Accelerated Onset of Liver Failure after Prolonged Adjuvant Tamoxifen Use in Breast Cancer Patients
A B S T R A C T
Use of adjuvant endocrine therapy for women with hormone-receptor (HR)-positive breast cancer has become the standard of care. Tamoxifen, an orally available selective estrogen receptor modulator (SERM), is a commonly used endocrine therapy agent currently recommended for use in pre- or post-menopausal women with HR-positive breast cancer. Current evidence suggests that prolonged tamoxifen use may be implicated in causing hepatotoxicity which may manifest as non-alcoholic steatohepatitis (NASH), cholestasis, cirrhosis, or hepatic necrosis. We herein present two cases of suspected tamoxifen-induced NASH resulting in fulminant liver failure. We also discuss literature surrounding tamoxifen-related hepatoxicity and implications in clinical practice.
Keywords
Tamoxifen, breast cancer, nonalcoholic steatohepatitis, NASH, hepatotoxicity
Introduction
Use of adjuvant endocrine therapy for women with hormone-receptor (HR)-positive breast cancer has become the standard of care. Tamoxifen, an orally available selective estrogen receptor modulator (SERM), is a commonly used endocrine therapy agent currently recommended for use in pre- or post-menopausal women with HR-positive breast cancer [1, 2]. Current guidelines recommend a total duration ranging from five to 10 years [1, 2]. Often, the decision to continue tamoxifen beyond the recommended time frame takes into consideration patient tolerability and quality of life. Endocrine therapy is often associated with adverse effects mimicking menopause including but not limited to the following: hot flashes, vaginal atrophy, diminished libido, mood changes, fatigue, hypercholesterolemia, arthralgia, and decreased bone density [3]. While tamoxifen is no exception to these adverse effects, there are more serious adverse effects unique to tamoxifen such as hepatotoxicity which may manifest as non-alcoholic steatohepatitis (NASH), cholestasis, cirrhosis, or hepatic necrosis [4-11]. In a study of 136 patients with breast cancer, hepatic steatosis was observed in approximately 38 percent of patients taking tamoxifen [11]. Notably, six of these patients had biopsy-confirmed NASH. Drug-induced NASH, which is thought to be associated with prolonged use of an offending agent, can be characterized by progressive steatohepatitis even after the offending agent has been discontinued [9]. The mechanism by which tamoxifen produces NASH may be two-fold as discussed previously [9, 11]. First, tamoxifen may directly produce steatohepatitis via inhibition of mitochondrial beta-oxidation of fatty acids [9]. Because tamoxifen is metabolized to active metabolites via CYP2D6, a cytochrome p450 enzyme that is polymorphically expressed, patients that are rapid metabolizers of tamoxifen may be further predisposed to hepatic accumulation and toxicity [4, 12]. Second, tamoxifen may indirectly cause NASH by accelerating risk factors for NASH such as hypertriglyceridemia [9]. While a correlation between tamoxifen use and hepatotoxicity is likely considering existing literature, it is unclear if tamoxifen expedites the development of fulminant liver failure in breast cancer patients with new hepatic metastases. We herein present a case report of two breast cancer patients who were previously managed on adjuvant tamoxifen therapy and experienced metastasis to the liver and subsequently, rapid liver failure.
