1), mediastinal window (A2). B) The pulmonary mass decreased, the inflammation subsided, and the pleural effusion was obviously absorbed after 20 days treatment. The lower lobe of the left lung hilar region see soft tissue mass with sharp edges, the size of about 3.4×3.0cm. The lower lobe of the left lung was seen several small nodules, some of which were slightly enhanced. Bilateral pleural thickening and adhesion, bilateral pleural effusion was absorbed significantly. Pulmonary window (B1), mediastinal window (B2). C) The pulmonary mass further decreased after 2 months treatment with icotinib. The lower lobe of the left lung hilar region showed soft tissue mass, about the size of 3.23×1.41cm. Pulmonary window (C1), Mediastinal window (C2). D) The pulmonary mass further decreased after 3 months treatment with icotinib. The lower lobe of the left lung hilar region showed soft tissue mass, about the size of 2.95×1.22cm. Pulmonary window (D1), mediastinal window (D2). E) The pulmonary mass further decreased after 4 months treatment with icotinib. The lower lobe of the left lung hilar region showed soft tissue mass, about the size of 4.55×1.68cm. Mediastinal lymph node enlargement can be seen. Bilateral pleural thickening and adhesion, moderate right pleural effusion can be seen. Pulmonary window (E1), mediastinal window (E2). F) The pulmonary mass further decreased after 7 months treatment with icotinib. The lower lobe of the left lung hilar region showed soft tissue mass, about the size of 6.69×4.44cm. Mediastinal lymph node enlargement can be seen, bilateral pleural effusion was further increased. Pulmonary window (F1), mediastinal window (F2).
Discussion
EGFR-TKIs inhibitor development has shown great superiority in the treatment of lung cancer, but it also brings a variety of skin toxicity [7]. Previous studies confirm that the presence and severity of rash may be a sign of clinical benefit for EGFR-TKIs, which can predict the risk of disease progression [8-10]. Thus, icotinib for the treatment of advanced non-small cell lung cancer and skin rash may be a good prognostic factor.
We report that this patient's face developed 4 degree rash after taking icotinib. According to evidence of withdrawal of adverse reactions to icotinib, it is generally recommended that patients stop taking the drug, but this patient refused to stop taking icotinib; but with active symptomatic treatments, the left temporal mass disappeared [11]. The PFS of this patient was 4.7 months and the OS was 8.0 months, which was similar to the results of ICOGEN studies [6]. And this patient's tumor markers CEA, CA125 decreased significantly, and the tumor was also significantly reduced.
In conclusion, icotinib for the treatment of advanced non-small cell lung cancer can be used as a choice, and the rash may be a good prognostic sign. Clinical workers need to further explore whether targeted therapy should be discontinued when severe adverse skin reactions occur. A large number of clinical trials are needed for that.
Acknowledgements
The authors thank the patient’s family for their collaboration.
Conflicts of Interest
The authors declare no conflicts of interest.
Consent
Written informed consent was obtained from the patient’s closest relatives for publication of this case report and any accompanying images. A copy of the written consent is available for review by the series editor of this journal.
Funding
This study was supported by the Research of Yunnan Provence Science and Technology Planning Project (Project No.202001AT070027); the National Natural Science Foundation of China (Project No. 81960423) and the Second Affiliated Hospital of Kunming Medical University Program (Project No. 2019YK001, Project No. RCPYXM2017-2-05).
Article Info
Article Type
Case ReportPublication history
Received: Wed 18, Nov 2020Accepted: Wed 02, Dec 2020
Published: Mon 14, Dec 2020
Copyright
© 2023 Jie Lin. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.ACO.2020.03.02
Author Info
Li Lv Lifeng Jiang Wenhui Zhang Xinyan Lu Tingting Li Jie Lin
Corresponding Author
Jie LinDepartment of Medical Oncology, The Second Affiliated Hospital of Kunming Medical University, Kunming, China
Figures & Tables
Table 1: Changes of tumor markers.
|
Date |
CEA (ng/ml) |
NSE (ng/ml) |
CA125 (U/ml) |
CA199 (U/ml) |
|
Oct. 21, 2014 |
54.99 |
- |
826.84 |
- |
|
Dec. 9, 2014 |
36.57 |
16.24 |
95.77 |
<2.00 |
|
Feb. 3, 2015 |
26.03 |
14.28 |
85.89 |
<2.00 |
|
Mar.27, 2015 |
19.67 |
13.36 |
75.24 |
0.01 |
CEA: Carcinoembryonic Antigen; CA: Carbohydrate Antigen; NSE: Neuron Specific Enolase; -: without detection; <: lower than.

References
- Camidge DR (2013) Icotinib: kick-starting the Chinese anticancer drug industry. Lancet Oncol 14: 913-914. [Crossref]
- Liu D, Zhang L, Wu Y, Jiang J, Tan F et al. (2015) Clinical pharmacokinetics, safety, and preliminary efficacy evaluation of icotinib in patients with advanced non-small cell lung cancer. Lung Cancer 89: 262-267. [Crossref]
- Zhang W, Zhang Y, Zhao Q, Liu X, Chen L et al. (2020) Long-term safety of icotinib in patients with non-small cell lung cancer: a retrospective, real-world study. J Thorac Dis 12: 639-650. [Crossref]
- Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT et al. (2009) Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med 361: 947-957. [Crossref]
- Burotto M, Manasanch EE, Wilkerson J, Fojo T (2015) Gefitinib and erlotinib in metastatic non-small cell lung cancer: a meta-analysis of toxicity and efficacy of randomized clinical trials. Oncologist 20: 400-410. [Crossref]
- Shi Y, Zhang L, Liu X, Zhou C, Zhang L et al. (2013) Icotinib versus gefitinib in previously treated advanced non-small-cell lung cancer (ICOGEN): a randomised, double-blind phase 3 non-inferiority trial. Lancet Oncol 14: 953-961. [Crossref]
- Nanney LB, Stoscheck CM, Jr LEK, Underwood RA, Holbrook KA (1990) Immunolocalization of Epidermal Growth Factor Receptors in Normal Developing Human Skin. J Invest Dermatol 94: 742-748. [Crossref]
- Bezjak A, Lee CW, Ding K, Brundage M, Winton T et al. (2008) Quality-of-Life Outcomes for Adjuvant Chemotherapy in Early-Stage Non–Small-Cell Lung Cancer: Results from a Randomized Trial, JBR.10. J Clin Oncol 26: 5052-5059. [Crossref]
- Perng RP, Yang CH, Chen YM, Chang GC, Lin MC et al. (2008) High efficacy of erlotinib in Taiwanese NSCLC patients in an expanded access program study previously treated with chemotherapy. Lung Cancer 62: 78-84. [Crossref]
- Lee Y, Shim HS, Park MS, Kim JH, Ha SJ et al. (2012) High EGFR gene copy number and skin rash as predictive markers for EGFR tyrosine kinase inhibitors in patients with advanced squamous cell lung carcinoma. Clin Cancer Res 18: 1760-1768. [Crossref]
- Kris MG, Natale RB, Herbst RS, Jr TJL, Prager D et al. (2003) Efficacy of gefitinib, an inhibitor of the epidermal growth factor receptor tyrosine kinase, in symptomatic patients with non-small cell lung cancer: a randomized trial. JAMA 290: 2149-2158. [Crossref]