Patient Case 1
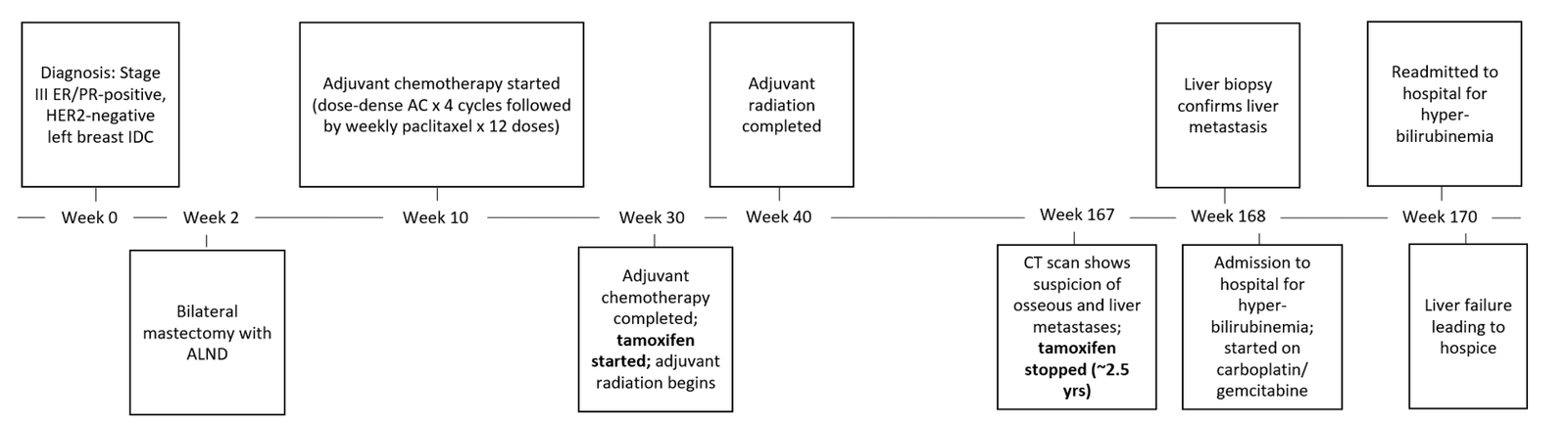
A 48-year-old, premenopausal, Caucasian female presented to clinic and was diagnosed with clinical stage III, estrogen receptor (ER)-positive, progesterone receptor (PR)-positive, HER2-negative left breast invasive ductal carcinoma (IDC). At the time of diagnosis, past medical history was significant only for obesity (Figure 1). The patient declined neoadjuvant chemotherapy and therefore underwent a bilateral mastectomy and left axillary lymph node dissection (ALND). Subsequently, the patient received four cycles of adjuvant chemotherapy with dose-dense doxorubicin and cyclophosphamide (AC) followed by 12 total doses of weekly paclitaxel. Adjuvant endocrine therapy with tamoxifen at the standard daily dose of 20 mg was initiated alongside adjuvant radiation. The patient remained stable on tamoxifen for approximately 2.5 years until a CT scan, performed for complaint of right upper quadrant pain, revealed the presence of sclerotic osseous lesions and innumerable hypodense lesions throughout the liver concerning for metastatic disease. Laboratory tests revealed notable transaminitis and an elevated total bilirubin level of 1.4 mg/dL. A biopsy of the hepatic lesion revealed the presence of hepatic adenocarcinoma consistent with breast cancer origin (ER/PR-positive, HER2-negative).
Tamoxifen was discontinued at this time due to progressive disease. Approximately two days following the liver biopsy, the patient presented to the emergency department with worsening shortness of breath and acute right upper quadrant abdominal pain. Further work-up revealed hyperbilirubinemia (3.7 mg/dL) and transaminitis, and a magnetic resonance cholangiopancreatography (MRCP) demonstrated the presence of hepatomegaly with diffuse hepatic metastases and heterogenous parenchyma in the absence of biliary or pancreatic ductal dilatation. The patient was started on palliative chemotherapy with carboplatin and gemcitabine, and she received one treatment of dose-reduced chemotherapy before being readmitted to the hospital for worsening hyperbilirubinemia (12.2 mg/dL). A repeat MRCP demonstrated near-complete replacement of the liver parenchyma by hepatic metastases in addition to new-onset ascites. Given worsening liver failure, severe pain, declining mental status, the patient decided to transition to hospice, and chemotherapy was not resumed. The patient passed away shortly after at the age of 51 years.
Figure 1: Timeline of Events for Patient Case 1.
AC: Doxorubicin, Cyclophosphamide; ALND: Axillary Lymph Node Dissection; ER: Estrogen Receptor; IDC: Invasive Ductal Carcinoma; PR: Progesterone Receptor.
Patient Case 2
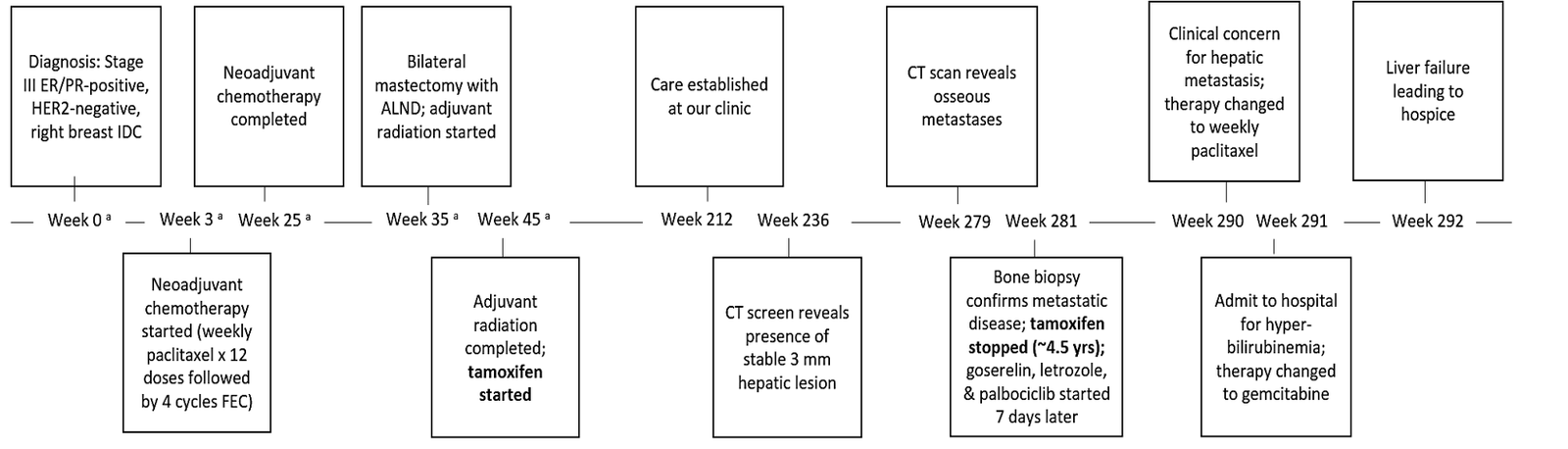
A 32-year-old, premenopausal, Caucasian female with clinical stage III, ER/PR-positive, HER2-negative, BRCA-positive right breast IDC presented to clinic after transferring care from an outside facility (Figure 2). The patient’s past medical history was significant only for hypothyroidism at the time of diagnosis. A review of the patient’s oncologic history revealed that the patient had previously received neoadjuvant chemotherapy consisting of 12 doses of weekly paclitaxel followed by four cycles of 5-fluorouracil, epirubicin, and cyclophosphamide (FEC). The patient then underwent a bilateral mastectomy and right ALND followed by adjuvant radiation therapy. Adjuvant endocrine therapy with tamoxifen was then initiated at a standard daily dose of 20 mg. Approximately four years from initial diagnosis, the patient established care at our cancer center, and adjuvant tamoxifen therapy was continued. Fifteen months later, a CT scan performed in the setting of back pain demonstrated findings consistent with osseous metastases in the appendicular and axial skeleton. A biopsy of lytic osseous lesions revealed presence of metastatic adenocarcinoma consistent with breast cancer origin (ER/PR-positive, HER2-negative).
Due to disease progression, the patient was transitioned from tamoxifen to letrozole, palbociclib, and goserelin. Approximately two months later, the patient was noted to have new onset transaminitis clinically concerning for hepatic metastases, and the patient was transitioned from palbociclib to weekly paclitaxel (three weekly doses per 28-day cycle). An MRI of the abdomen performed at this time unfortunately revealed the presence of several enhancing lesions throughout all hepatic lobes consistent with diffuse metastatic disease. One week later, the patient was directly admitted to the hospital for worsening transaminitis, new-onset hyperbilirubinemia (10 mg/dL), and complaint of abdominal pain. In response to the clinical concern for biliary obstruction, and abdominal ultrasound and MRCP were performed confirming the presence of innumerable hepatic lesions in the absence of biliary or pancreatic ductal dilatation. The patient’s chemotherapy regimen was then emergently switched from paclitaxel to gemcitabine. Unfortunately, the patient’s transaminitis and hyperbilirubinemia (16.4 mg/dL) only worsened after receiving only one dose of gemcitabine, and the patient decided to transition to hospice for worsening liver failure, up trending INR, and pancytopenia. The patient passed away shortly after at the age of 38 years.
Figure 2: Timeline of Events for Patient Case 2.
ALND: Axillary Lymph Node Dissection; ER: Estrogen Receptor; FEC: 5-Fluorouracil/Epirubicin/Cyclophosphamide; IDC: Invasive Ductal Carcinoma; PR: Progesterone Receptor.
a = Exact timing unclear given patient treated at outside facility during this timeframe.
Discussion
In the case reports described above, each patient experienced fulminant liver failure with history of tamoxifen use. While there are case reports of acute exacerbation of pre-existing liver dysfunction after months to years of tamoxifen use, we believe that the prolonged use of tamoxifen in our patients may have directly precipitated NASH, ultimately accelerating fulminant liver failure in two patients whose breast cancer metastasized to the liver. NASH is suspected in both patient cases given the progressive hepatic decline that occurred even upon discontinuation of tamoxifen. Furthermore, comorbid obesity in the first patient case described may have increased predisposition to NASH [9]. Taken together, tamoxifen-induced steatohepatitis and malignant infiltration of the liver may have synergistically induced hepatic insult and rapid liver failure. It should be noted, however, that a liver biopsy was not able to be obtained in both cases to confirm the clinical suspicion of NASH.
The aggressive hepatic decline observed in both cases elicits discussion regarding prevention of NASH in patients receiving prolonged tamoxifen therapy. Indeed, tamoxifen-induced NASH warrants early detection in order to improve patient outcomes and reduce mortality. To date, little guidance is available on monitoring for tamoxifen-induced hepatotoxicity in patients with pre-existing hepatic comorbidities. According to manufacturer labeling, liver function tests (LFTs) and bilirubin levels should be obtained periodically while a patient is receiving tamoxifen [4]. Considering the progressive nature of tamoxifen-induced hepatotoxicity in the above cases, we suggest that these parameters be monitored more frequently in patients with underlying hepatic comorbidities or risk factors for NASH such as obesity, insulin resistance, and lipid abnormalities.
These case reports have prompted a change in our personal practice in that patients receiving prolonged tamoxifen therapy will have comprehensive metabolic panels (CMPs) obtained every six months as opposed to every 12 months. Cessation of therapy should be considered in patients who experience new onset LFT elevations without other explanation. Currently, manufacturer labeling does not provide guidance for tamoxifen dose adjustments in the setting of hepatic dysfunction [4]. However, a reduced tamoxifen dose of 10 mg every other day was shown to help normalize seemingly supratherapeutic tamoxifen and metabolite serum levels in one case report of a patient with underlying hepatic dysfunction [8]. Finally, though not routinely performed at most centers, tamoxifen metabolite (endoxifen) serum monitoring and/or CYP2D6 genotype testing may be of utility and deserves further investigation in preventing tamoxifen-related hepatotoxicity.
Conclusion
While causality is not confirmed, the case reports described above suggest that prolonged tamoxifen could have contributed to fulminant liver failure in patients who developed metastasis to the liver. Tamoxifen-induced NASH may be a plausible explanation. Closer LFT monitoring and more stringent criteria for tamoxifen discontinuation or dose reduction should be considered in patients with otherwise unexplained significant LFT abnormalities. Further studies are warranted at this time.
Funding
None.
Ethics Approval, Statement of Compliance, and Clinical Trial Transparency
• IRB ethics committee approval obtained (IRB-exempt for the purpose of this case report).
• Informed patient consent not applicable.
Compliance with relevant laws and institutional guidelines for human and animal subjects including:
• Experiments involving humans: The Code of Ethics of the World Medical Association (Declaration of Helsinki).
• Experiments involving animals: ARRIVE guidelines and carried out in accordance with the U.K. Animals (Scientific Procedures) Act 1986 and associated guidelines, EU Directive 2010/63/EU on the protection of animals used for scientific purposes, or the U.S. Public Health Service Policy on Humane Care and Use of Laboratory Animals and, as applicable, the Animal Welfare Act.
• Clinical trial transparency.
Acknowledgement
None.
Conflicts of Interest
None.
Article Info
Article Type
Case ReportPublication history
Received: Sat 11, Apr 2020Accepted: Mon 27, Apr 2020
Published: Wed 29, Apr 2020
Copyright
© 2023 Hayley M. Heers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.COR.2020.04.11
Author Info
Hayley M. Heers Lida Mina Shakeela W Bahadur Shannon Lim
Corresponding Author
Hayley M. HeersPGY2 Oncology Pharmacy Resident, Banner MD Anderson Cancer Center, Arizona, USA
Figures & Tables
AC: Doxorubicin, Cyclophosphamide; ALND: Axillary Lymph Node Dissection; ER: Estrogen Receptor; IDC: Invasive Ductal Carcinoma; PR: Progesterone Receptor.
ALND: Axillary Lymph Node Dissection; ER: Estrogen Receptor; FEC: 5-Fluorouracil/Epirubicin/Cyclophosphamide; IDC: Invasive Ductal Carcinoma; PR: Progesterone Receptor.
a = Exact timing unclear given patient treated at outside facility during this timeframe.
References
- Breast Cancer: National Comprehensive Cancer Network (NCCN) (2019) Clinical Practice Guidelines in Oncology Version 3.2019.
- Visvanathan K, Fabian CJ, Bantug E, Brewster AM, Davidson NE et al. (2019) Use of Endocrine Therapy for Breast Cancer Risk Reduction: ASCO Clinical Practice Guideline Update. J Clin Oncol 37: 3152-3165. [Crossref]
- Amir E, Seruga B, Niraula S, Carlsson L, Ocana A (2011) Toxicity of adjuvant endocrine therapy in postmenopausal breast cancer patients: a systematic review and meta-analysis. J Natl Cancer Inst 103: 1299-1309. [Crossref]
- Tamoxifen [prescribing information]: Fort Lauderdale (2015) FL: Actavis Laboratories FL.
- Fisher B, Dignam J, Bryant J, Wolmark N (2001) Five versus more than five years of tamoxifen for lymph node-negative breast cancer: updated findings from the National Surgical Adjuvant Breast and Bowel Project B-14 randomized trial. J Natl Cancer Inst 93: 684-690. [Crossref]
- Rabaglio M, Ruepp B, Soft/Text/Perche Steering Committee (2010) Death due to liver failure during endocrine therapy for premenopausal breast cancer. Acta Oncol 49: 874-876. [Crossref]
- Lin Y, Liu J, Zhang X, Li L, Hu R et al. (2014) A prospective, randomized study on hepatotoxicity of anastrozole compared with tamoxifen in women with cancer. Cancer Sci 105: 1182-1188. [Crossref]
- Floren LC, Herbert MF, Venook AP, Jordan VC, Cisneros A et al. (1998) Tamoxifen in liver disease: potential exacerbation of hepatic dysfunction. Ann Oncol 9: 1123-1126. [Crossref]
- Farrell GC (2002) Drugs and steatohepatitis. Semin Liver Dis 22: 185-194. [Crossref]
- Maruyama S, Hirayama C, Abe J, Tanaka J, Matsui K (1995) Chronic active hepatitis and liver cirrhosis in association with combined tamoxifen/tegafur adjuvant therapy. Dig Dis Sci 40: 2602-2607. [Crossref]
- Murata Y, Ogawa Y, Saibara T, Nishioka A, Fujiwara Y et al. (2000) Unrecognized hepatic steatosis and non-alcoholic steatohepatitis in adjuvant tamoxifen for breast cancer patients. Oncol Rep 7: 1299-1304. [Crossref]
- Fox P, Balleine RL, Lee C, Gao B, Balakrishnar B et al. (2016) Dose Escalation of Tamoxifen in Patients with Low Endoxifen Level: Evidence for Therapeutic Drug Monitoring-The TADE Study. Clin Cancer Res 22: 3164-3171. [Crossref]
